The Essential Fuel: How dNTPs Power PCR Amplification for Research and Diagnostics
This comprehensive guide explores the critical, multifaceted roles of deoxynucleoside triphosphates (dNTPs) in Polymerase Chain Reaction (PCR), tailored for researchers, scientists, and drug development professionals.
The Essential Fuel: How dNTPs Power PCR Amplification for Research and Diagnostics
Abstract
This comprehensive guide explores the critical, multifaceted roles of deoxynucleoside triphosphates (dNTPs) in Polymerase Chain Reaction (PCR), tailored for researchers, scientists, and drug development professionals. The article details the foundational biochemistry of dNTP incorporation, examines best practices for their use in various PCR methodologies (including qPCR and multiplex PCR), provides targeted troubleshooting strategies for common reaction failures linked to dNTPs, and offers a comparative analysis of commercial sources and novel analogs. The goal is to empower practitioners with the knowledge to optimize dNTP usage for maximum yield, fidelity, and efficiency in biomedical research and clinical assay development.
The Building Blocks of Replication: A Deep Dive into dNTP Biochemistry and PCR Fundamentals
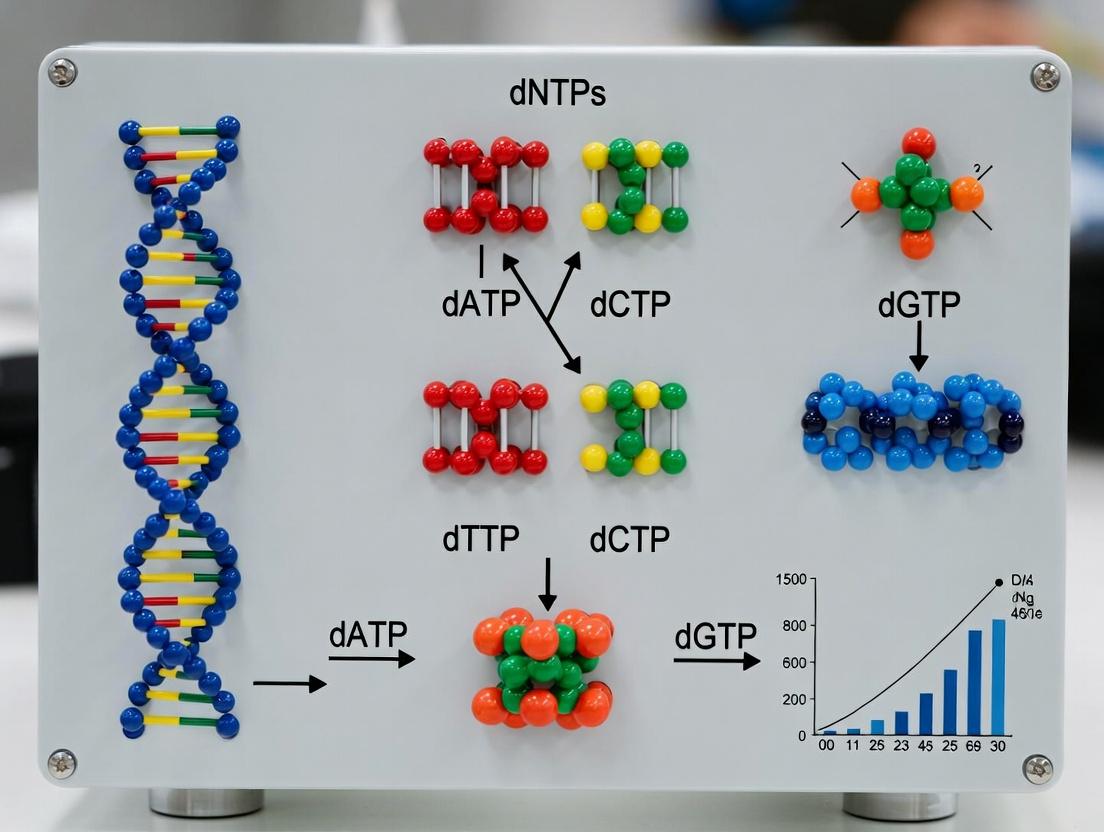
Deoxynucleotide triphosphates (dNTPs) are the essential molecular building blocks for DNA synthesis. In the context of Polymerase Chain Reaction (PCR) amplification research, they serve as the substrates for DNA polymerase, enabling the exponential, template-directed amplification of specific DNA sequences. The efficiency and fidelity of PCR are fundamentally governed by the availability, quality, and precise incorporation of dNTPs (dATP, dTTP, dCTP, and dGTP). This whitepaper provides an in-depth technical analysis of dNTP chemical structure, their function in DNA polymerization, and the critical role of phosphoanhydride bond energy in driving nucleic acid synthesis, with a focus on applications in modern molecular research and drug development.
Chemical Structure of dNTPs
A dNTP is composed of three key moieties:
- A Nitrogenous Base: A purine (adenine or guanine) or pyrimidine (cytosine or thymine).
- A Deoxyribose Sugar: A 5-carbon sugar where the 2' carbon lacks a hydroxyl group (distinguishing it from ribose in NTPs).
- A Triphosphate Chain: Three phosphate groups linked sequentially by phosphoanhydride bonds.
The structure is denoted as dNTP (where "N" represents any of the four bases) and the general molecular formula is C₁₀H₁₂N₅O₁₂P₃⁴⁻ (for dATP) or its analogs. The triphosphate chain is attached to the 5' carbon of the deoxyribose sugar via a phosphoester bond.
Table 1: Structural Components of Standard dNTPs
| dNTP | Nitrogenous Base | Base Type | Molecular Formula | Molecular Weight (g/mol) |
|---|---|---|---|---|
| dATP | Adenine | Purine | C₁₀H₁₂N₅O₁₂P₃ | 491.2 |
| dTTP | Thymine | Pyrimidine | C₁₀H₁₇N₂O₁₄P₃ | 482.2 |
| dCTP | Cytosine | Pyrimidine | C₉H₁₂N₃O₁₃P₃ | 467.2 |
| dGTP | Guanine | Purine | C₁₀H₁₂N₅O₁₃P₃ | 507.2 |
Visual Representation of dNTP Structure:
Function in DNA Synthesis and the Role of Phosphoanhydride Bond Energy
The primary function of dNTPs is to serve as substrates for DNA polymerases during DNA replication, repair, and PCR amplification. The mechanism is energy-driven:
- Nucleophilic Attack: The 3'-OH group of the primer strand attacks the α-phosphate of the incoming complementary dNTP.
- Phosphoanhydride Bond Cleavage: The bond between the α- and β-phosphates is cleaved (a hydrolysis reaction).
- Dimer Formation: A phosphodiester bond is formed between the primer and the dNTP's deoxyribose, with the release of pyrophosphate (PPi: β- and γ-phosphates).
The high free energy of hydrolysis (ΔG°') of the phosphoanhydride bonds (-7 to -8 kcal/mol per bond under cellular conditions) provides the thermodynamic driving force for the otherwise endergonic phosphodiester bond formation. The subsequent hydrolysis of the released pyrophosphate by inorganic pyrophosphatase further drives the reaction to completion.
Table 2: Thermodynamic and Kinetic Parameters of dNTP Incorporation in PCR
| Parameter | Typical Value / Range | Functional Significance in PCR |
|---|---|---|
| ΔG°' of Phosphoanhydride Hydrolysis | ~ -7.6 kcal/mol | Provides energy for phosphodiester bond formation. |
| Optimal dNTP Concentration | 200 µM each (standard PCR) | Balance between yield, fidelity, and speed. Too low: poor yield; Too high: reduced fidelity. |
| Kₘ (DNA Polymerase for dNTPs) | 10 - 50 µM | Affinity of polymerase for dNTP substrates. Influences optimal concentration. |
| Misincorporation Rate (Taq Polymerase) | ~ 1 x 10⁻⁴ to 1 x 10⁻⁵ | Error rate per base pair; dependent on dNTP concentration, [Mg²⁺], and buffer conditions. |
Experimental Protocol: Optimizing dNTP Concentration for High-Fidelity PCR
Objective: To determine the optimal dNTP concentration that maximizes yield while maintaining high fidelity for a specific DNA polymerase blend (e.g., a high-fidelity mix containing a proofreading enzyme).
Methodology:
- Reagent Setup: Prepare a master mix containing buffer, MgCl₂ (typically 1.5 - 2.5 mM final), forward/reverse primers (0.2 - 0.5 µM each), DNA template (10² - 10⁵ copies), and polymerase blend (0.02 - 0.05 U/µL).
- dNTP Titration: Aliquot the master mix into 8 PCR tubes. Spike each tube with a dNTP mix (equimolar dATP, dTTP, dCTP, dGTP) to achieve final concentrations of: 50, 100, 200, 400, 600, 800, 1000, and 1500 µM total dNTP (i.e., 12.5 - 375 µM each).
- PCR Amplification: Run the thermocycling profile appropriate for the primers and template (e.g., 98°C initial denaturation; 30 cycles of 98°C/10s, 60°C/30s, 72°C/30s/kb; final extension 72°C/2min).
- Yield Analysis: Run 5 µL of each product on an agarose gel. Quantify band intensity with a gel imaging system. Plot yield (relative intensity) vs. dNTP concentration.
- Fidelity Analysis (via Sequencing): Purify the PCR products from the 200 µM, 400 µM, and 800 µM reactions. Clone into a sequencing vector (e.g., TA cloning). Sequence 10-20 clones per condition. Calculate error rate (mutations/bp/duplication) by comparing to the known template sequence.
The Scientist's Toolkit: Key Reagents for dNTP/PCR Experiments
| Reagent / Material | Function / Role | Example Product/Note |
|---|---|---|
| Ultra-Pure dNTP Set | Provides high-quality, nuclease-free substrates for DNA synthesis. Reduces non-specific amplification. | Thermo Scientific, NEB, Sigma-Aldrich. Supplied as neutral pH solutions. |
| High-Fidelity DNA Polymerase Blend | Enzyme mix with proofreading (3'→5' exonuclease) activity for low error rates. Essential for cloning. | Q5 High-Fidelity (NEB), Phusion (Thermo), Pfu-based blends. |
| MgCl₂ Solution | Essential cofactor for polymerase activity. Concentration critically influences fidelity and primer annealing. | Often included with PCR buffer. Titration is key for optimization. |
| dNTP Analogs (e.g., dUTP) | Used in PCR for carry-over contamination prevention (with uracil-DNA glycosylase). | Replaces dTTP in the mix. |
| PCR Clean-Up / Cloning Kit | For purifying amplification products prior to sequencing or downstream applications. | Qiagen, Macherey-Nagel, or Zymo Research kits. |
| Sanger Sequencing Service/Kit | Gold standard for verifying sequence and quantifying point mutation rates. | Eurofins, Genewiz, or plasmid-based sequencing kits. |
Advanced Applications and Modifications in Research
Modified dNTPs are crucial tools in advanced research and drug development:
- Labeled dNTPs (e.g., Cy5-dUTP, Digoxigenin-dUTP): For generating probes for FISH, microarray, or detection assays.
- Chain-Terminating dNTPs (ddNTPs): Used in Sanger sequencing, where lack of a 3'-OH halts elongation.
- Therapeutic Analogs: Drugs like Acyclovir (antiviral) or Cytarabine (chemotherapeutic) are nucleoside analogs that, once phosphorylated, inhibit viral or cellular DNA polymerases after incorporation.
dNTPs are far more than simple reagents; they are the energized fuel for the DNA synthesis machinery. Their defined chemical structure, particularly the high-energy phosphoanhydride bonds, is directly harnessed by DNA polymerases to build DNA strands with high precision. In PCR amplification research, understanding the quantitative aspects of dNTP concentration, kinetics, and energy utilization is critical for optimizing assays for yield, speed, and—most importantly for drug development and genetic analysis—fidelity. Mastery of dNTP chemistry and function remains a cornerstone of molecular biology, enabling everything from basic gene cloning to cutting-edge diagnostic and therapeutic development.
The fidelity, efficiency, and specificity of Polymerase Chain Reaction (PCR) amplification are fundamentally governed by the quality and function of its foundational substrates: the deoxyribonucleoside triphosphates (dNTPs). This whitepaper posits that a comprehensive understanding of dNTPs—from their biosynthetic origins within the cell (the Central Dogma's precursor pool) to their optimized use as PCR substrates—is critical for advancing high-fidelity amplification techniques essential for modern genomics, diagnostics, and drug development. The precise concentration, purity, and stability of dATP, dTTP, dCTP, and dGTP directly influence error rates, yield, and the detection of rare genetic variants.
The Biochemical Pathway: Cellular dNTP Synthesis
dNTPs are not primary substrates but are synthesized through a complex, regulated network originating from ribonucleotides.
Key Pathway: The de novo synthesis pathway involves the reduction of ribonucleoside diphosphates (NDPs) to deoxyribonucleoside diphosphates (dNDPs) by the enzyme ribonucleotide reductase (RNR). dNDPs are then phosphorylated to dNTPs. Salvage pathways recycle deoxynucleosides.
Diagram 1: Cellular dNTP Synthesis & Regulation Pathway (100 chars)
dNTPs as PCR Substrates: Quantitative Parameters
The following table summarizes critical quantitative parameters for dNTPs in standard and specialized PCR applications, derived from current manufacturer specifications and recent literature.
Table 1: Optimal dNTP Parameters for PCR Amplification
| Parameter | Standard PCR | High-Fidelity PCR | Long-Range PCR | Notes/Source |
|---|---|---|---|---|
| Final Concentration (each dNTP) | 200 µM | 200 - 250 µM | 350 - 500 µM | Excess can inhibit Taq; too low reduces yield. |
| Equimolar Ratio | Critical | Critical | Critical | Imbalances increase misincorporation rate. |
| Recommended Stock Purity | >99% | >99.5% (HPLC-purified) | >99.5% (HPLC-purified) | Reduces PCR artifacts from contaminants. |
| pH (in stock solution) | 7.0 - 7.5 | 7.0 - 7.5 | 7.0 - 7.5 | Ensures stability and prevents Mg²⁺ chelation. |
| Mg²⁺:dNTP Ratio | ~0.7-1.0:1 (mol/mol) | ~0.7-1.0:1 (mol/mol) | Adjustable | Free Mg²⁺ is a cofactor; dNTPs compete for Mg²⁺. |
| Typical Error Rate Influence | ~1 x 10⁻⁴ (Taq) | ~1 x 10⁻⁶ (High-fidelity enzymes) | Varies | dNTP pool imbalances can increase error rate 10-fold. |
Experimental Protocol: Assessing dNTP Purity and Its Impact on PCR Fidelity
Objective: To evaluate commercial dNTP preparations for contaminating NTPs/rNTPs and measure the subsequent effect on PCR amplicon yield and sequence fidelity.
Materials: See "The Scientist's Toolkit" below.
Methodology:
dNTP Purity Analysis by LC-MS/MS:
- Prepare 1:100 dilutions of dNTP stocks in LC-MS grade water.
- Inject samples onto a reverse-phase C18 column coupled to a tandem mass spectrometer.
- Use a gradient elution with 10mM ammonium acetate (pH 5.3) and methanol.
- Quantify dNTPs and any contaminating NTPs/rNTPs by comparison to pure external standards. Calculate percentage purity.
PCR Amplification with Varied dNTP Sources:
- Design primers for a 2kb target sequence from a standard genomic DNA (e.g., human β-actin).
- Set up identical 50µL reaction mixtures for three dNTP sources: (A) Standard grade, (B) HPLC-purified grade, (C) HPLC-purified with deliberate 0.1% rNTP spike.
- Master Mix per reaction: 1X High-fidelity buffer, 1.5mM MgSO₄, 200µM each dNTP (from source A, B, or C), 0.3µM each primer, 50ng template DNA, 1 unit of a high-fidelity DNA polymerase.
- Thermocycling: Initial denaturation: 98°C, 30s; 35 cycles of [98°C, 10s; 62°C, 30s; 72°C, 2 min]; final extension: 72°C, 5 min.
Yield and Fidelity Assessment:
- Yield: Quantify amplicon yield via fluorescent dsDNA assay (e.g., Qubit) and agarose gel electrophoresis.
- Fidelity: Clone 10-20 individual amplicons from each condition into a sequencing vector. Perform Sanger sequencing on individual colonies. Align sequences to the reference and calculate mutation frequency.
Diagram 2: dNTP Purity & PCR Fidelity Experimental Workflow (99 chars)
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents for dNTP-Centric PCR Research
| Item | Function & Relevance | Example Vendor/Product Type |
|---|---|---|
| HPLC-Purified dNTPs | Ultra-pure dNTPs free of rNTP/NTP contaminants, essential for high-fidelity and long-range PCR to minimize misincorporation. | Thermo Fisher Scientific UltraPure, NEB dNTP Solution Set |
| dNTP/NTP Analysis Kit | Validated standards and buffers for quantifying dNTP purity and detecting contaminants via LC-MS or enzymatic assays. | Biovision dNTP Assay Kit, Agilent Nucleoside/Nucleotide Standards |
| High-Fidelity DNA Polymerase | Enzyme with 3'→5' exonuclease (proofreading) activity, whose performance is highly sensitive to dNTP quality and concentration. | NEB Q5, Takara PrimeSTAR, KAPA HiFi |
| Mg²⁺/MgSO₄ Solution | Separate, optimized cofactor solution allowing independent titration of Mg²⁺ concentration relative to dNTPs. | Included with most polymerase systems |
| PCR Clean-Up & Cloning Kit | For purifying amplicons away from residual dNTPs/primer dimers before sequencing or cloning for fidelity analysis. | Qiagen QIAquick, Zymo DNA Clean & Concentrator |
| Next-Gen Sequencing Library Prep Kit | For deep sequencing to comprehensively assess PCR-induced error spectra from different dNTP conditions. | Illumina Nextera, Swift Biosciences Accel-NGS |
This technical guide examines the biochemical mechanism of deoxynucleotide triphosphate (dNTP) incorporation by DNA polymerase during DNA replication, with a specific focus on implications for Polymerase Chain Reaction (PCR) amplification fidelity and efficiency. Within the broader thesis on the function of dNTPs in PCR, this paper details the enzymatic catalysis, kinetic proofreading, and experimental analysis of this fundamental process, providing a resource for research and therapeutic development.
DNA polymerases are the engines of DNA replication and PCR, but they require dNTPs (dATP, dTTP, dCTP, dGTP) as the fundamental building blocks and energy source. Each incorporation event involves the nucleophilic attack by the 3'-OH of the primer strand on the α-phosphate of the incoming dNTP, releasing pyrophosphate (PPi) and forming a phosphodiester bond. The fidelity of this process, governed by base pairing and polymerase geometry, is critical for accurate PCR amplification. Imbalances in dNTP pools are a documented source of replication errors and reduced amplification yield.
Molecular Mechanism of dNTP Incorporation
The Catalytic Cycle
The incorporation follows a multi-step mechanism:
- Template Binding: DNA polymerase binds the primed template DNA.
- dNTP Binding: The correct, complementary dNTP enters the active site.
- Conformational Change: A rate-limiting open-to-closed transition in the polymerase fingers domain aligns catalytic residues.
- Chemical Catalysis: A two-metal-ion mechanism facilitates the nucleotidyl transfer reaction.
- Pyrophosphate Release & Translocation: PPi is released, and the polymerase translocates to the next template position.
Proofreading and Fidelity
High-fidelity polymerases (e.g., Taq, Pfu) possess a 3'→5' exonuclease domain that excizes misincorporated nucleotides, a critical checkpoint in PCR for reducing error rates.
Quantitative Analysis of dNTP Incorporation Kinetics
Key kinetic parameters govern incorporation efficiency. The following table summarizes data from recent studies on family A DNA polymerases (e.g., Taq) relevant to PCR.
Table 1: Kinetic Parameters for Correct dNTP Incorporation
| Parameter | Symbol | Typical Value (Correct dNTP) | Typical Value (Incorrect dNTP) | Explanation |
|---|---|---|---|---|
| Michaelis Constant | Km | 1 - 20 µM | 50 - 500 µM | Binding affinity; lower Km indicates tighter binding. |
| Maximal Velocity | kcat | 1 - 50 s-1 | 0.001 - 0.1 s-1 | Catalytic rate constant at saturation. |
| Incorporation Efficiency | kcat/Km | ~105 - 106 M-1s-1 | ~101 - 103 M-1s-1 | Overall measure of substrate specificity. |
| Error Rate (Fidelity) | -- | ~10-4 to 10-5 | -- | Mistakes per nucleotide incorporated (without proofreading). |
Table 2: Impact of dNTP Pool Imbalance on PCR Fidelity
| Condition | Error Rate Increase (Fold) | Primary Mutation Type | Effect on Amplification Yield |
|---|---|---|---|
| Equimolar dNTPs (Baseline) | 1x | -- | Optimal |
| 10-Fold Excess of One dNTP | 5-15x | Transversions | Slight decrease |
| dNTP Limitation (All < 20 µM) | -- | -- | Significant yield reduction, early plateau |
| Mg2+:dNTP Ratio > 8:1 | Increased | Frameshifts/ Mismatches | Variable |
Experimental Protocols for Studying dNTP Incorporation
Pre-steady-state Kinetic Analysis (Stopped-Flow)
This protocol measures the elemental steps of a single dNTP incorporation.
Methodology:
- Solution Preparation:
- Solution A: 50-100 nM polymerase (e.g., Taq) pre-mixed with 5'-32P-labeled DNA primer/template (200 nM) in reaction buffer (50 mM Tris-HCl pH 8.8, 50 mM KCl).
- Solution B: Variable concentrations of a single dNTP (0.5-200 µM) in the same buffer with 10-15 mM MgCl2.
- Rapid Mixing: Solutions A and B are rapidly mixed in a stopped-flow apparatus at 37°C.
- Quenching: Reactions are quenched with 0.5 M EDTA at time points from 5 ms to several seconds.
- Product Analysis: Quenched samples are separated on denaturing polyacrylamide gels. The amount of extended primer is quantified via phosphorimaging.
- Data Fitting: The burst phase (kobs) is plotted against [dNTP] to determine Kd(dNTP) and the maximum rate of incorporation (kpol).
Steady-State Kinetic Analysis
Determines Km and kcat under multiple-turnover conditions.
Methodology:
- Reaction Setup: Set up reactions with excess DNA substrate (primer/template) over polymerase (e.g., 100 nM DNA, 5 nM polymerase).
- Variable Substrate: Vary the concentration of one dNTP (e.g., 1-100 µM) while keeping the other three dNTPs at a saturating level (e.g., 200 µM).
- Initiation & Time Points: Initiate reactions with MgCl2 at 37°C. Aliquot at regular time intervals (e.g., 0, 15, 30, 60, 90 s) into EDTA.
- Analysis: Separate products via gel electrophoresis, quantify, and calculate the initial velocity (v0) in nM/s.
- Michaelis-Menten Fit: Plot v0 vs. [dNTP] and fit to the Michaelis-Menten equation to derive Km and kcat.
Visualization of Mechanisms and Workflows
Title: DNA Polymerase Catalytic Cycle for dNTP Addition
Title: Stopped-Flow Kinetics Assay Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for dNTP Incorporation Studies
| Reagent/Material | Function in Experiment | Key Considerations for PCR Research |
|---|---|---|
| High-Fidelity DNA Polymerase (e.g., Pfu, Q5) | Catalyzes templated dNTP incorporation. Provides proofreading (3'→5' exonuclease) activity for high-fidelity PCR. | Choice affects error rate, yield, and amplicon length. |
| Ultra-Pure dNTP Set (neutral pH) | Provides substrates for DNA synthesis. Quality is critical for fidelity and reproducibility. | Degraded dNTPs (e.g., from freeze-thaw) increase error rates. Stock concentration must be verified. |
| MgCl2 Solution | Essential cofactor for polymerase activity. Mg2+ coordinates the dNTP triphosphate and catalytic residues. | Optimal concentration is template- and polymerase-dependent. The Mg2+:dNTP ratio is crucial. |
| Radioactive [α-32P] dNTP or 5'-32P-labeled Primer | Allows sensitive detection and quantification of incorporated nucleotides or primer extension products. | Essential for gel-based kinetic assays. Requires appropriate safety protocols. |
| Synthetic DNA Oligonucleotides (Primer/Template) | Defines the sequence context for studying dNTP incorporation kinetics. | High-purity, HPLC-purified oligos recommended to avoid artifacts. |
| Rapid Kinetics Instrument (Stopped-Flow) | Enables measurement of fast, pre-steady-state enzymatic reactions (millisecond timescale). | Critical for elucidating the elemental rate constants (kpol, Kd). |
| Thermocycler with Gradient | Standard instrument for PCR amplification studies. Used to test effects of dNTP concentration/balance on yield and fidelity under cycling conditions. | Allows optimization of annealing/extension temperatures. |
| Next-Generation Sequencing (NGS) Kit | For comprehensive analysis of PCR error rates and mutational spectra resulting from dNTP pool manipulations or polymerase variants. | Provides a deep, quantitative measure of fidelity. |
This whitepaper elucidates the critical distinctions between deoxynucleoside triphosphates (dNTPs), nucleoside triphosphates (NTPs), and dideoxynucleoside triphosphates (ddNTPs). The discussion is framed within the broader thesis on the Function of dNTPs in PCR amplification research, where the precise biochemical properties of these molecules govern the efficiency, fidelity, and application of nucleic acid synthesis. For researchers and drug development professionals, a rigorous understanding of these building blocks is fundamental to experimental design and interpretation.
Core Chemical Structures and Functional Roles
The primary distinction lies in the structure of the sugar moiety and the resulting biochemical function.
- NTPs (e.g., ATP, UTP, GTP, CTP): Contain a ribose sugar with hydroxyl groups (-OH) at both the 2' and 3' positions. They are the substrates for RNA polymerases during transcription and other RNA synthesis processes.
- dNTPs (dATP, dTTP, dGTP, dCTP): Contain a deoxyribose sugar, lacking the 2'-OH group (replaced by -H). This absence is crucial for DNA stability. dNTPs are the essential substrates for DNA polymerases during DNA replication, repair, and crucially, PCR amplification.
- ddNTPs (ddATP, ddTTP, ddGTP, ddCTP): Contain a dideoxyribose sugar, lacking hydroxyl groups at both the 2' and 3' positions. This absence prevents the formation of a phosphodiester bond with the next incoming nucleotide, causing chain termination.
Quantitative Comparison Table
Table 1: Structural and Functional Comparison of NTPs, dNTPs, and ddNTPs
| Feature | NTP (Ribonucleotide) | dNTP (Deoxyribonucleotide) | ddNTP (Dideoxyribonucleotide) |
|---|---|---|---|
| Sugar Molecule | Ribose | 2'-Deoxyribose | 2',3'-Dideoxyribose |
| 2' Carbon | -OH (Hydroxyl) | -H (Hydrogen) | -H (Hydrogen) |
| 3' Carbon | -OH (Hydroxyl) | -OH (Hydroxyl) | -H (Hydrogen) |
| Primary Polymerase | RNA Polymerase | DNA Polymerase | DNA Polymerase (terminator) |
| Function | RNA synthesis | DNA synthesis & amplification (PCR) | Chain termination (Sanger sequencing) |
| Key Application | Transcription, in vitro transcription | PCR, cDNA synthesis, DNA cloning | Sanger sequencing, fragment analysis |
The Central Role of dNTPs in PCR Amplification Research
Within PCR, dNTPs serve as the fundamental building blocks. The thesis on their function encompasses:
- Substrate Saturation: Optimal and balanced dNTP concentration is critical for amplification efficiency and fidelity. Insufficient dNTPs lead to premature termination, while excessive concentrations can increase error rate by reducing polymerase specificity.
- Fidelity Impact: The choice of DNA polymerase interacts with dNTP pool composition. High-fidelity polymerases often have optimized binding pockets that discriminate against incorrect dNTPs, but an imbalanced dNTP pool can still promote misincorporation.
- Modified dNTPs: Research extends to modified dNTPs (e.g., biotinylated, digoxigenin-labeled, or fluorescently tagged dNTPs) for applications like probe generation, labeling, and next-generation sequencing library prep.
Experimental Protocol: Optimizing dNTP Concentration for High-Fidelity PCR
Objective: To determine the optimal dNTP concentration for maximum yield and fidelity in a specific PCR assay.
Methodology:
- Reaction Setup: Prepare a master mix containing: 1X high-fidelity polymerase buffer, 0.3 µM each primer, 1.5 U of a high-fidelity DNA polymerase (e.g., Pfu, Q5), and a fixed amount of template DNA (e.g., 10-100 ng genomic DNA).
- dNTP Titration: Aliquot the master mix into 8 PCR tubes. Add dNTPs from a pre-mixed stock to final concentrations of: 50 µM, 100 µM, 200 µM (standard), 300 µM, 400 µM, 500 µM, 750 µM, and 1000 µM per dNTP.
- PCR Cycling: Run the following thermocycling protocol: Initial denaturation: 98°C for 30 sec; 30 cycles of [98°C for 10 sec, 60-72°C (primer-specific) for 20 sec, 72°C for 30 sec/kb]; Final extension: 72°C for 2 min.
- Yield Analysis: Analyze 5 µL of each product by agarose gel electrophoresis (1-2%) stained with SYBR Safe. Quantify band intensity using gel documentation software.
- Fidelity Analysis (Optional): Clone the PCR products from the 200 µM, 400 µM, and 800 µM reactions into a TA or blunt-end cloning vector. Sequence 10-20 colonies per condition and calculate the error rate (mutations/bp/duplication).
Experimental Workflow Diagram
Diagram 1: dNTP Optimization Workflow
Chain Termination: The Mechanism of ddNTPs in Sequencing
The function of ddNTPs is defined by their role in Sanger sequencing. When a ddNTP is incorporated by DNA polymerase, extension ceases. Modern sequencing uses fluorescently labeled ddNTPs or primers.
Signaling Pathway: ddNTP Incorporation & Chain Termination
Diagram 2: Chain Termination by ddNTP
Experimental Protocol: Sanger Sequencing Reaction
Objective: To perform a standard cycle sequencing reaction.
Methodology:
- Reagent Mix: In a thin-walled PCR tube, combine:
- Template DNA (PCR product/plasmid): 50-100 ng.
- Sequencing Primer: 3.2 pmol (1 µL of 3.2 µM stock).
- Sequencing Mix (contains buffer, dNTPs, ddNTPs, dye-labeled terminators): 4 µL.
- Sterile water to a final volume of 10 µL.
- Thermal Cycling: Place in a thermocycler. Program: 96°C for 1 min; then 25 cycles of [96°C for 10 sec, 50°C for 5 sec, 60°C for 4 min]; Hold at 4°C.
- Cleanup: Post-cycling, add 10 µL of sterile water and 10 µL of 125 mM EDTA to each tube. Alternatively, use a column- or bead-based purification kit to remove unincorporated terminators.
- Analysis: Resuspend purified product in formamide loading buffer. Denature at 95°C for 5 mins, then analyze by capillary electrophoresis on a sequencer.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Nucleotide-Based Experiments
| Reagent / Material | Function in Research | Key Consideration for Use |
|---|---|---|
| Ultra-pure dNTP Mix (100 mM) | Provides balanced, equimolar substrates for high-efficiency PCR and DNA synthesis. | Aliquot to avoid freeze-thaw cycles; use at 200 µM (final) as a starting point for PCR optimization. |
| High-Fidelity DNA Polymerase (e.g., Q5, Pfu) | Catalyzes DNA synthesis with low error rates, essential for cloning and expression constructs. | Often requires specific buffers; check Mg2+ and dNTP concentration compatibility. |
| Modified dNTPs (e.g., Cy3-dUTP) | Incorporates labels for detection, enabling FISH, microarray labeling, and probe generation. | Incorporation efficiency varies by polymerase; may require adjusted ratios with natural dNTPs. |
| ddNTP Sequencing Mixes | Pre-mixed solutions containing dNTPs and fluorescently labeled ddNTPs for Sanger sequencing. | Kit-specific; optimized for cycle sequencing protocols and specific capillary electrophoresis systems. |
| dNTP Removal Kit | Cleans PCR products or enzymatic reactions of excess dNTPs prior to downstream steps (e.g., sequencing, ligation). | Critical for preventing interference in subsequent enzymatic steps. |
| NTP Set for in vitro Transcription | Provides pure ATP, UTP, GTP, and CTP for synthesizing RNA transcripts for studies in RNA biology or therapeutics. | Must be RNase-free; often used with T7, SP6, or T3 RNA polymerases. |
Within the broader thesis on the function of deoxynucleotide triphosphates (dNTPs) in PCR amplification research, it is imperative to dissect the critical parameters governing their use in standard PCR formulations. dNTPs serve as the fundamental building blocks for DNA polymerase, and their concentration, balance (relative molar ratios), and purity are non-negotiable determinants of amplification efficiency, fidelity, and specificity. This technical guide provides an in-depth analysis of these parameters, underpinned by current experimental data and methodologies.
dNTPs (dATP, dTTP, dCTP, dGTP) are substrates for DNA polymerase during the extension phase of PCR. Their incorporation follows base-pairing rules, and their physicochemical properties directly influence the reaction kinetics. Deviations from optimal parameters can lead to reduced yield, increased error rates, primer-dimer formation, and even complete amplification failure.
Quantitative Analysis of Critical Parameters
Table 1: Optimal Ranges and Effects of Deviations for dNTP Parameters in Standard PCR
| Parameter | Optimal Range (Standard PCR) | Effect of Low Value | Effect of High Value |
|---|---|---|---|
| Total dNTP Concentration | 200 - 500 µM (each dNTP) | Reduced yield, early plateau | Increased error rate, inhibition of Taq polymerase, non-specific amplification |
| dNTP Balance (Equimolar) | 1:1:1:1 molar ratio | Bias in nucleotide composition, sequence-dependent amplification failure | Depletion of limiting dNTP halts synthesis, favors misincorporation |
| Purity (HPLC-grade) | ≥ 99% purity, free of pyrophosphates | Reduced efficiency due to inhibitors (e.g., dNDPs, salts) | N/A; high purity is always the goal |
Table 2: Impact of dNTP Imbalance on PCR Fidelity (Error Rate)
| Imbalance Scenario (Relative Concentration) | Estimated Error Rate (per bp per duplication) | Primary Type of Mutation Induced |
|---|---|---|
| Balanced (1:1:1:1 Control) | ~1 x 10⁻⁵ | Baseline substitution |
| dCTP or dGTP in 5-fold excess | ~1 x 10⁻⁴ | C→T or G→A transitions |
| dTTP in 5-fold excess | ~1 x 10⁻⁴ | A→G transitions |
| All dNTPs at 10-fold optimal | ~1 x 10⁻³ | Multiple transitions & transversions |
Experimental Protocols for Parameter Validation
Protocol 1: Titration of Total dNTP Concentration
Objective: To determine the optimal total dNTP concentration for a specific amplicon and polymerase system.
- Master Mix Preparation: Prepare a series of 50 µL PCR reactions with a fixed concentration of template (10 ng genomic DNA), primers (0.2 µM each), buffer (1X), and Taq DNA polymerase (1.25 U). Vary only the dNTP mix.
- dNTP Gradient: Create a dNTP stock series where each dNTP is at: 50 µM, 100 µM, 200 µM, 400 µM, 800 µM.
- Cycling Conditions: Use standard cycling: Initial denaturation (95°C, 2 min); 30 cycles of denaturation (95°C, 30 s), annealing (Tm-5°C, 30 s), extension (72°C, 1 min/kb); final extension (72°C, 5 min).
- Analysis: Run 10 µL of each product on a 1.5% agarose gel. Quantify band intensity. Plot yield vs. concentration. The lowest concentration giving maximal yield is optimal.
Protocol 2: Assessing dNTP Balance via Sequencing Fidelity Assay
Objective: To evaluate the impact of dNTP imbalance on polymerase error rate.
- Template and Design: Use a well-characterized, low-complexity plasmid template (e.g., pUC19) and primers amplifying a 1-2 kb region.
- Imbalanced Reactions: Set up four 50 µL reactions with a balanced 200 µM each dNTP control. Set up test reactions where one dNTP is at 50 µM (limiting) while the other three remain at 200 µM.
- Amplification: Use a high-fidelity polymerase (e.g., Pfu) with its recommended buffer. Use 20 cycles to avoid jackpot mutations.
- Cloning & Sequencing: Clone the PCR products into a blunt-end vector. Transform competent E. coli. Pick 20-50 colonies per condition and Sanger sequence the entire insert.
- Data Calculation: Align sequences to the known template. Count mismatches. Calculate error rate = (total mismatches) / (total bp sequenced).
Protocol 3: Testing dNTP Purity by Spiking Recovery Experiment
Objective: To detect the presence of inhibitors in a dNTP stock.
- Setup: Prepare two identical "master" PCR mixes with all components except dNTPs, known to work for a robust 500 bp amplicon.
- Spike Test: To mix A, add the dNTP stock under test (200 µM each final). To mix B, add a fresh, certified pure dNTP stock (200 µM each final).
- Inhibition Control: Create mix C by adding 90% of the certified pure dNTPs from B and 10% of the test dNTPs from A.
- Amplification & Analysis: Run PCR. Compare yields on an agarose gel. A significant drop in yield in Mix A or C versus Mix B indicates the presence of inhibitors (e.g., pyrophosphate) in the test stock.
Visualization of dNTP Function and Parameter Impact
Diagram Title: dNTP Role in PCR Cycle and Governing Parameters
Diagram Title: Impact of Sub-Optimal dNTPs on PCR Outcomes
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for dNTP-Centric PCR Research
| Reagent / Solution | Function & Importance in dNTP Research |
|---|---|
| Ultrapure, HPLC-verified dNTP Set | Provides the foundational substrates with guaranteed chemical purity (>99%), absence of contaminants (e.g., pyrophosphate, salts), and accurate concentration, ensuring reproducible kinetics. |
| Individual dNTP Stocks (100 mM) | Allows for precise creation of custom-balanced or imbalanced dNTP mixes to study nucleotide bias, fidelity, and for optimization of difficult targets (e.g., high GC-content). |
| dNTP Neutralization Buffer (for storage) | Typically 10 mM Tris-HCl, pH 7.5-8.0. Maintains dNTP stability by preventing acid-catalyzed hydrolysis during long-term storage at -20°C. |
| MgCl₂ Solution (25-50 mM) | Mg²⁺ is a crucial cofactor for polymerase activity and forms the active complex with dNTPs. Its concentration must be optimized relative to total dNTP concentration. |
| Thermostable DNA Polymerase (Standard & High-Fidelity) | Taq for robustness; high-fidelity enzymes (e.g., Pfu, Q5) for fidelity studies. Different polymerases have varying affinities (Km) for dNTPs, affecting optimal concentration. |
| ddNTPs (for sequencing control) | Used in Sanger sequencing to verify PCR product sequence and directly measure error rates resulting from dNTP imbalance or impurity in fidelity assays. |
| PCR Clean-up / Gel Extraction Kit | Essential for purifying amplification products away from residual dNTPs and primers before downstream applications like cloning or sequencing, which are sensitive to these leftovers. |
The concentration, balance, and purity of dNTPs are interdependent pillars of a successful PCR formulation. As detailed in this guide, deviation from optimal ranges has quantifiable and often detrimental effects on amplification success and data integrity. Within the thesis of dNTP function, mastering these parameters is not merely a procedural step but a fundamental exercise in controlling the biochemical environment to ensure the reliability of genetic data—a non-negotiable standard for research and drug development.
Precision in Practice: Optimizing dNTP Use for Advanced PCR Applications and Techniques
Within the broader thesis on the Function of dNTPs in PCR Amplification Research, establishing optimal deoxynucleotide triphosphate (dNTP) concentrations is a cornerstone of robust assay development. dNTPs serve as the essential building blocks for DNA synthesis, and their concentration directly impacts PCR efficiency, fidelity, specificity, and yield. Imbalanced or suboptimal dNTP levels can lead to reduced amplification efficiency, increased error rates due to polymerase misincorporation, and primer-dimer formation. This technical guide provides an in-depth protocol for determining the optimal dNTP concentration range and integrating it into a reliable Master Mix design for research and diagnostic applications.
dNTPs (dATP, dTTP, dCTP, dGTP) are substrates for DNA polymerase. Each dNTP is incorporated into the nascent DNA strand complementary to the template. Key functional considerations include:
- Concentration & Balance: Equimolar concentrations of all four dNTPs are typically required to prevent polymerase errors and stalling.
- Interplay with Mg²⁺: Mg²⁺ is a critical cofactor for polymerase activity and forms a complex with dNTPs. The total dNTP concentration directly influences the free Mg²⁺ available in the reaction, which must be optimized separately.
- Impact on Fidelity: High dNTP concentrations can reduce polymerase fidelity by promoting misincorporation, while very low concentrations can limit yield and efficiency.
Experimental Protocol: Determining Optimal dNTP Concentration
Objective
To empirically determine the optimal concentration range for a standard dNTP mixture that maximizes PCR product yield and specificity for a given primer-template system and polymerase.
Materials & Master Mix Preparation
A standard 50 µL reaction is used as a basis. Prepare a 2X concentrated Master Mix to minimize pipetting errors. The variable component will be the dNTP solution.
Table 1: Master Mix Components (2X Concentrate)
| Component | Final 1X Concentration | Volume in 2X Master Mix (per reaction) | Function & Notes |
|---|---|---|---|
| PCR Buffer (10X) | 1X | 10 µL | Provides optimal pH, ionic strength. Often includes MgCl₂. |
| Forward Primer | 0.2 - 1.0 µM | Variable | Target-specific oligonucleotide. |
| Reverse Primer | 0.2 - 1.0 µM | Variable | Target-specific oligonucleotide. |
| DNA Polymerase | 0.5 - 2.5 U/50µL | Variable | Thermostable enzyme (e.g., Taq, high-fidelity). |
| Template DNA | Variable (e.g., 10-100 ng) | Variable | Genomic DNA, cDNA, or plasmid. |
| dNTP Mix | Variable (Test Range) | Variable | The target of optimization. |
| MgCl₂ (if not in buffer) | 1.0 - 4.0 mM (optimize) | Variable | Essential cofactor. Concentration depends on dNTP level. |
| Nuclease-free Water | To volume | To volume | Solvent. |
Step-by-Step Procedure
- Design the dNTP Concentration Gradient: Prepare a dNTP dilution series. A standard starting range is 50 µM to 1000 µM for each dNTP (i.e., 200 µM to 4000 µM for total dNTPs). Suggested test points: 50, 100, 200, 400, 600, 800, 1000 µM per dNTP.
- Prepare the Master Mix (without dNTPs): For 'n' test reactions + 10% extra, combine PCR buffer, primers, polymerase, water, and MgCl₂ (if required). Mix gently by vortexing and brief centrifugation.
- Aliquot the Master Mix: Dispense equal volumes (e.g., 45 µL if the final dNTP addition is 5 µL) into each PCR tube.
- Add Variable dNTP: Add the corresponding volume of each dNTP stock from the gradient series to each tube.
- Add Template DNA: Add a constant amount of template DNA to each tube. Include a no-template control (NTC) using a mid-range dNTP concentration.
- Run PCR: Use standard cycling conditions appropriate for the primer pair and polymerase.
- Analyze Results: Separate PCR products by agarose gel electrophoresis. Analyze for:
- Yield: Band intensity of the target product.
- Specificity: Presence/absence of non-specific bands or primer-dimers.
- Fidelity: May require sequencing of products from extremes of the range.
Data Analysis and Interpretation
Quantify gel band intensities using densitometry software. Plot relative yield versus dNTP concentration.
Table 2: Example Experimental Results for dNTP Optimization
| dNTP Concentration (each, µM) | Total dNTP (µM) | Relative Product Yield (%) | Specificity Score (1-5)* | Notes |
|---|---|---|---|---|
| 50 | 200 | 15 | 5 | Very clean, low yield. |
| 100 | 400 | 65 | 5 | High specificity, good yield. |
| 200 | 800 | 100 | 4 | Optimal yield, slight non-specific bands. |
| 400 | 1600 | 95 | 3 | High yield, increased background. |
| 600 | 2400 | 80 | 2 | Reduced yield, significant non-specific amplification. |
| 800 | 3200 | 50 | 2 | Poor yield and specificity. |
*1 = Low specificity (many spurious bands), 5 = High specificity (single sharp band).
Conclusion from Example Data: The optimal range for this specific assay is 100-200 µM per dNTP, balancing high yield with good specificity. 200 µM provides maximum yield, while 100 µM provides the cleanest reaction.
Master Mix Design Principles with Optimized dNTPs
Once the optimal dNTP concentration is determined, design a stable, ready-to-use Master Mix.
Key Considerations:
- Stability: dNTPs in aqueous solution are susceptible to hydrolysis. Prepare aliquots at -20°C. In a ready-made Master Mix, ensure pH is stabilized.
- Compatibility: The chosen dNTP concentration must be compatible with the recommended Mg²⁺ concentration for the polymerase. As a rule, free Mg²⁺ should be 0.5-2.5 mM above total dNTP concentration.
- Formulation: A standard 2X Master Mix formulation includes: buffer, MgCl₂ (at optimized level), optimized dNTP mix, stabilizers (e.g., glycerol, trehalose), and polymerase. Primers and template are added separately.
Protocol for Preparing a 2X qPCR Master Mix (with SYBR Green)
- Calculate Requirements: Determine total volume needed (number of reactions * volume per reaction + ~20% overage).
- Combine in Order: In a nuclease-free tube on ice, combine:
- Nuclease-free water (to final volume)
- 2X Concentrated Reaction Buffer
- MgCl₂ stock to achieve final optimized concentration (e.g., 3-4 mM final in 1X mix)
- Optimized dNTP Mix (e.g., 400 µM each final in 1X mix)
- SYBR Green I dye (at manufacturer's recommended dilution)
- Passive Reference Dye (e.g., ROX, if required by instrument)
- Hot-start DNA Polymerase (add last, gently mix)
- Mix Gently: Vortex briefly and centrifuge. Aliquot if desired.
- Quality Control: Test the Master Mix against a known standard template and primer set to verify performance (Cq value, amplification efficiency, specificity).
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for dNTP & Master Mix Optimization
| Item | Function in Protocol | Key Considerations |
|---|---|---|
| Ultra-pure dNTP Set (100 mM each) | Provides the nucleotide substrates. Stock solutions for creating precise gradients and master mixes. | Verify pH (~7.0), avoid freeze-thaw cycles, check for degradation. |
| Hot-Start DNA Polymerase | Catalyzes DNA synthesis. Reduces non-specific amplification at low temperatures. | Choose based on fidelity, speed, and template type (e.g., high-fidelity for cloning). |
| 10X PCR Buffer (with/without MgCl₂) | Provides optimal chemical environment (pH, salts) for polymerase activity. | Mg²⁺-free buffers allow independent optimization of Mg²⁺ concentration. |
| 25 mM MgCl₂ Solution | Essential cofactor for polymerase. Concentration is critically dependent on dNTP levels. | Must be titrated in conjunction with the optimized dNTP concentration. |
| Nuclease-free Water | Solvent for all reactions. Prevents degradation of reagents. | Must be PCR-grade to avoid contaminating nucleases. |
| dNTP Compatible Stabilizers (e.g., Trehalose) | Added to ready-made Master Mixes to enhance long-term stability of enzymes and dNTPs. | Reduces freeze-thaw stress and maintains reaction efficiency. |
| Quantitative PCR (qPCR) Dyes | For real-time monitoring of amplification (e.g., SYBR Green, EvaGreen). | Choice affects cost, specificity detection, and multiplexing capability. |
Visualizations
Workflow for dNTP Concentration Optimization
Interaction Between dNTPs and Magnesium Ions
This document serves as an in-depth technical guide on the application-specific optimization of deoxynucleotide triphosphates (dNTPs) within various polymerase chain reaction (PCR) methodologies. It is framed within the broader thesis that the function of dNTPs extends beyond their role as simple building blocks; their concentration, purity, balance, and formulation are critical, modifiable parameters that directly influence the efficiency, fidelity, specificity, and yield of diverse PCR applications. For researchers, scientists, and drug development professionals, mastering these nuances is essential for robust experimental outcomes.
Core Function of dNTPs in PCR
dNTPs (dATP, dTTP, dCTP, dGTP) are the essential substrates for DNA polymerase, providing both the energy and the nucleotides for DNA strand elongation. Key functional parameters include:
- Concentration: Typically 200 μM of each dNTP is standard for routine PCR, but this must be adjusted for specialized applications. Insufficient dNTPs lead to early reaction cessation, while excess can chelate Mg²⁺ (a required cofactor) and reduce polymerase fidelity.
- Balance: Equimolar concentrations of all four dNTPs are crucial to prevent polymerase errors and premature termination.
- Purity: The presence of contaminants like pyrophosphates or nucleotidases can severely inhibit amplification.
- Stability: dNTPs are susceptible to degradation by repeated freeze-thaw cycles or improper storage, impacting reaction consistency.
Application-Specific Formulations and Protocols
Quantitative PCR (qPCR)
qPCR demands high efficiency and linearity over a wide dynamic range. dNTP formulation impacts the kinetics of the reaction and the accuracy of quantification.
- Optimal Concentration: Lower than standard PCR, typically 200-500 μM total dNTPs (50-125 μM each). This minimizes background fluorescence and reagent costs while maintaining efficiency.
- Critical Factor: Use of high-purity, PCR-grade dNTPs free from contaminants that could inhibit the polymerase or quench fluorescence.
- Protocol Snippet: qPCR Optimization with dNTP Titration
- Prepare a master mix containing buffer, MgCl₂ (3-5 mM), hot-start Taq polymerase, SYBR Green I dye, primers, and template.
- Aliquot the master mix into separate tubes.
- Spike each tube with a different dNTP mix to achieve final total dNTP concentrations of: 100 μM, 200 μM, 300 μM, 400 μM, and 500 μM (ensure equimolar ratios).
- Run qPCR with a standardized template dilution series.
- Calculate amplification efficiency (E) from the standard curve slope: E = [10^(-1/slope)] - 1. The ideal dNTP concentration yields E closest to 1 (100%) with the lowest Cq values and highest linearity (R² > 0.99).
Reverse Transcription PCR (RT-PCR)
RT-PCR involves a reverse transcription step to generate cDNA from RNA, followed by PCR. dNTPs are critical for both enzymatic steps.
- Two-Step RT-PCR: The RT and PCR steps are separate. The RT reaction typically uses 500 μM of each dNTP to ensure full-length cDNA synthesis from complex RNA templates.
- One-Step RT-PCR: Combines RT and PCR in a single tube. A compromise dNTP concentration is used (typically 400-600 μM total) to satisfy both enzymes without inhibiting the subsequent PCR. Commercial one-step kits provide optimized, balanced formulations.
- Protocol Snippet: One-Step RT-PCR Setup
- On ice, combine: 1x Reaction Buffer, MgSO₄ (or MgCl₂, as specified), 500 μM of each dNTP, 0.5-1 μM each primer, 1 μL enzyme mix (reverse transcriptase + Taq DNA polymerase), RNase inhibitor, and 1 pg–1 μg RNA template.
- Incubate for reverse transcription: 45–50°C for 10–30 minutes.
- Inactivate RT and activate hot-start polymerase: 94–95°C for 2 minutes.
- Perform 35–40 cycles of PCR.
- Analyze products by agarose gel electrophoresis.
Long-Range PCR
Amplification of fragments >5 kb requires high-processivity polymerases and optimized dNTP conditions to support continuous, error-free elongation.
- Higher Concentration: Standard is 500-600 μM of each dNTP. This ensures a sufficient local substrate pool for the polymerase during long extension steps.
- Balanced dNTP/Mg²⁺ Ratio: The increased dNTP concentration necessitates a proportional increase in Mg²⁺ concentration (often 2.0-2.5 mM) to prevent chelation and ensure polymerase activity.
- Stability: Use of stabilized dNTP solutions (e.g., mixed with Tris buffer) is recommended to prevent degradation during long cycle times.
- Protocol Snippet: Long-Range PCR Amplification
- Use a specialized long-range PCR enzyme blend (e.g., mixture of Taq and a proofreading polymerase).
- Prepare a 50 μL reaction: 1x proprietary buffer, 2.2 mM Mg²⁺, 550 μM of each dNTP, 0.3 μM each primer, 2.5 U enzyme blend, and 100–500 ng genomic DNA.
- Use a "hot-start" activation: 94°C for 2 min.
- Cycle: Denature at 94°C for 10 sec, anneal at 60°C for 30 sec, and extend at 68°C for 1 min per kb of product.
- Perform a final extension at 68°C for 7–10 minutes.
Multiplex PCR
The simultaneous amplification of multiple targets requires stringent optimization to prevent primer-dimer formation and ensure uniform yield of all amplicons.
- Increased Concentration: Often requires 400-600 μM total dNTPs to support the amplification of multiple templates simultaneously and compete against non-specific interactions.
- Ultra-Pure dNTPs: Essential to minimize baseline noise from any enzymatic inhibition, which is magnified in a complex primer mix.
- Balanced with MgCl₂: Mg²⁺ concentration must be titrated in conjunction with dNTPs. A typical starting point is 2.0-3.0 mM MgCl₂ with 500 μM each dNTP.
- Protocol Snippet: Multiplex PCR Optimization
- Design primers with similar Tm (within 2°C) and minimal complementarity.
- Perform a matrix optimization of MgCl₂ (1.5, 2.0, 2.5, 3.0 mM) and dNTPs (300, 400, 500 μM each).
- Use a hot-start, high-specificity polymerase.
- Use a touchdown or two-step cycling program to enhance specificity: Initial denaturation 95°C, 2 min; 10 cycles of 95°C 30 sec, 65–55°C (-1°C/cycle) 30 sec, 72°C 1 min; then 25 cycles of 95°C 30 sec, 55°C 30 sec, 72°C 1 min.
- Analyze on a high-resolution agarose or capillary electrophoresis system.
Table 1: Recommended dNTP Formulations by PCR Application
| PCR Application | Recommended [dNTP] each (μM) | Total [dNTP] (μM) | Key Rationale | Critical Co-factor Adjustment |
|---|---|---|---|---|
| Standard PCR | 200 | 800 | Balanced for routine yield & specificity. | MgCl₂ ~1.5-2.0 mM |
| Quantitative PCR (qPCR) | 50-125 | 200-500 | Maximizes efficiency, minimizes background/ cost. | MgCl₂ ~3-5 mM (often optimized with dNTPs) |
| RT-PCR (Two-Step) | 500 (RT step) | 2000 | Ensures complete cDNA synthesis. | MgCl₂ per enzyme specs. |
| RT-PCR (One-Step) | 100-150 | 400-600 | Compromise for dual enzyme activity. | Mg²⁺ as per kit formulation. |
| Long-Range PCR | 500-600 | 2000-2400 | Maintains pool for processive polymerases over long extensions. | Increase Mg²⁺ to 2.0-2.5 mM to counter chelation. |
| Multiplex PCR | 100-150 | 400-600 | Supports multiple amplicons, outcompetes non-specific priming. | Titrate MgCl₂ (2.0-3.0 mM) with dNTPs. |
Table 2: Impact of dNTP Imbalance on PCR Fidelity
| dNTP Condition | Error Rate (mutations/bp/duplication) | Primary Effect | Application Most Affected |
|---|---|---|---|
| Perfect Balance (200 μM each) | ~1 x 10⁻⁵ (for standard Taq) | Baseline fidelity. | All, especially cloning & sequencing. |
| One dNTP at 50% (100 μM) | Can increase 2-5 fold | Misincorporation, premature termination. | Long-range PCR, multiplex. |
| One dNTP at 200%, others at 100% | Can increase >10 fold | Severe misincorporation, sequence skew. | qPCR (affects quantification), NGS library prep. |
| With Proofreading Polymerase | ~1 x 10⁻⁶ | 3’→5’ exonuclease corrects errors. | High-fidelity applications (e.g., gene cloning). |
Visualizations
Diagram 1: dNTP Strategy Selection for PCR Types
Diagram 2: One-Step RT-PCR dNTP Utilization
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for dNTP-Optimized PCR
| Reagent/Material | Function & Rationale | Example/Criteria for Selection |
|---|---|---|
| Ultra-Pure, PCR-Grade dNTP Set | Provides the core substrates. High purity is non-negotiable for sensitive applications (qPCR, multiplex) to avoid inhibitors. | Solutions pH-stabilized to 7.0, supplied at 100 mM each, HPLC-purified. |
| MgCl₂ or MgSO₄ Solution | Essential co-factor for polymerase activity. Concentration must be optimized relative to dNTP concentration. | Separate, certified nuclease-free stock solutions (typically 25-50 mM). |
| Thermostable DNA Polymerase | Enzyme that catalyzes synthesis. Choice depends on application (fidelity, processivity, hot-start). | Standard Taq (routine), Blends (long-range), High-Fidelity (cloning), Hot-Start (multiplex, qPCR). |
| Optimized Reaction Buffer | Provides optimal pH, ionic strength, and often includes stabilizers. Commercial buffers are matched to their polymerase. | 10x concentration, often with (NH₄)₂SO₄ or KCl, and sometimes with enhancers like DMSO or betaine. |
| Nuclease-Free Water | Solvent for all reactions. Must be free of nucleases and contaminants to ensure reproducibility. | DEPC-treated and autoclaved or commercially certified nuclease-free water. |
| dNTP/Mg²⁺ Optimization Kit | Facilitates systematic titration of these critical components for challenging applications. | Pre-formulated matrices of dNTP and Mg²⁺ concentrations. |
| PCR Plates/Tubes with Seals | Ensure efficient thermal conductivity and prevent evaporation/cross-contamination. | Thin-walled, skirted plates compatible with thermal cyclers and real-time detectors. |
| Positive Control Template & Primers | Validates the entire reaction setup, including dNTP functionality. | A well-characterized, amplifiable DNA fragment with known primers. |
This whitepaper is a core component of a comprehensive thesis investigating the Function of dNTPs in PCR Amplification Research. While dNTPs are universally recognized as the fundamental building blocks for DNA synthesis, their precise concentrations, ratios, and purity exert a profound, yet often underappreciated, influence on the fidelity of DNA polymerases. This guide elucidates the mechanistic relationship between dNTP pools and replication accuracy, providing a framework for selecting optimal polymerases and reaction conditions to minimize errors in high-stakes applications such as cloning, next-generation sequencing library preparation, and diagnostic assays.
Mechanistic Interplay: dNTP Pools and Polymerase Fidelity
DNA polymerase fidelity is governed by a multi-step kinetic proofreading mechanism. The incorporation of an incorrect nucleotide (mismatch) is less favored due to a slower incorporation rate and a more rapid dissociation rate. The concentration of dNTPs directly impacts this process through the dNTP concentration gradient effect.
- High dNTP Concentrations: Can increase the rate of misincorporation by promoting the binding and incorporation of incorrect nucleotides before the polymerase can reject them. This can saturate the polymerase's exonuclease proofreading activity.
- Balanced dNTP Ratios: Imbalances (e.g., low [dCTP] relative to others) can force the polymerase to incorporate an incorrect nucleotide (e.g., dUTP) more frequently when the correct one is scarce, increasing error rates.
- dNTP Purity: Contaminants like oxidized dNTPs (e.g., 8-oxo-dGTP) are direct substrates for misincorporation, leading to transversion mutations.
Diagram 1: dNTP Influence on Polymerase Fidelity Pathway
Quantitative Data: Polymerase Error Rates and dNTP Modulation
Error rates are typically measured as mutations per base per duplication (error rate) or as a fidelity multiplier relative to a reference. The following table synthesizes current data on high-fidelity polymerases and the impact of dNTP conditions.
Table 1: Fidelity Characteristics of Common High-Fidelity Polymerases
| Polymerase (Commercial Example) | Intrinsic Error Rate (approx.) | Proofreading (3'→5' Exo) | Key Feature | Optimal [dNTP] for Max Fidelity* |
|---|---|---|---|---|
| Q5 High-Fidelity (NEB) | ~2.8 x 10⁻⁷ | Yes | High-processivity, mutant B. cereus Pol | 200 µM each |
| Phusion / Fusion (Thermo) | ~4.4 x 10⁻⁷ | Yes | Chimeric Pfu-like enzyme | 200 µM each |
| Kapa HiFi (Roche) | ~2.6 x 10⁻⁷ | Yes | Pyrococcus-like enzyme, optimized buffer | 200-300 µM each |
| PrimeSTAR GXL (Takara) | ~9.0 x 10⁻⁷ | Yes | Blend with high processivity | 200 µM each |
| Platinum SuperFi II (Invitrogen) | ~1.4 x 10⁻⁷ | Yes | Engineered for ultralow error rates | 200 µM each |
| Taq Wild-Type | ~2.0 x 10⁻⁵ | No | Low-cost, standard PCR | 200-500 µM each |
Note: Optimal [dNTP] is typically vendor-recommended and balances fidelity with yield. Lower concentrations (e.g., 50 µM) may increase fidelity but reduce yield and processivity.
Table 2: Impact of dNTP Conditions on Observed Error Frequency
| Condition Variable | Typical Experimental Range | Effect on Fidelity (Direction) | Mechanistic Rationale | Recommended for High Accuracy |
|---|---|---|---|---|
| Total [dNTP] | 50 µM – 1000 µM each | ↓ at very high concentrations (>500µM) | Increased misincorporation kinetics, proofreading saturation | 100 – 200 µM each |
| dNTP Ratio Imbalance | 1:1:1:1 vs. 10:1:1:1 | ↓↓ with severe imbalance | Depletion of one dNTP forces misincorporation | Strict 1:1:1:1 molar ratio |
| Presence of dUTP | 0 – 200 µM (in carryover prevention) | ↓ (increases A•G mismatches) | dUTP competes with dTTP, pairs with adenine | Use uracil-DNA glycosylase (UDG) with care |
| Oxidized dNTPs (e.g., 8-oxo-dGTP) | Contaminant at >0.1% | ↓↓↓ | Direct incorporation opposite A or C | Use high-purity, stabilized dNTPs, fresh aliquots |
| Mg²⁺:[dNTP] Ratio | Constant Mg²⁺, varying dNTP | ↓ if ratio is incorrect | Alters polymerase active site geometry & fidelity | Optimize Mg²⁺ after setting [dNTP]; aim for ~0.5-1 mM free Mg²⁺ |
Experimental Protocols for Assessing Fidelity
Protocol 1:lacIForward Mutation Assay (Classical Method)
This assay measures the frequency of inactivating mutations in the lacI gene of E. coli after PCR amplification.
- Amplify: PCR amplify the lacI gene (~1.2 kb) from a plasmid template using the polymerase and dNTP conditions under test.
- Clone: Ligate the purified PCR products into a suitable vector and transform into an E. coli host strain competent for alpha-complementation (e.g., lacI⁻ host).
- Plate: Plate transformed cells on media containing X-gal and IPTG. Incubate.
- Score: Wild-type lacI colonies are blue (repressor functional, β-galactosidase suppressed). Mutant lacI colonies are white or light blue (repressor non-functional, β-galactosidase expressed).
- Calculate: Fidelity = (Total plaques / Mutant plaques) / (Number of target bases). Error rate = 1 / Fidelity.
Protocol 2: Next-Generation Sequencing (NGS)-Based Error Rate Analysis
A modern, high-throughput method for direct error quantification.
- Amplify with Barcodes: Perform PCR on a known, error-free template (e.g., plasmid, gBlock) using test conditions. Use primers containing unique dual-index barcodes to identify each product molecule and distinguish PCR errors from sequencing errors.
- Purify & Quantify: Clean up PCR products and quantify precisely.
- Library Prep & Sequencing: Prepare an NGS library (avoiding additional PCR if possible) and sequence on a high-accuracy platform (e.g., Illumina MiSeq) with sufficient coverage (>1000x per original molecule).
- Bioinformatics Analysis:
- Align sequences to the reference template.
- Identify variants, filtering out low-quality calls.
- Use barcode information to cluster reads derived from the same initial molecule. True PCR errors will appear in all reads from that cluster.
- Calculate error rate: (Total confirmed errors) / (Total bases sequenced in consensus reads).
Diagram 2: NGS-Based PCR Fidelity Analysis Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for High-Fidelity PCR Applications
| Reagent / Solution | Function in High-Fidelity Context | Key Considerations for Fidelity |
|---|---|---|
| Ultrapure, Stabilized dNTPs (e.g., PCR Grade) | Provides balanced, clean nucleotide substrates. | Minimizes contaminants (oxo-dGTP, nuclease); ensures consistent 1:1:1:1 ratio; often supplied in pH-stabilized buffers. |
| Proofreading DNA Polymerase (e.g., Q5, Phusion, Kapa HiFi) | Catalyzes DNA synthesis with 3'→5' exonuclease activity to remove mismatches. | Select based on error rate, processivity, and template compatibility (e.g., GC-rich). |
| Optimized High-Fidelity Buffer | Provides optimal ionic strength, pH, and cofactors (Mg²⁺). | Often includes components that enhance polymerase specificity and stability. Mg²⁺ concentration is critical. |
| Betaine or GC Enhancer | Additive to reduce secondary structure in GC-rich templates. | Prevents polymerase stalling, which can indirectly increase error incorporation. |
| DMSO | Additive to lower melting temperature of secondary structure. | Use at low concentrations (<3%); can sometimes affect polymerase fidelity; requires optimization. |
| High-Purity Nuclease-Free Water | Solvent for all reaction components. | Prevents nuclease contamination and unintended ionic changes. |
| Uracil-DNA Glycosylase (UDG) | Enzyme to degrade carryover dU-containing PCR products. | Requires use of dUTP in PCR, which can lower fidelity. Best used in post-amplification cleanup for sensitive applications. |
| PCR Cleanup/Sizing Kits (e.g., bead-based) | Purifies amplicons from unused dNTPs, primers, and polymerase. | Essential for downstream applications like cloning or sequencing. Removes potential error sources. |
Within the broader thesis on the Function of dNTPs in PCR Amplification Research, it is critical to expand beyond canonical deoxynucleoside triphosphates (dNTPs). Standard PCR utilizes dATP, dTTP, dCTP, and dGTP as the fundamental building blocks for enzymatic DNA synthesis. However, the strategic incorporation of modified nucleotides—specialized dNTPs—has revolutionized molecular biology applications. These analogs are chemically altered at the base, sugar, or phosphate moiety, enabling precise manipulation of nucleic acids for advanced cloning strategies, sensitive detection via labeling, and novel sequencing technologies. This guide details the technical principles, protocols, and applications of these indispensable tools.
Classes of Modified dNTPs and Their Core Functions
Modified dNTPs are categorized by their functional modification and application.
A. For Cloning and Manipulation:
- 5-Methyl-dCTP: Incorporates methyl groups at cytosine residues, protecting PCR products from cleavage by restriction enzymes sensitive to CpG methylation.
- dUTP: Used in PCR to produce uracil-containing amplicons. Subsequent treatment with Uracil-DNA Glycosylase (UDG) prevents carryover contamination from previous PCRs, a cornerstone of cloning sterile environments.
- α-Thio-dNTPs (dNTPαS): Feature a sulfur atom replacing a non-bridging oxygen on the α-phosphate. They create nuclease-resistant phosphorothioate bonds, useful for generating strand-specific breaks or protecting linkers.
B. For Labeling and Detection:
- Biotin-dNTPs (e.g., Biotin-16-dUTP): Incorporate biotin, enabling strong non-radioactive detection via streptavidin-enzyme conjugates (alkaline phosphatase, horseradish peroxidase) for blotting, in situ hybridization, and pull-down assays.
- Digoxigenin-dNTPs (e.g., DIG-11-dUTP): Hapten-labeled nucleotides detected with high-affinity anti-digoxigenin antibodies, offering exceptional sensitivity with low background.
- Fluorophore-dNTPs (e.g., Cy3-dUTP, Fluorescein-dCTP): Enable direct fluorescent labeling of DNA for techniques like fluorescent in situ hybridization (FISH), microarray analysis, and next-generation sequencing (NGS) library construction.
C. For Sequencing and Functional Genomics:
- 2',3'-Dideoxynucleotides (ddNTPs): The foundational reagents for Sanger sequencing, lacking the 3'-hydroxyl group to terminate chain elongation.
- Reversibly Terminating dNTPs: Used in cyclic-array sequencing (e.g., Illumina). Each nucleotide contains a cleavable blocker at the 3'-OH and a fluorescent label, allowing single-base incorporation and imaging per cycle.
- 8-Oxo-dGTP: An oxidized guanine derivative used to study oxidative damage and mutagenesis.
Table 1: Key Classes of Specialized dNTPs and Their Properties
| Modification Class | Example dNTP | Key Chemical Feature | Primary Application(s) | Typinal Incorporation Efficiency vs. Wild-Type* |
|---|---|---|---|---|
| Backbone Modification | dATPαS | Phosphorothioate linkage | Nuclease resistance, site-directed mutagenesis | ~80-95% |
| Base Modification (Hapten) | DIG-11-dUTP | Digoxigenin linked to base | Non-radioactive detection (blotting, FISH) | ~70-90% |
| Base Modification (Fluorophore) | Cy5-dCTP | Cy5 dye linked to base | Direct fluorescence, microarray labeling, NGS | ~50-75% |
| Base Modification (Biotin) | Biotin-16-dUTP | Biotin linker on base | Affinity capture, detection, pull-down assays | ~75-90% |
| Sugar Modification | ddTTP | Lacks 3'-OH group | Chain termination (Sanger sequencing) | 100% (terminates) |
| Epigenetic Marker | 5-Methyl-dCTP | Methyl group at C5 of cytosine | Studying methylation, protecting from restriction | ~85-98% |
*Efficiency depends on polymerase, buffer conditions, and ratio to canonical dNTPs. Values are generalized from current literature.
Experimental Protocols
Protocol 1: PCR Incorporation of Hapten-Labeled dNTPs for Southern Blot Detection
Objective: Generate a digoxigenin (DIG)-labeled DNA probe via PCR for high-sensitivity Southern hybridization.
Materials:
- Template DNA (10-100 ng).
- Sequence-specific primers (0.2-1.0 µM each).
- PCR Master Mix: 1x PCR buffer (with Mg²⁺), 0.2 mM each dATP, dCTP, dGTP, 0.13 mM dTTP, 0.07 mM DIG-11-dUTP, 1.25-2.5 U proofreading polymerase (e.g., Pfu).
- Thermal cycler.
Methodology:
- Prepare Reaction Mix: In a 50 µL reaction, combine template, primers, and PCR Master Mix. The dTTP:DIG-11-dUTP ratio is critical (~65:35) to ensure sufficient incorporation while maintaining amplicon yield.
- Amplify: Use standard cycling conditions appropriate for the primer-template system, but extend the elongation time by 20-30% to accommodate the modified nucleotide.
- Purify Probe: Purify the PCR product using a spin column or ethanol precipitation to remove unincorporated DIG-dUTP.
- Hybridize & Detect: Denature the probe and add to hybridization buffer. Perform Southern blot per standard protocol. Detect using anti-DIG-alkaline phosphatase conjugate and chemiluminescent substrate (e.g., CSPD).
Protocol 2: Primer Extension with α-Thio-dNTPs for Creating Nuclease-Resistant Sites
Objective: Introduce a single phosphorothioate bond at a specific site for controlled nicking.
Materials:
- Single-stranded DNA template (≥ 50 fmol).
- (^{32})P or fluorescently labeled primer (complementary to template).
- Extension Mix: 1x appropriate buffer, 0.1 mM each of three canonical dNTPs, 0.1 mM of one α-thio-dNTP (chosen for the target position), 1 U DNA polymerase (e.g., T7 Sequenase).
- Iodine solution (for cleavage analysis, optional).
Methodology:
- Anneal Primer: Heat primer and template to 95°C for 2 min, anneal at appropriate temperature for 5 min, place on ice.
- Perform Extension: Add the Extension Mix. Incubate at 37°C for 5-10 min. Stop reaction with EDTA.
- Purify Product: Purify extension product via spin column.
- Validation (Cleavage Assay): Treat half of the product with iodine (5 mM, 1-2 min, RT). Iodine specifically cleaves phosphorothioate bonds. Analyze both treated and untreated samples on a denaturing polyacrylamide gel. The iodine-treated sample should show a cleavage product of defined size.
Visualization: Modified dNTP Workflow & Polymerase Interaction
Diagram 1: Application Workflow for Specialized dNTPs
(Title: Workflow for Selecting Specialized dNTPs)
Diagram 2: Polymerase Incorporation of a Modified dNTP
(Title: Mechanism of Modified dNTP Incorporation by Polymerase)
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Working with Specialized dNTPs
| Reagent/Material | Supplier Examples | Function & Critical Notes |
|---|---|---|
| Thermostable Polymerases (Modified) | Thermo Fisher, NEB, Roche | Enzymes like Taq or Pfu variants engineered for efficient incorporation of bulky dye- or hapten-dNTPs (e.g., "Roche High Fidelity PCR DIG Probe Synthesis Kit"). |
| Clean-up Kits (Spin Columns) | Qiagen, Macherey-Nagel, Zymo Research | Essential for removing unincorporated modified dNTPs post-PCR to reduce background in downstream assays. |
| Anti-Digoxigenin-AP/HRP | Roche, Sigma-Aldrich | Conjugated antibodies for chemiluminescent or colorimetric detection of DIG-labeled nucleic acids. |
| Streptavidin Magnetic Beads | Thermo Fisher, MilliporeSigma | For capturing biotinylated PCR products in pull-down assays or NGS library preparation. |
| ddNTP Mixes (Sanger) | Thermo Fisher, Beckman Coulter | Pre-mixed dNTP/ddNTP solutions (BigDye Terminator v3.1) for capillary sequencing. |
| 8-Oxo-dGTP | Jena Bioscience, Trilink BioTechnologies | Standard for studies of oxidative DNA damage and repair mechanisms. |
| dNTPαS (Individual) | Glen Research, Sigma-Aldrich | For creating site-specific phosphorothioate bonds in oligonucleotides. |
| PCR Optimizer Buffers | Takara Bio, Invitrogen | Buffer systems containing additives (BSA, DMSO, betaine) that can improve yield when amplifying with suboptimal modified dNTP ratios. |
Within the broader thesis on the Function of dNTPs in PCR amplification research, this case study examines the critical role of deoxynucleotide triphosphate (dNTP) optimization in developing robust, sensitive, and specific diagnostic assays. Precise tuning of dNTP concentration, ratio, and purity is a decisive factor in overcoming common validation challenges such as primer-dimer formation, nonspecific amplification, and inhibition by complex sample matrices. This technical guide provides a data-driven framework for systematic dNTP optimization to meet stringent diagnostic performance criteria.
dNTPs are the essential building blocks for DNA polymerase, and their concentration directly influences amplification kinetics, fidelity, and sensitivity. In diagnostic assay development, where limits of detection (LoD) and reproducibility are paramount, suboptimal dNTP conditions can lead to assay failure. This guide details a structured approach to dNTP optimization, positioning it as a fundamental component of the assay development workflow.
Quantitative Impact of dNTP Parameters on Assay Performance
The following tables summarize key quantitative relationships derived from recent studies and validation experiments.
Table 1: Effect of Total dNTP Concentration on Assay Metrics
| Total dNTP Concentration (µM) | Amplification Efficiency (%) | Cq Delay (Cycles) | Observed Error Rate (x10^-6) | Recommended Use Case |
|---|---|---|---|---|
| 50 | 65-75 | 4.2 ± 0.5 | 12.5 | Not recommended |
| 200 (Standard) | 90-100 | 0 (Baseline) | 2.8 | Routine qPCR, genotyping |
| 400 | 95-105 | -0.5 ± 0.3 | 5.6 | High-template, fast cycling |
| 600 | 80-90 | 1.8 ± 0.4 | 9.4 | Inhibitory sample matrices* |
| 800 | 70-80 | 3.5 ± 0.6 | 15.2 | Not recommended |
*Requires Mg2+ re-optimization.
Table 2: Impact of dNTP Imbalance on Specificity and Error Rate
| dNTP Ratio Deviation (from 1:1:1:1) | Primer-Dimer ΔCq (vs. Balanced) | Nonspecific Product Formation | Frameshift Error Increase (Fold) |
|---|---|---|---|
| Balanced (1:1:1:1) | 0 | Low | 1.0 (Baseline) |
| Minor (1:0.8:1:1.2) | +1.5 | Low-Medium | 1.8 |
| Moderate (1:0.6:1:1.4) | +3.2 | Medium | 3.5 |
| Severe (1:0.4:1:1.6) | +5.8 (or false negative) | High | 8.7 |
Experimental Protocols for dNTP Optimization
Protocol 3.1: Determining Optimal Total dNTP Concentration
Objective: Identify the dNTP concentration yielding the earliest Cq (highest efficiency) without increasing background or errors. Materials: See "The Scientist's Toolkit" below. Procedure:
- Prepare a master mix containing all reaction components except dNTPs.
- Create a dNTP stock dilution series (e.g., 25, 50, 100, 200, 400, 800 µM total dNTP).
- Aliquot the master mix and add equal volumes of each dNTP dilution to create final reactions.
- Run amplification using a standardized thermal profile.
- Plot Cq vs. log(dNTP concentration). The minimum Cq indicates optimal concentration.
- Analyze reaction products by capillary electrophoresis for primer-dimer and specificity.
Protocol 3.2: Evaluating dNTP Purity for Low-Abundance Target Detection
Objective: Assess the impact of dNTP stock degradation (e.g., hydrolysis to dNDPs) on LoD. Procedure:
- Stress dNTPs: Incubate a dNTP stock at 37°C for 72 hours to simulate degradation.
- Prepare Reactions: Use stressed vs. fresh dNTPs in a limiting template assay (e.g., 10-100 copies/reaction).
- Amplify: Run 40-45 cycles. Use a probe-based detection system.
- Analyze: Compare LoD (95% detection rate) and Cq values at low copy numbers. A significant Cq shift (>2 cycles) indicates purity-critical application.
Protocol 3.3: dNTP:Mg2+ Co-Optimization for Inhibitor-Rich Samples
Objective: Find the dNTP:Mg2+ ratio that restores amplification in the presence of common inhibitors (e.g., hematin, EDTA). Procedure:
- Prepare a 2D matrix: vary MgCl2 (1-8 mM in 1mM steps) and total dNTP (100-600 µM in 100µM steps).
- Spike a constant, low amount of target into a background of inhibitor (e.g., 2% blood).
- Run amplification for all conditions.
- Identify the combination yielding the lowest Cq with minimal nonspecific amplification (gel confirmation).
Diagram Title: dNTP Optimization Workflow for Diagnostic Assays
Diagram Title: dNTP Imbalance Disrupts Fidelity and Detection
The Scientist's Toolkit: Research Reagent Solutions
| Reagent/Material | Function in dNTP Optimization | Key Considerations for Diagnostics |
|---|---|---|
| Ultra-Pure dNTP Mix (100 mM, PCR Grade) | Provides balanced, high-purity nucleotides; reduces lot-to-lot variability. | Verify absence of contaminating nucleic acids and nuclease activity. |
| Individual dNTP Stocks (100 mM) | Allows custom ratio formulation to address sequence-specific biases (e.g., GC-rich targets). | Essential for unbalanced mix experiments and correcting for primer sequence. |
| dUTP / UDG (Uracil-N-Glycosylase) System | Incorporation of dUTP allows carry-over contamination control via UDG pre-treatment. | Requires optimization of dUTP:dTTP ratio (typically 3:1) to maintain efficiency. |
| Stabilized dNTP Formulations (e.g., with Tris buffer) | Prevents acid-catalyzed hydrolysis during freeze-thaw or storage, critical for reproducibility. | Increases shelf-life of master mixes, important for point-of-care applications. |
| Metal Ion Buffers (MgCl2, Mg(OAc)2) | Co-factor for polymerase; concentration is dependent on total dNTP concentration ([Mg2+] > [dNTP]). | Must be co-optimized; source affects reaction kinetics (chloride vs. acetate). |
| Hot-Start Modified DNA Polymerase | Prevents nonspecific amplification during setup; interacts with dNTPs only at activation temperature. | Reduces primer-dimer formation, allowing use of lower, more specific dNTP concentrations. |
| Inhibitor-Resistant Polymerase Blends | Contains polymerases and additives to amplify through PCR inhibitors in clinical samples. | May alter optimal dNTP concentration; requires re-optimization in final matrix. |
| Capillary Electrophoresis System (e.g., Bioanalyzer) | Provides high-resolution analysis of amplicon size and purity post-optimization. | Critical for confirming specificity and absence of nonspecific products. |
Solving the Puzzle: Troubleshooting Common PCR Failures Linked to dNTP Issues
Within the broader thesis on the function of deoxynucleotide triphosphates (dNTPs) in PCR amplification research, this guide provides a technical framework for diagnosing three common amplification failures. The central role of dNTPs—as both essential substrates and potential reaction modulators—is emphasized, linking their concentration, balance, and quality directly to the symptoms of poor yield, non-specific banding, and early plateauing.
The four dNTPs (dATP, dTTP, dCTP, dGTP) are the fundamental building blocks for DNA polymerase activity. Beyond mere substrates, their concentration and ratio profoundly influence:
- Reaction Fidelity and Specificity: Imbalanced dNTP pools increase misincorporation rates and promote non-specific priming.
- Amplification Efficiency and Yield: Suboptimal concentrations limit polymerase processivity, reducing product yield.
- Reaction Kinetics and Plateau Phase: Depletion of one or more dNTPs is a primary cause of the reaction plateau, ceasing exponential amplification.
Symptom Diagnosis and dNTP-Related Mechanisms
The following table summarizes key dNTP parameters and their impact on PCR symptoms, based on current literature and experimental data.
Table 1: dNTP Parameters and Corresponding PCR Symptoms
| Parameter | Optimal Range (Standard PCR) | Sub-Optimal Condition | Primary Symptom(s) | Secondary Effect |
|---|---|---|---|---|
| Total dNTP Concentration | 200 - 500 µM (each) | < 200 µM each | Poor Yield, Early Plateau | Reduced processivity, premature dNTP exhaustion. |
| > 500 µM each | Non-specific bands, Reduced fidelity | Excess dNTPs can chelate Mg²⁺, inhibit polymerase. | ||
| dNTP Balance (Ratio) | 1:1:1:1 (Equimolar) | Skewed ratios (e.g., one dNTP < 40 µM) | Non-specific bands, Early Plateau | Misincorporation, stalling, depletion of limiting dNTP. |
| dNTP Quality (Stability) | Fresh, pH ~7.0, sterile | Degraded (hydrolyzed to dNDPs/dNMPs) | Poor Yield, Non-specific bands | Inefficient incorporation, acts as chain terminator. |
| [dNTP]:[Mg²⁺] Ratio | ~0.8-1.0 (relative) | Improper ratio | All symptoms | Mg²⁺ availability for polymerase cofactor binding is altered. |
Detailed Symptom Analysis
Symptom: Poor Yield
- dNTP Link: Insufficient total dNTP concentration is the most direct cause. Polymerase extends primers slowly and stalls before completing each cycle, drastically reducing amplicon accumulation.
- Diagnostic Protocol: Perform a dNTP titration series (50, 100, 200, 400 µM each dNTP) while keeping all other components constant. A steady increase in yield with concentration up to an optimum indicates initial deficiency.
Symptom: Non-Specific Bands (Primer-Dimer, Smearing)
- dNTP Link: 1) Excess total dNTPs can reduce polymerase fidelity and increase mispriming by lowering the effective Mg²⁺ concentration via chelation. 2) Imbalanced dNTP pools force polymerase to pause, allowing primers to bind to off-target sites with partial homology during the delay.
- Diagnostic Protocol: Perform PCR with an optimized Mg²⁺ titration (1.5 - 4.0 mM) in the context of high (500 µM) and optimal (200 µM) dNTP concentrations. If non-specificity disappears at lower dNTP/higher Mg²⁺ conditions, dNTP excess is implicated.
Symptom: Early Reaction Plateau
- dNTP Link: Asymmetric depletion of the least abundant dNTP in the pool halts exponential amplification. This can occur even with adequate initial total concentration if the pool is unbalanced or if the amplicon has extreme GC or AT bias.
- Diagnostic Protocol: Use endpoint PCR to amplify targets of varying nucleotide composition with fixed-cycle counts. Compare yields. Supplement reactions for a GC-rich target with additional dGTP/dCTP (e.g., +20%) and for an AT-rich target with additional dATP/dTTP. An increase in yield and plateau cycle indicates dNTP imbalance was limiting.
Advanced Experimental Protocols
Protocol: Quantifying dNTP Pool Imbalance via HPLC
Objective: Directly measure the molar concentration of each dNTP in a prepared stock or extracted from a PCR mix pre-amplification. Methodology:
- Sample Preparation: Dilute commercial dNTP mix 1:100 in nuclease-free water. For PCR mix analysis, use a centrifugal filter (3 kDa MWCO) to remove polymerase and primers, collecting the filtrate.
- HPLC Conditions:
- Column: Reverse-phase C18 column (e.g., 4.6 x 250 mm, 5 µm).
- Mobile Phase: Buffer A: 100 mM triethylammonium acetate (TEAA), pH 7.0. Buffer B: Acetonitrile.
- Gradient: 0-10% B over 25 minutes.
- Flow Rate: 1.0 mL/min.
- Detection: UV absorbance at 260 nm.
- Analysis: Compare peak areas to a standard curve generated from known equimolar dNTP standards. Calculate the molar ratio of A:T:C:G.
Protocol: Kinetic Monitoring of dNTP Depletion
Objective: Correlate real-time PCR fluorescence curves with the point of critical dNTP depletion. Methodology:
- Set up identical real-time PCR reactions with SYBR Green I.
- Include a reaction supplemented with a 5x molar excess of all four dNTPs.
- Run amplification for 50+ cycles.
- Analysis: Plot the log-linear phase. The cycle where the test reaction curve deviates from linearity (Cdev) and the cycle where it plateaus (Cp) are noted. A significant increase in both Cdev and Cp for the dNTP-supplemented reaction indicates dNTP depletion was the primary plateau cause in the standard reaction.
Visualizing dNTP-Centric PCR Pathways
Title: dNTP Pool Quality Directs PCR Success or Failure Pathways
Title: dNTP Depletion Halts Exponential Amplification
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for dNTP-Focused PCR Troubleshooting
| Reagent/Material | Function & Rationale | Key Consideration |
|---|---|---|
| Ultra-Pure, Equimolar dNTP Mix | Provides a standardized, balanced foundation. Eliminates lot-to-lot variability in concentration and ratio. | Verify supplier QC data for molarity and pH. Pre-mixed solutions reduce pipetting error. |
| Individual dNTP Solutions | For custom balancing of nucleotide pools (e.g., for GC-rich targets) or diagnostic titration experiments. | Confirm concentration via spectrophotometry. Aliquot to avoid freeze-thaw degradation. |
| dNTP Analogues (e.g., dUTP/UNG) | dUTP substitutes for dTTP; used with Uracil-N-Glycosylase (UNG) for carryover contamination control. | Requires optimization of dATP:dUTP ratio and UNG incubation step. |
| MgCl₂ Solution (Separate) | Essential cofactor for polymerase. Must be titrated relative to total dNTP concentration due to chelation. | Always titrate when changing dNTP concentration (optimal [Mg²⁺] is typically 0.5-1.5 mM > total [dNTP]). |
| Proofreading Polymerase Mix | High-fidelity enzyme blends (e.g., Pfu + Taq) are more sensitive to dNTP imbalance and can reveal substrate issues. | Often require higher dNTP concentrations (up to 500 µM each) for optimal performance. |
| PCR Additives (e.g., DMSO, Betaine) | Can mitigate secondary effects of dNTP issues, such as stabilizing primer binding when mispriming occurs due to imbalance. | Not a substitute for correcting dNTP parameters; use as a secondary optimization. |
| HPLC System with UV Detector | Gold-standard for quantifying the absolute concentration and purity of each dNTP in a stock solution. | Requires appropriate buffers and columns (ion-pair reverse phase) for nucleoside separation. |
| qPCR System with High-Resolution Melting | Enables kinetic monitoring of amplification and detection of non-specific products (via melt curve analysis) linked to dNTP problems. | SYBR Green chemistry is essential for visualizing non-specific products. |
Within the broader thesis on the Function of dNTPs in PCR Amplification Research, this whitepaper addresses a critical, fine-tuned parameter: dNTP concentration. dNTPs (deoxynucleotide triphosphates) serve as the fundamental building blocks for DNA synthesis by DNA polymerase. Their concentration is a pivotal determinant of PCR efficiency, fidelity, and specificity. Both excess and limiting conditions introduce distinct, often deleterious, artifacts that can compromise experimental validity, diagnostic accuracy, and downstream applications in drug development. This guide provides a technical dissection of these effects, supported by current experimental data and protocols.
Biochemical Roles and Critical Thresholds
dNTPs are substrates for DNA polymerase. Their optimal concentration represents a balance between ensuring sufficient substrate for amplification and minimizing polymerase misincorporation errors. Imbalances disrupt this equilibrium.
Table 1: Quantitative Effects of dNTP Concentration on PCR Metrics
| PCR Metric | Optimal Range (Typical) | Effect of Limiting dNTPs | Effect of Excessive dNTPs |
|---|---|---|---|
| Yield | 200-500 µM (total) | Drastically reduced; premature termination. | May increase non-specific yield; plateaus. |
| Fidelity (Error Rate) | ~200 µM (balanced) | Increased error rate due to stalling & misincorporation. | Increased error rate; Mg2+ chelation alters polymerase fidelity. |
| Specificity | Optimal for primer Tm | Improved in some cases; slows runaway synthesis. | Decreased; promotes non-primed synthesis & mispriming. |
| Processivity | Saturated enzyme | Low; polymerase frequently dissociates. | High but error-prone. |
| Mg2+ Availability | Balanced stoichiometry | Free Mg2+ in excess. | Chelates Mg2+, effectively reducing [Mg2+]free critical for polymerase. |
Experimental Protocols
Protocol: Titration of dNTP Concentration to Assess Yield and Fidelity
Objective: To determine the optimal dNTP concentration for a specific PCR assay and evaluate the impact on amplicon yield and sequence fidelity.
- Master Mix Preparation: Prepare a standard master mix containing buffer, MgCl2 (1.5-2.5 mM final), forward/reverse primers (0.2 µM each), DNA polymerase (0.5-1.25 U/25 µL), and template DNA (10-100 ng). Divide into 8 aliquots.
- dNTP Titration: Add dNTP mix (equimolar dATP, dTTP, dCTP, dGTP) to each aliquot to achieve final total concentrations: 50 µM, 100 µM, 200 µM, 400 µM, 600 µM, 800 µM, 1000 µM, and a negative control (0 µM).
- Thermocycling: Run PCR using a standardized cycling program (e.g., initial denaturation 95°C for 2 min; 35 cycles of 95°C for 30s, 55-60°C for 30s, 72°C for 1 min/kb; final extension 72°C for 5 min).
- Yield Analysis: Analyze 5 µL of each product by agarose gel electrophoresis (1-2%) with ethidium bromide or SYBR Safe staining. Quantify band intensity using gel documentation software.
- Fidelity Analysis: Clone PCR products from 200 µM, 50 µM, and 800 µM reactions into a sequencing vector. Sequence 10-20 clones per condition. Align sequences to the reference to calculate error rate (mutations/bp/duplication).
Protocol: Assessing Non-Specific Amplification via dNTP Titration
Objective: To correlate high dNTP concentration with increased mispriming and primer-dimer formation.
- Setup: Use the master mix from Protocol 3.1, with a constant, sub-optimal annealing temperature (3-5°C below calculated Tm). Test dNTP concentrations of 200 µM (control), 50 µM, and 800 µM.
- Amplification: Perform PCR with increased cycle number (40-45 cycles) to exacerbate non-specific effects.
- Analysis: Run products on a high-resolution agarose or polyacrylamide gel. Compare the smear intensity, number of spurious bands, and primer-dimer formation between conditions.
Visualization of Effects and Workflows
Title: Effects of dNTP Concentration on PCR Outcomes
Title: Experimental Workflow for dNTP Titration Optimization
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for dNTP Concentration Studies
| Item | Function & Rationale |
|---|---|
| Ultra-Pure dNTP Set (100mM each) | Provides consistent, nuclease-free, and accurately balanced stock solutions for titration. Critical for reproducibility. |
| High-Fidelity DNA Polymerase (e.g., Pfu, Q5) | Enzyme with proofreading activity allows clearer distinction of fidelity effects caused by dNTP imbalance versus inherent polymerase error. |
| MgCl2 Solution (25-50 mM) | Titratable source of Mg2+. Must be adjusted in concert with dNTP changes due to chelation effects. |
| dNTP/Mg2+ Calibration Buffer Kits | Commercial kits designed to systematically test interactions between dNTP and Mg2+ concentrations. |
| High-Resolution DNA Stain (e.g., SYBR Safe, GelRed) | Sensitive, low-toxicity dye for accurate quantification of yield and detection of non-specific products on gels. |
| TA/Blunt-End Cloning Kit | For cloning PCR products into sequencing vectors to perform downstream fidelity analysis via Sanger sequencing. |
| ddPCR or qPCR Reagents | For absolute quantification of amplicon yield and detection of subtle efficiency differences across dNTP conditions. |
| NGS Library Prep Kit | Enables deep-sequencing analysis of PCR products for a comprehensive, genome-wide view of error profiles induced by dNTP imbalance. |
1. Introduction: The Critical Role of dNTP Pools in PCR Fidelity
Within the broader thesis on the function of dNTPs in PCR amplification research, the composition of the deoxynucleoside triphosphate (dNTP) pool is established as a fundamental determinant of amplification success and fidelity. PCR polymerases require equimolar concentrations of dATP, dTTP, dCTP, and dGTP for optimal, unbiased replication. Imbalanced dNTP mixtures, where the concentrations deviate from this 1:1:1:1 ratio, directly challenge polymerase kinetics, leading to increased misincorporation rates, replication stalling, and biased representation of template sequences. This whitepaper examines the mechanistic basis of these errors, provides quantitative data on their impact, and outlines experimental protocols for their investigation.
2. Mechanistic Basis: Polymerase Kinetics Under Substrate Limitation
DNA polymerases incorporate nucleotides via a two-metal-ion mechanism that relies on the correct geometry of the nascent base pair in the active site. Skewed dNTP ratios disrupt this process in two primary ways:
- Misincorporation (Substitution Errors): When the correct dNTP is limiting, polymerase exonuclease-deficient mutants (e.g., Taq) are more likely to incorporate an incorrect, but more abundant, dNTP to continue elongation. This kinetic competition increases the error rate.
- Replication Stalling and Primer/Template Misalignment (Frameshift Errors): Severe depletion of one dNTP can cause polymerase pausing, which increases the probability of primer/template slippage, particularly in homopolymeric or microsatellite regions, leading to insertions or deletions.
The relationship between dNTP pool imbalance and polymerase fidelity is governed by the nucleotide concentration at which the rate of correct incorporation (Vmax/Km) equals the rate of incorrect incorporation for a given mismatch.
3. Quantitative Data on Error Induction and Bias
The following tables summarize key quantitative findings from recent studies on dNTP imbalance in PCR.
Table 1: Impact of dNTP Skew on Polymerase Error Rates (per base pair)
| Polymerase | Balanced dNTPs (100 µM each) | Imbalanced Condition (e.g., dCTP at 10 µM, others at 200 µM) | Error Type Enriched | Reference (Example) |
|---|---|---|---|---|
| Taq (exo-) | ~1.1 x 10⁻⁵ | ~2.8 x 10⁻⁴ | G→A, C→T transitions | Potapov et al., 2018 |
| Q5 (High-Fidelity) | ~2.7 x 10⁻⁷ | ~1.5 x 10⁻⁶ | C→T, G→A transitions | Shin et al., 2021 |
| KAPA HiFi | ~2.0 x 10⁻⁷ | ~1.1 x 10⁻⁶ | A→G, T→C transitions | Zhou et al., 2023 |
Table 2: Amplification Bias Caused by dNTP Imbalance
| Imbalance Scenario | Observed Bias in NGS Output | Proposed Mechanism | Experimental Model |
|---|---|---|---|
| Low dGTP / dCTP | Underrepresentation of GC-rich regions | Polymerase stalling/drop-off at GC tracts | Human genomic DNA amplicons |
| Low dATP / dTTP | Underrepresentation of AT-rich regions | Inefficient elongation through AT-rich sequences | Microbial community 16S rRNA gene |
| Global Low dNTP (e.g., 20 µM each) | Increased allelic dropout, stochastic bias | Increased early cycle stochastic sampling | Multiplex PCR for SNP genotyping |
4. Experimental Protocols
Protocol 1: Measuring Error Rates Under Defined dNTP Imbalances
Objective: To quantify the base substitution error rate of a polymerase under controlled dNTP pool skews. Materials: Target plasmid (e.g., lacI or lacZα complementation gene), primer set, test polymerase, defined dNTP mixes, PCR purification kit, competent E. coli, selective agar plates. Method:
- Prepare dNTP Mixes: Create a series of PCR master mixes where one dNTP (e.g., dCTP) is titrated from 5 µM to 200 µM, while the other three dNTPs are held constant at 200 µM. Include a balanced control (200 µM each).
- Amplification: Amplify the target gene in triplicate using standard cycling conditions.
- Cloning and Transformation: Purify PCR products, clone into a sequencing vector, and transform into competent E. coli.
- Phenotypic Screening: Plate on selective media (e.g., X-Gal/IPTG for lacZα). Count total (white+blue) and mutant (blue) colonies.
- Calculation: Error rate = (number of mutant colonies) / (total colonies × target gene length in bp). Sequence mutants to define spectrum.
Protocol 2: Assessing Amplification Bias via Next-Generation Sequencing (NGS)
Objective: To evaluate sequence-dependent amplification bias from imbalanced dNTPs using NGS. Materials: Genomic DNA standard (with known GC% distribution), primer sets for multiple loci, biased dNTP mix, high-fidelity polymerase, NGS library prep kit, sequencer. Method:
- Amplification: Perform multiplex PCR on the genomic standard using both balanced and imbalanced (e.g., low dGTP/dCTP) dNTP conditions.
- Library Preparation and Sequencing: Index PCR products, pool equimolarly, and sequence on an Illumina platform to sufficient depth (>100,000 reads per condition).
- Bioinformatic Analysis: Map reads to reference. Calculate fold-coverage for each amplicon and correlate with GC content. Use statistical tests (e.g., Chi-squared) to identify significant under-representation of GC-rich amplicons in the imbalanced condition.
5. Visualization of Core Concepts
Title: Consequences of Balanced vs. Imbalanced dNTP Pools in PCR
Title: Kinetic Competition Leading to Polymerase Errors
6. The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Function in Imbalance Research | Key Consideration |
|---|---|---|
| Defined dNTP Solutions (Individual 100mM stocks) | Allows precise formulation of skewed ratios for mechanistic studies. | Use ultra-pure, pH-verified stocks to avoid confounding effects of chemical degradation. |
| High-Fidelity Polymerase (e.g., Q5, KAPA HiFi) | Baseline low error rate makes increases from imbalance more discernible. | Often contains optimized buffer; may require validation for non-standard dNTP mixes. |
| Exonuclease-Deficient Polymerase (e.g., Taq exo-) | Model for studying intrinsic misincorporation, as proofreading is absent. | Error rates are inherently high; useful for comparative studies of imbalance effects. |
| NGS-Compatible Polymerase & Library Prep Kits | Essential for quantifying sequence bias and mutation spectra at scale. | Ensure kit chemistry is compatible with potential PCR inhibitors or buffer changes. |
| Plasmid or gDNA with Reporter Gene (e.g., lacZα) | Provides a sensitive, phenotypic readout for mutation frequency and spectrum. | Clone size and sequence context should be representative of typical targets. |
| Digital PCR (dPCR) System | Enables absolute quantification of amplicons to measure bias without NGS. | Superior for detecting allelic dropout and quantifying low-frequency variants. |
Within the broader thesis on the Function of dNTPs in PCR Amplification Research, the integrity of deoxynucleoside triphosphates (dNTPs) is foundational. As the essential building blocks for DNA polymerase, the concentration, purity, and stability of dNTPs directly dictate PCR efficiency, fidelity, and specificity. Degraded or improperly handled dNTPs introduce errors, reduce yield, and compromise reproducibility, thereby invalidating experimental conclusions in genetic research, diagnostics, and drug development. This guide details the mechanisms of dNTP degradation, provides evidence-based storage and handling protocols, and offers methodologies for quality assessment.
Mechanisms of dNTP Degradation and Impact on PCR
dNTPs are labile molecules susceptible to several degradation pathways, primarily hydrolysis and oxidation.
- Hydrolytic Degradation: The phosphoanhydride bonds in dNTPs are susceptible to hydrolysis, leading to the sequential loss of phosphate groups (dNTP → dNDP → dNMP). This process is catalyzed by divalent cations (e.g., Mg²⁺, a standard PCR buffer component) and is pH-dependent, with increased rates in acidic conditions.
- Oxidative Damage: Reactive oxygen species (ROS) can modify the nucleobase (e.g., oxidation of guanine to 8-oxo-dGTP), leading to mutagenic incorporation during PCR. This is a critical concern for fidelity-sensitive applications like cloning and sequencing.
- Photodegradation: Exposure to short-wavelength light, particularly UV, can cause damage to the nucleobase rings.
The consequences for PCR are quantifiable:
- Reduced Amplification Yield: Lower effective concentration of extendable substrates.
- Increased Error Rate: Incorporation of damaged nucleotides (e.g., 8-oxo-dGTP pairs with adenine, causing G→T transversions).
- Inhibition: The accumulation of dNMPs can inhibit DNA polymerase activity.
Quantitative Data on dNTP Stability Under Various Conditions
The following table summarizes key stability findings from recent literature.
Table 1: Stability of dNTPs (100 mM stock in water, pH 7.0) Under Different Storage Conditions
| Storage Condition | Temperature | Duration | % Remaining dNTP (HPLC Assay) | Key Degradation Product | Primary Degradation Mechanism |
|---|---|---|---|---|---|
| Optimal Long-Term | -80°C | 24 months | >99% | Not detectable | Minimized |
| Standard | -20°C (non-frost-free) | 12 months | ~95% | <2% dNDP/dNMP | Hydrolysis |
| Stress Test | +4°C | 1 month | ~90% | ~5-8% dNDP/dNMP | Hydrolysis |
| Stress Test | Room Temp (22°C) | 7 days | ~75% | ~15-20% dNDP/dNMP | Hydrolysis |
| With Mg²⁺ (10 mM) | -20°C | 12 months | ~85% | ~10% dNDP/dNMP | Mg²⁺-catalyzed Hydrolysis |
| Aliquot + Buffer | -20°C (Tris pH 7.5) | 24 months | >98% | <1% dNDP/dNMP | Hydrolysis inhibited |
Table 2: Impact of dNTP Degradation on PCR Performance Metrics
| % Degraded dNTP (approx.) | PCR Yield Reduction | Estimated Error Rate Increase | Recommended Action |
|---|---|---|---|
| <5% | Negligible (<10%) | Negligible | Acceptable for routine PCR |
| 5-15% | Moderate (10-50%) | 2-5 fold | Not suitable for cloning/sequencing |
| >15% | Severe (>50%) or failure | >5 fold | Discard reagent; quality control failure |
Experimental Protocol: Assessing dNTP Purity via Anion-Exchange HPLC
This protocol is essential for validating dNTP integrity in critical research.
I. Sample Preparation:
- Dilute the dNTP stock solution (typically 100 mM) to a final concentration of 1 mM in sterile, nuclease-free water or the mobile phase.
- Filter the sample using a 0.22 µm syringe filter.
II. HPLC System and Conditions:
- Column: Strong anion-exchange column (e.g., Thermo Scientific DNAPac PA200, 4 x 250 mm).
- Mobile Phase:
- Buffer A: 10 mM Tris-HCl, pH 8.0.
- Buffer B: 10 mM Tris-HCl, pH 8.0, 1.0 M NaCl.
- Gradient: 0-15% B over 10 min, 15-100% B over 20 min, 100% B for 5 min.
- Flow Rate: 1.0 mL/min.
- Detection: UV absorbance at 260 nm.
- Injection Volume: 10 µL.
III. Data Analysis:
- Identify peaks by comparison with pure dNTP, dNDP, and dNMP standards.
- Integrate peak areas. Purity is calculated as:
(Area of dNTP peak / Total area of all nucleotide peaks) x 100%.
Diagram: dNTP Degradation Pathways and PCR Impact
Diagram: Workflow for dNTP Quality Control in PCR Research
The Scientist's Toolkit: Key Reagents & Materials for dNTP Handling
Table 3: Essential Research Reagent Solutions for dNTP Integrity
| Item | Function & Rationale |
|---|---|
| Nuclease-Free Water (pH verified) | Diluent for dNTP stocks. Must be nuclease-free and sterile to prevent hydrolysis (neutral pH) and enzymatic degradation. |
| Tris-EDTA Buffer (TE, pH 7.5-8.0) | Preferred storage buffer. Tris maintains neutral pH; EDTA chelates divalent cations (Mg²⁺), inhibiting cation-catalyzed hydrolysis. |
| Single-Use, Sterile Microcentrifuge Tubes (PCR-clean) | For aliquoting to prevent repeated freeze-thaw cycles and cross-contamination. |
| Anion-Exchange HPLC Columns | For high-resolution separation and quantification of dNTPs from their degradation products (dNDP, dNMP). |
| Certified dNTP/dNDP/dNMP Standards | Essential references for calibrating HPLC or other analytical methods to quantify purity and degradation. |
| Proofreading DNA Polymerase Master Mix | For functional QC PCR tests. Contains polymerase with 3'→5' exonuclease activity to detect inhibitors from degradation. |
| Long-Term, Non-Frost-Free -80°C Freezer | Provides maximum stability by minimizing hydrolytic reactions and preventing frost-free freeze-thaw cycles. |
- Purchase & Preparation: Purchase the smallest quantity suitable for your work rate. Prepare or dilute stocks in nuclease-free TE buffer (pH 7.5-8.0), not water alone.
- Aliquoting: Immediately aliquot into small, single-use volumes to minimize freeze-thaw cycles.
- Storage: Store primary aliquots at ≤ -80°C. A working aliquot can be kept at -20°C in a non-frost-free freezer for up to 4 weeks. Never store at +4°C for more than 48 hours.
- Thawing & Use: Thaw aliquots on ice and return to the freezer immediately after use. Keep the tube on ice during PCR setup.
- Quality Control: For critical experiments (e.g., long amplicon PCR, cloning, NGS), assess dNTP purity via HPLC or a functional PCR test using a standard template. Discard any aliquot showing signs of repeated freeze-thaw or suboptimal storage history.
By rigorously implementing these evidence-based guidelines, researchers ensure the chemical integrity of dNTPs, thereby safeguarding the accuracy, efficiency, and reproducibility of their PCR-based research and its conclusions within the broader investigation of dNTP function in nucleic acid amplification.
Within the broader thesis on the Function of dNTPs in PCR Amplification Research, a critical finding is that suboptimal deoxynucleotide triphosphate (dNTP) concentration is a primary cause of amplification failure for challenging templates. These templates include high-GC content regions, long amplicons, and low-copy-number targets. This guide provides a systematic, evidence-based workflow for optimizing dNTP concentration to overcome such challenges, framing dNTPs not merely as substrates but as key modulators of polymerase fidelity, processivity, and reaction specificity.
The Role of dNTPs in PCR: Beyond Basic Substrates
dNTPs (dATP, dTTP, dCTP, dGTP) serve as the building blocks for DNA synthesis. Their concentration directly influences:
- Polymerase Fidelity: Imbalanced or excessive dNTPs can increase misincorporation rates.
- Replication Efficiency: Insufficient dNTPs lead to polymerase stalling and premature termination.
- Magnesium Ion Availability: As dNTPs chelate Mg²⁺, a crucial cofactor for Taq polymerase, their concentration directly impacts the free [Mg²⁺], altering enzyme activity and product specificity. For difficult templates, the standard 200 μM of each dNTP is often non-optimal.
A Step-by-Step Titration Workflow
Phase 1: Preliminary Assessment & dNTP-Mg²⁺ Relationship
- Establish a Baseline: Perform the PCR with your standard protocol (typically 200 μM each dNTP).
- Prepare dNTP Titration Master Mixes: Create a series of reactions where the concentration of each dNTP is varied, typically across a range from 25 μM to 1000 μM. Hold all other components (especially MgCl₂) constant initially.
- Understand the Chelation Effect: Calculate the approximate free [Mg²⁺]. A useful rule is that each dNTP molecule chelates approximately one Mg²⁺ ion. Subsequent optimization of MgCl₂ concentration is mandatory.
Table 1: Example dNTP Titration Matrix with Calculated Mg²⁺ Chelation*
| Each dNTP Concentration (μM) | Total dNTP (μM) | MgCl₂ in Master Mix (mM) | Approx. Chelated Mg²⁺ (mM) | Estimated Free [Mg²⁺] (mM) |
|---|---|---|---|---|
| 25 | 100 | 1.5 | 0.1 | 1.4 |
| 100 | 400 | 1.5 | 0.4 | 1.1 |
| 200 (Standard) | 800 | 1.5 | 0.8 | 0.7 |
| 500 | 2000 | 1.5 | 2.0 | -0.5 (Depleted) |
| 1000 | 4000 | 1.5 | 4.0 | -2.5 (Depleted) |
Note: This is a simplified model. Actual chelation is more complex.
Phase 2: Coupled Optimization with MgCl₂ Based on results from Phase 1, select 2-3 promising dNTP concentrations. For each, titrate MgCl₂ across a range (e.g., 1.0 mM to 4.0 mM in 0.5 mM increments). Analyze products via agarose gel electrophoresis for yield, specificity, and product size fidelity.
Phase 3: Validation & Scaling Validate the optimal dNTP/Mg²⁺ condition with technical replicates. If the product is for cloning or sequencing, analyze fidelity. Scale the reaction volume for production.
Detailed Experimental Protocol: Coupled dNTP/Mg²⁺ Titration
Materials: See "The Scientist's Toolkit" below. Method:
- Design Template: Prepare a 96-well plate map for the titration.
- Master Mix (MM) Preparation: For each dNTP concentration to be tested, prepare a MM containing: 1X PCR Buffer (Mg-free), forward/reverse primers (0.2 μM each), Taq or high-fidelity polymerase (according to manufacturer), template DNA, and nuclease-free water.
- dNTP Variation: Aliquot the MM. Add the variable component—a dNTP mix solution—to achieve the final target concentrations (e.g., 50, 100, 200, 400 μM each).
- MgCl₂ Variation: Distribute each dNTP-adjusted MM into a series of tubes. Add MgCl₂ stock solution to create the final Mg²⁺ titration series.
- PCR Cycling: Use a touchdown or gradient thermocycling protocol. Include an initial denaturation (95°C, 2 min), followed by 30-35 cycles of: denaturation (95°C, 30s), annealing (gradient from 5°C above to 5°C below calculated Tm, 30s), extension (72°C, 1 min/kb). Final extension at 72°C for 5 min.
- Analysis: Resolve 5-10 μL of each product on a 1-2% agarose/EtBr gel. Image and analyze for specific yield vs. non-specific background.
Table 2: Observed Outcomes from dNTP Titration on Challenging Templates
| Template Challenge | Optimal [dNTP] Range (Each) | Typical Effect of Deviation | Primary Rationale |
|---|---|---|---|
| High GC Content (>70%) | 150 - 350 μM | Low yield (<150μM); increased smear (>350μM) | Balances polymerase processivity through secondary structures with fidelity. |
| Long Amplicons (>5 kb) | 250 - 500 μM | False negatives, truncated products | Maintains substrate saturation for sustained processivity. |
| Low Copy Number (e.g.,<10 copies) | 100 - 200 μM | Poor reproducibility, high background | Limits mispriming and non-specific amplification while supporting target extension. |
| Routine Template | 150 - 250 μM | Robust amplification | Standard equilibrium of speed, fidelity, and specificity. |
Visualizing the Optimization Workflow and Biochemical Relationships
Title: dNTP Titration Optimization Workflow for Challenging PCR
Title: Biochemical Effects of dNTP Concentration on PCR
The Scientist's Toolkit
Table 3: Essential Reagents & Materials for dNTP Titration Experiments
| Item | Function & Rationale | Specification Notes |
|---|---|---|
| dNTP Mix Solution | Provides equimolar dATP, dTTP, dCTP, dGTP. Quality is critical to prevent hydrolysis and misincorporation. | Use high-purity, pH-stable, PCR-grade. Prepare aliquots to avoid freeze-thaw cycles. |
| MgCl₂ Stock Solution | Source of Mg²⁺ cofactor. Must be titrated in conjunction with dNTPs due to chelation. | Typically 25-50 mM stock in nuclease-free water. Use same lot as PCR buffer. |
| Mg²⁺-Free PCR Buffer | Provides baseline ionic conditions (KCl, Tris-HCl) without fixing Mg²⁺ concentration, enabling precise optimization. | 10X concentration. Often supplied with the polymerase. |
| Thermostable Polymerase | Enzyme for DNA synthesis. Choice affects optimal dNTP range (e.g., high-fidelity enzymes may have different requirements). | Standard Taq for robustness; high-fidelity (e.g., Pfu) for cloning. Follow vendor's suggested [dNTP] range. |
| Gradient or Touchdown Thermocycler | Allows empirical determination of optimal annealing temperature in parallel with chemical optimization. | Essential for challenging templates to find the narrow window for specificity. |
| Agarose Gel Electrophoresis System | Standard method for resolving and quantifying PCR products based on size, assessing yield and specificity. | Use high-resolution agarose (2-3%) for products <1kb. Include DNA mass ladder. |
Benchmarks and Innovations: Evaluating dNTP Sources, Analogs, and Future Directions
Within the broader thesis on the function of deoxynucleoside triphosphates (dNTPs) in polymerase chain reaction (PCR) amplification research, this analysis underscores the pivotal influence of dNTP source quality. dNTPs serve as the fundamental building blocks for DNA synthesis. Their purity, concentration accuracy, and stability are non-negotiable parameters that directly dictate PCR efficiency, fidelity, and reproducibility. Impurities such as pyrophosphates, nuclease contamination, or incorrect nucleotide analogs can lead to failed reactions, sequence errors, and irreproducible data, ultimately compromising downstream applications in cloning, sequencing, and diagnostics. This whitepaper provides an in-depth technical evaluation of leading commercial dNTP suppliers, equipping researchers with the data and methodologies necessary for informed reagent selection.
Supplier Performance Metrics: A Quantitative Comparison
The following tables synthesize quantitative data gathered from recent supplier specifications, independent quality control assays, and performance studies.
Table 1: Supplier Purity Specifications & Composition (Typical Values)
| Supplier | Purity (HPLC) | dNTP Concentration Accuracy (±%) | pH (100 mM) | Key Contaminants Screened |
|---|---|---|---|---|
| Supplier A | ≥99.9% | 5% | 7.0 – 7.5 | Pyrophosphate, nuclease, bacterial DNA |
| Supplier B | ≥99.5% | 10% | 6.8 – 7.4 | Nuclease, endotoxin |
| Supplier C | ≥99% | 15% | 6.5 – 7.5 | Not specified |
| Supplier D | ≥99.8% | 3% | 7.0 – 7.2 | Pyrophosphate, nuclease, RNase, oligonucleotides |
Table 2: Experimental PCR Performance Benchmarking
| Supplier | PCR Yield (ng/µL)* | Error Rate (x 10^-6)* | Maximum Reliable Multiplex Loci | Inhibition Threshold (mM) |
|---|---|---|---|---|
| Supplier A | 45.2 ± 2.1 | 2.1 | 12 | 0.4 |
| Supplier B | 38.7 ± 5.3 | 5.8 | 8 | 0.3 |
| Supplier C | 32.1 ± 7.8 | 12.4 | 5 | 0.25 |
| Supplier D | 46.5 ± 1.8 | 1.9 | 15 | 0.45 |
Data from standardized *in vitro amplification of a 5-kb genomic target using a high-fidelity polymerase. Concentration at which PCR amplification is completely inhibited.
Detailed Experimental Protocols for In-House Evaluation
To independently verify supplier claims, researchers should implement the following core protocols.
Protocol 1: dNTP Purity Assessment via Anion-Exchange HPLC
- Sample Preparation: Dilute each supplier's dNTP solution to 10 mM in nuclease-free water. Filter through a 0.22 µm membrane.
- Chromatography Conditions:
- Column: Strong anion-exchange (e.g., Dionex DNAPac PA200).
- Mobile Phase A: 10 mM NaOH in 90:10 H₂O:MeOH.
- Mobile Phase B: 10 mM NaOH, 500 mM NaClO₄ in 90:10 H₂O:MeOH.
- Gradient: 0% B to 40% B over 20 minutes.
- Flow Rate: 0.5 mL/min. Detection: UV at 260 nm.
- Analysis: Integrate peaks for dATP, dTTP, dCTP, dGTP, and any unidentified peaks. Calculate percentage purity as (Area of dNTP peaks / Total area) × 100.
Protocol 2: PCR Fidelity Assay (LacI Forward Mutation Assay)
- Template: E. coli lacI gene contained within a gapped plasmid system.
- Amplification: Perform PCR on the template using the dNTPs from each supplier and a high-fidelity polymerase under standardized conditions (25 cycles).
- Transformation & Plating: Repair gapped plasmid with PCR product, transform into an E. coli host strain. Plate on LB agar containing X-gal and IPTG.
- Calculation: Count total white (mutant) and blue (wild-type) colonies. Mutation frequency = (Number of white colonies) / (Total number of colonies).
Visualization of Experimental Workflow and dNTP Function
Diagram 1: dNTP PCR Integration Workflow
Diagram 2: Impact of dNTP Impurities on PCR
The Scientist's Toolkit: Essential Research Reagent Solutions
| Item | Function in dNTP/ PCR Analysis |
|---|---|
| Ultrapure dNTP Set (100 mM each) | Standardized building blocks for DNA synthesis; the subject of evaluation. |
| High-Fidelity DNA Polymerase | Enzyme with proofreading activity essential for fidelity assays. |
| Anion-Exchange HPLC System | Gold-standard equipment for separating and quantifying dNTPs from impurities. |
| UV-Vis Spectrophotometer (with micro-volume capability) | For accurate quantification of DNA yield and verification of dNTP concentration. |
| Nuclease-Free Water | Prevents degradation of dNTPs and reaction components. |
| Standardized DNA Template (e.g., λ phage DNA) | Provides a consistent, complex substrate for performance benchmarking. |
| LacI Forward Mutation Assay Kit | Validated system for quantitatively measuring polymerase error rates. |
| dNTP-Compatible Storage Buffer (e.g., 10 mM Tris-HCl, pH 7.5) | Maintains dNTP stability at -20°C or -80°C for long-term storage. |
This comparative analysis demonstrates significant variability in the purity and performance of dNTPs from different commercial suppliers. These differences have a direct and measurable impact on critical PCR outcomes, including yield, accuracy, and robustness in complex applications. For research that forms the foundation of the broader thesis on dNTP function—particularly studies demanding high fidelity, reproducibility, and sensitivity—selection of a supplier with verified ultra-high purity (≥99.8%), minimal contaminants, and lot-to-lot consistency is paramount. The provided experimental protocols empower researchers to move beyond datasheet claims and conduct rigorous, in-house validation, ensuring the integrity of their PCR amplification research.
The foundational role of deoxynucleoside triphosphates (dNTPs) as the essential building blocks in PCR amplification is well-established. The efficacy of PCR—its fidelity, yield, and specificity—is directly governed by the quality and properties of the dNTPs used. This principle extends powerfully into the field of in vitro transcription (IVT) for mRNA synthesis, where nucleoside triphosphates (NTPs) serve an analogous function. Within this broader thesis on dNTP function, we explore the next-generation evolution of these critical reagents: thermostable dNTPs and CleanCap cap analogs. These advanced analogs are engineered to address key limitations in conventional mRNA production, directly translating lessons from high-fidelity PCR to enable more robust, efficient, and translatable mRNA therapeutics and vaccines.
Core Technology: Defining Next-Generation Analogs
Thermostable dNTPs
Traditional dNTPs can degrade under prolonged thermal cycling, leading to decreased PCR performance. Thermostable dNTPs are chemically modified (e.g., with stabilizing salts or via purification processes) to resist heat-induced decomposition.
- Primary Function: Enhance stability and reliability in long-amplicon or high-cycle-number PCR, which is critical for generating high-quality DNA templates for IVT.
- Impact on mRNA Synthesis: A pure, high-yield PCR template is the first prerequisite for efficient mRNA synthesis. Using thermostable dNTPs ensures template integrity, maximizing the full-length product yield for downstream IVT.
CleanCap Cap Analogs
The 5' cap structure (Cap-0: m7GpppN) is vital for mRNA stability, translation efficiency, and immune evasion. Traditional cap analogs (e.g., ARCA) are co-transcriptionally incorporated but result in ~20% of mRNA being synthesized in the reverse, non-functional orientation. CleanCap technology uses a trinucleotide cap analog (e.g., CleanCap AG, m7GpppA2'-OmepA2'-OG).
- Primary Function: Enables co-transcriptional capping with >90% efficiency and >95% proper orientation by leveraging the initiating nucleotide preference of T7 RNA polymerase.
- Impact on mRNA Synthesis: Streamlines production by combining capping and transcription into a single step, eliminates the need for post-transcriptional enzymatic capping, and produces a superior cap structure that enhances protein expression in vivo.
Quantitative Data Comparison
Table 1: Performance Comparison of Standard vs. Next-Generation dNTPs/NTPs in Template & mRNA Production
| Parameter | Standard dNTPs / NTPs / Cap Analogs | Next-Gen Thermostable dNTPs | Next-Gen CleanCap Analog | Key Implication for mRNA Synthesis |
|---|---|---|---|---|
| Thermal Stability (95°C) | Significant degradation after 60 min. | >95% remaining after 120 min. | Not Applicable | Enables robust, multi-kb template PCR for large mRNA constructs. |
| PCR Yield (10 kb amplicon) | ~50 ng/µL | ~120 ng/µL | Not Applicable | Higher template concentration directly feeds into higher mRNA yield. |
| Co-transcriptional Capping Efficiency | ~80% (with ARCA) | Not Applicable | >90% (with CleanCap AG) | Reduces uncapped mRNA, minimizing immune activation (TLR response). |
| Correct Cap Orientation | ~80% | Not Applicable | >95% | Nearly all mRNA molecules are translationally competent. |
| Resultant mRNA Translation Output | Baseline (100%) | Indirect boost via template quality | 300-500% increase in vitro | Higher protein yield per dose of therapeutic/vaccine. |
Table 2: Research Reagent Solutions for Advanced mRNA Workflow
| Reagent / Material | Function in the Workflow | Key Consideration |
|---|---|---|
| Thermostable High-Fidelity dNTP Mix | Synthesis of plasmid or linear PCR template DNA with ultra-low error rates. | Essential for producing error-free templates encoding the antigen/protein of interest. |
| CleanCap Analog (e.g., CleanCap AG) | Single-step, co-transcriptional 5' capping during IVT. | Must match the first transcribed nucleotide of the template (A for AG analog). |
| Modified NTPs (e.g., N1-Methylpseudouridine, 5-methoxyUTP) | Substitute for standard UTP to decrease immunogenicity and increase translational efficiency. | Used in conjunction with CleanCap for synergistic enhancement of mRNA performance. |
| T7 RNA Polymerase (High-Yield Mutant) | Drives efficient in vitro transcription from the T7 promoter. | Optimized enzyme blends are often paired with CleanCap for maximum yield. |
| DNase I (RNase-free) | Digests the DNA template post-IVT. | Critical purification step to prevent unwanted immune responses to plasmid DNA. |
| Lipid Nanoparticle (LNP) Formulation Kit | For in vivo delivery of synthesized mRNA. | The final delivery vehicle; formulation efficiency dictates in vivo efficacy. |
Detailed Experimental Protocols
Protocol: Generating High-Fidelity DNA Template Using Thermostable dNTPs
Objective: Amplify a linear DNA template containing a T7 promoter, 5' UTR, antigen sequence, and 3' UTR from a plasmid backbone.
Reaction Setup (50 µL):
- 1x High-Fidelity PCR Buffer
- Thermostable dNTP Mix: 200 µM each dNTP
- Forward Primer (specific to T7 promoter): 0.5 µM
- Reverse Primer (specific to 3' UTR/poly-T tail): 0.5 µM
- Plasmid DNA template: 10-50 ng
- High-Fidelity DNA Polymerase: 1.25 units
- Nuclease-free water to 50 µL.
Thermal Cycling:
- 98°C for 30 sec (initial denaturation)
- 35 cycles: 98°C for 10 sec, 65°C for 20 sec, 72°C for 90 sec/kb
- 72°C for 5 min (final extension)
- Hold at 4°C.
Purification: Use a spin-column-based PCR purification kit. Quantify DNA yield via spectrophotometry (Nanodrop). Analyze integrity by agarose gel electrophoresis.
Protocol: One-Step IVT mRNA Synthesis with CleanCap Analog
Objective: Synthesize 5'-capped, modified mRNA in a single reaction.
Reaction Setup (100 µL):
- 1x IVT Reaction Buffer
- NTP Mix: 7.5 mM each ATP, CTP, GTP, and modified UTP (e.g., N1-Methylpseudouridine-5'-TP)
- CleanCap Analog (e.g., CleanCap AG): 6 mM
- Purified linear DNA template (from Protocol 3.1): 1 µg
- T7 RNA Polymerase mix: 10 µL
- Nuclease-free water to 100 µL.
- Optional: Add RNase inhibitor (40 U).
Incubation: 37°C for 2-4 hours.
Template Digestion: Add 2 µL of DNase I (RNase-free) to the reaction. Mix gently and incubate at 37°C for 15 min.
mRNA Purification: Purify using a lithium chloride precipitation method or a dedicated mRNA purification kit (e.g., oligo-dT cellulose). Elute in nuclease-free water. Quantify yield and assess integrity by capillary electrophoresis (e.g., Fragment Analyzer).
Visualizing the Workflow and Impact
Title: Advanced mRNA Synthesis Workflow
Title: Key Drivers of mRNA Quality
Within the broader research on the function of dNTPs in PCR amplification, fidelity—the accuracy of DNA replication by a polymerase—is a paramount concern. The inherent error rate of PCR can introduce mutations that compromise downstream applications, from cloning and sequencing to diagnostic assays and gene therapy vector production. This guide examines the core experimental approach for quantifying fidelity by measuring error rates resulting from the interplay between dNTP substrate quality/concentration and polymerase intrinsic proofreading activity. The systematic comparison of these combinations provides a foundational metric for selecting optimal reagents for high-fidelity amplification.
Key Experimental Protocols for Fidelity Measurement
The gold-standard method for quantifying polymerase error rates is the lacIα forward mutation assay.
2.1. lacIα Forward Mutation Assay Protocol
- Target Amplification: Amplify the lacIα gene (∼190 bp) from a plasmid (e.g., pUC19) using the dNTP/polymerase combination under test. Use a minimum of 16 independent PCR reactions per condition to ensure statistical power.
- Cloning: Purify the pooled PCR products, digest with appropriate restriction enzymes, and ligate into a gapped vector backbone. Transform into an E. coli host strain deficient in lacI and β-galactosidase activity (e.g., NR9165).
- Selection and Screening: Plate transformed bacteria on medium containing X-gal and IPTG. The functional lacIα gene product represses β-galactosidase, resulting in white colonies. Any mutation that inactivates the lacIα gene leads to blue colony formation.
- Calculation: The error rate (E) is calculated using the formula: E = M / (B × N), where M is the number of mutant (blue) colonies, B is the number of total (blue + white) colonies screened, and N is the target length in base pairs. Results are typically expressed as error rate per base per duplication.
2.2. Next-Generation Sequencing (NGS)-Based Error Profiling Protocol
- Amplicon Preparation: Amplify a defined, heterologous genomic target (∼1-2 kb) with the test combination. Perform ultra-deep, high-fidelity library preparation (e.g., using unique molecular identifiers, UMIs) to distinguish PCR errors from sequencing errors.
- Sequencing: Sequence on a platform such as Illumina MiSeq to achieve >10,000x average coverage.
- Bioinformatic Analysis: Use a pipeline (e.g., with DADA2, PEAR) to cluster reads by UMI, generate consensus sequences, and call variants against the known reference sequence. Filter for true PCR-derived mutations.
- Calculation: The error rate is calculated as the total number of mutation events divided by the total number of bases sequenced across all consensus amplicons.
Data Presentation: Quantitative Comparisons
Table 1: Error Rates of Common Polymerase/dNTP Combinations
| Polymerase (Proofreading Status) | dNTP Condition | Reported Error Rate (per bp per duplication) | Assay Used | Key Reference (Recent) |
|---|---|---|---|---|
| Taq (non-proofreading) | Standard purity, 200 µM each | 1.0 x 10⁻⁴ to 2.0 x 10⁻⁴ | lacIα | Potapov & Ong, 2017 |
| Taq | Ultra-pure (HPLC), 200 µM each | ~8.0 x 10⁻⁵ | lacIα | - |
| Phusion (Pfu-derived) | Standard purity, 200 µM each | 4.4 x 10⁻⁷ | lacIα | - |
| Q5 (engineered) | Standard purity, 200 µM each | 2.8 x 10⁻⁷ | NGS-based | Abbot, 2022 |
| KAPA HiFi | Standard purity, 200 µM each | 3.5 x 10⁻⁷ | NGS-based | - |
| Taq + [Mn²⁺] | Standard dNTPs, [Mn²⁺]=0.5 mM | ~1.0 x 10⁻³ | lacIα | Classic control |
| Multiple High-Fidelity | dNTP imbalance (e.g., dGTP at 50 µM, others at 200 µM) | Increase by 5-10 fold | NGS-based | Schacherl, 2023 |
Table 2: Impact of dNTP Quality and Concentration on Fidelity
| Experimental Variable | Test Condition | Effect on Observed Error Rate (Relative to Optimal) | Primary Mechanism |
|---|---|---|---|
| dNTP Purity | Presence of dUTP contaminants | Increased (substitution errors) | Misincorporation of uracil |
| dNTP Purity | Oxidized dNTPs (e.g., 8-oxo-dGTP) | Greatly Increased | Mispairing (8-oxo-dG:A) |
| dNTP Concentration | Sub-optimal (e.g., 20 µM each) | Increased | Increased misincorporation rate due to polymerase stalling |
| dNTP Concentration | Supra-optimal (e.g., 1 mM each) | Slightly Increased | Reduced polymerase selectivity |
| dNTP Ratio Balance | One dNTP at 10% of others | Dramatically Increased | Depletion leading to misincorporation and frameshifts |
Visualizations of Workflows and Pathways
Title: Two primary workflows for PCR fidelity measurement.
Title: Key factors influencing PCR fidelity and error rates.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function & Rationale |
|---|---|
| Ultra-Pure dNTP Solution Set (HPLC-purified) | Minimizes errors caused by dNTP analogs (dUTP) or oxidized bases (8-oxo-dGTP). Essential for baseline high-fidelity work. |
| Proofreading Polymerase Mix (e.g., Q5, Phusion, KAPA HiFi) | Provides intrinsic 3'→5' exonuclease activity to excise misincorporated bases, lowering error rates 50-100x vs. non-proofreading enzymes. |
| dNTP Analog: 8-oxo-dGTP | Used as a positive control to induce a known high error rate in fidelity assays by promoting mispairing. |
| Manganese Chloride (MnCl₂) Solution | A known fidelity disruptor; substituting Mn²⁺ for Mg²⁺ in the buffer dramatically increases error rates, serving as a negative control. |
| lacIα Assay Kit | Commercial kit containing the gapped vector and competent E. coli reporter strain, standardizing the classic mutation assay. |
| NGS Library Prep Kit with UMIs | Enables accurate distinction of PCR-derived mutations from sequencing errors in modern, high-throughput error profiling. |
| Thermostable Pyrophosphatase | Prevents dNTP degradation and inhibition from pyrophosphate accumulation during long or high-cycle number PCR, maintaining fidelity. |
| Balanced Mg²⁺ Buffer | Optimized, dedicated buffer for the polymerase to ensure correct cation concentration, which is critical for polymerase selectivity and fidelity. |
This whitepaper addresses a critical pillar within the broader thesis on the Function of dNTPs in PCR Amplification Research. The central thesis posits that dNTPs are not merely inert substrates but are dynamic co-factors whose concentration, purity, and stability fundamentally govern polymerase fidelity, amplification efficiency, and ultimately, the accuracy of diagnostic results. In clinical diagnostics, where PCR results directly inform patient management, the validation of dNTP quality control (QC) transitions from an academic concern to a stringent regulatory and practical imperative. This document provides an in-depth technical guide for implementing a rigorous dNTP QC protocol to ensure reproducible, sensitive, and reliable diagnostic PCR assays.
The Critical Role of dNTP Quality in Clinical PCR Performance
Suboptimal dNTP quality manifests in assay failure through several mechanisms:
- Reduced Sensitivity: Impurities (e.g., RNA, DNA, nucleases) or unbalanced concentrations can cause premature plateauing, increasing the limit of detection (LoD).
- False Negatives/Npositives: DNase/RNase contamination degrades templates or probes. Chemical modifications (e.g., oxidation of dGTP) can promote misincorporation, affecting sequencing or melting curve analysis.
- Irreproducibility: Lot-to-lot variability in dNTP purity or concentration introduces inter-assay variance, compromising longitudinal monitoring.
Core dNTP QC Parameters and Quantitative Specifications
A comprehensive QC profile must assess the following parameters against established acceptance criteria.
Table 1: Essential dNTP QC Parameters and Acceptance Criteria for Clinical PCR
| QC Parameter | Method of Analysis | Typical Acceptance Criteria | Impact on PCR if Out-of-Spec |
|---|---|---|---|
| Concentration & Balance | HPLC, UV Spectrophotometry | Individual dNTP concentration: ±5% of stated value. Molar ratio of all four dNTPs: 1:1:1:1 ± 5%. | Reduced yield, biased amplification, increased misincorporation. |
| Purity (Chemical) | HPLC (Peak Purity), Mass Spectrometry | Single, symmetric peak for each dNTP. Purity ≥ 99%. Free nucleotides < 0.1%. | Inhibition, aberrant melting curves, sequencing errors. |
| pH & Buffering | pH Meter | pH 7.0 - 7.5 (in aqueous solution). | Optimal Taq polymerase activity is pH-dependent. |
| Sterility & Nuclease Contamination | Microbial Culture, Nuclease Activity Assays | No microbial growth. Undetectable DNase/RNase activity in functional assays. | Template degradation, false negatives. |
| Stability | Real-Time Stability Studies (e.g., -20°C) | ≥ 95% potency after 24 months at recommended storage temperature. | Declining sensitivity over assay lifetime. |
| PCR-Functional Performance | Standardized Amplification Assay (See Protocol 1) | LoD within 95% CI of reference standard. Amplification efficiency (E): 90–105%. No non-specific amplification. | Direct measure of clinical assay suitability. |
Detailed Experimental QC Protocols
Protocol 1: PCR-Functional Validation of dNTPs
- Purpose: To validate dNTP performance in a standardized amplification system mimicking assay conditions.
- Materials: Candidate dNTP mix, reference (QC-pass) dNTP mix, master mix components (excluding dNTPs), validated template (serial dilutions covering LoD), primers, calibrated thermocycler.
- Procedure:
- Prepare two identical master mixes, differing only in the dNTP source (Candidate vs. Reference).
- Amplify template serial dilutions (e.g., 10^6 to 10^1 copies/reaction) in quadruplicate.
- Run qPCR using the clinical assay's cycling conditions.
- Data Analysis: Generate standard curves for each dNTP set. Compare Amplification Efficiency (E = [10^(-1/slope)] - 1), y-intercept (correlates to Cq at 1 copy), and LoD (lowest concentration with ≥95% detection). The candidate dNTPs must produce results statistically non-inferior to the reference.
Protocol 2: HPLC Analysis for Concentration and Purity
- Purpose: To quantify dNTP concentration and identify chemical impurities.
- Method: Anion-exchange or reversed-phase HPLC with UV detection (260 nm).
- Procedure:
- Prepare standard solutions of individual dNTPs of known concentration.
- Inject the candidate dNTP mix (diluted appropriately).
- Data Analysis: Integrate peak areas. Calculate concentration by comparing to standard curves. Assess purity by examining chromatogram for extra peaks (degradation products, free nucleotides) and verifying each dNTP peak is symmetric (Gaussian).
Workflow and Logical Relationships
Diagram 1: Clinical dNTP QC Validation Workflow
Diagram 2: Impact Pathway of Poor dNTP Quality on PCR
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Materials for dNTP QC
| Item | Function in dNTP QC | Key Consideration for Clinical Use |
|---|---|---|
| HPLC System with UV Detector | Gold-standard for quantifying dNTP concentration and assessing chemical purity. | Must be calibrated with NIST-traceable standards. System suitability tests required. |
| Validated Nuclease Assay Kit | Detects contaminating DNase/RNase activity via fluorescent or colorimetric readout. | Use a kit with sensitivity relevant to single-copy PCR templates. |
| PCR Master Mix (dNTP-free) | Used in functional validation to isolate the dNTP variable. | Should be a consistent, well-characterized lot for all QC comparisons. |
| Standardized DNA Template & Primers | Creates a controlled system for functional QC (Protocol 1). | Template should be clinically relevant (e.g., plasmid with viral target) at a quantified copy number. |
| Reference dNTP Mix | Provides the benchmark for functional performance comparison. | Must be from a lot previously validated for the intended clinical assay. Store in single-use aliquots. |
| Sterile, Nuclease-Free Water | Diluent for all solutions to prevent introduction of contaminants. | Certificates of Analysis for nuclease testing are mandatory. |
Within the broader thesis on the Function of dNTPs in PCR Amplification Research, it is critical to examine not only the canonical role of deoxynucleotide triphosphates (dNTPs) as the fundamental building blocks for DNA polymerase but also the emerging technologies that challenge or augment this standard paradigm. This whitepaper explores innovative substrates and methodologies that offer enhanced fidelity, speed, multiplexing capability, and utility in specialized applications such as next-generation sequencing (NGS) and drug development.
Several key technologies are positioning themselves as alternatives or complements to standard dNTP-based amplification.
1.1. Modified Nucleotide Analogs These are structurally altered dNTPs designed to confer specific advantages during PCR.
- Purpose-Built dNTPs (e.g., CleanAmp dNTPs): Thermally labile derivatives that are inactive at room temperature, enabling hot-start PCR without enzyme modification, reducing non-specific amplification.
- Reverse Transcriptase qPCR (RT-qPCR) Enhancers: Additives like Tandem Duplex Stabilizers that work with dNTPs to improve cDNA synthesis efficiency and qPCR robustness, particularly for difficult templates.
1.2. Isothermal Amplification Technologies These methods bypass the thermal cycling requirement of PCR, often using specialized enzyme mixes with alternative nucleotide requirements or mechanisms.
- Recombinase Polymerase Amplification (RPA): Uses recombinase-primer complexes, single-stranded DNA-binding proteins, and strand-displacing polymerases. It typically employs standard dNTPs but operates at constant low temperatures (37-42°C), enabling rapid field-deployable diagnostics.
- Loop-mediated Isothermal Amplification (LAMP): Employs a DNA polymerase with high strand displacement activity and 4-6 primers recognizing 6-8 distinct regions. It often uses dNTPs supplemented with loop-inducing additives like betaine for efficiency.
1.3. Sequencing-Driven Substrates For NGS library preparation, nucleotides are modified to incorporate adapters or unique molecular identifiers (UMIs).
- Tagged dNTPs: Nucleotides conjugated with specific adapter sequences or priming sites, enabling direct "tagmentation" or in-situ labeling during amplification.
- Non-natural Nucleotides for Barcoding: Nucleotides with cleavable linker arms for attaching barcodes post-incorporation, facilitating ultra-multiplexed sequencing.
1.4. Synthetic Biology & Xenobiology The exploration of artificial genetic systems expands the chemical space beyond natural dNTPs.
- XNA (xeno nucleic acid) Synthesis: Polymerases engineered to utilize XNA triphosphates (e.g., FANA, HNA) as substrates, creating nucleic acid polymers with novel biophysical properties for therapeutic aptamer development.
- Base-Modified Triphosphates: dNTPs with extended functional groups (e.g., biotin, fluorophores, azide handles) for click chemistry, enabling direct post-amplification conjugation without complex probe design.
Quantitative Comparison of Technologies
Table 1: Performance Metrics of Alternative Amplification Technologies vs. Standard PCR
| Technology | Primary Nucleotide Substrate | Typical Rxn Temp | Amplification Speed (time to 10^9 copies) | Multiplexing Potential | Primary Application | Key Limitation |
|---|---|---|---|---|---|---|
| Standard PCR | dNTPs | 55-95°C (Cycling) | 30-60 min | High | General cloning, genotyping | Thermal cycling equipment |
| Hot-Start PCR (CleanAmp) | Modified dNTPs | 55-95°C (Cycling) | 30-60 min | High | High-fidelity, sensitive assays | Higher cost per rxn |
| RPA | dNTPs | 37-42°C (Isothermal) | 10-20 min | Low-Moderate | Point-of-care diagnostics | Primer design constraints, sensitivity to inhibitors |
| LAMP | dNTPs + Betaine | 60-65°C (Isothermal) | 15-30 min | Low | Pathogen detection, field testing | Complex primer design (4-6 primers) |
| XNA Synthesis | XNTPs (e.g., FANA-TPs) | 37-55°C | 60-120 min | Low | Synthetic biology, aptamer generation | Limited polymerase availability, high substrate cost |
Table 2: Commercial Reagent Kits Featuring Alternative Nucleotide Workflows (2024 Data)
| Product Name (Supplier) | Core Technology | Claimed Efficiency/ Yield vs. Standard dNTP PCR | Approx. Cost per Rxn (USD) | Key Additive/Modification |
|---|---|---|---|---|
| CleanAmp PCR Kit (TriLink) | Hot-Start via Modified dNTPs | 3-5x reduction in primer-dimer artifacts | $2.50 | CleanAmp dNTPs (thermolabile) |
| TwistAmp Basic (TwistDx) | Recombinase Polymerase Amplification (RPA) | >95% sensitivity in 10 min at 39°C | $4.00 | Recombinase, SSB, strand-displ. polymerase |
| WarmStart LAMP Kit (NEB) | Loop-Mediated Isothermal Amplification | 10^9 copies in <20 min at 65°C | $3.00 | Bst 2.0/3.0 Polymerase, optimized buffer |
| ClickSeq Library Prep | Click Chemistry-Compatible dNTPs | N/A (for sequencing) | $8.00 (library) | 5-Ethynyl-dUTP for post-amplification barcoding |
Detailed Experimental Protocols
Protocol 3.1: Evaluating Hot-Start dNTPs (CleanAmp) vs. Standard dNTPs in Sensitive PCR Objective: To compare non-specific amplification and yield in a low-template-number assay. Materials: See "The Scientist's Toolkit" below. Method:
- Master Mix Preparation (Two Reactions):
- Reaction A (Standard): 10 µL 2X Standard Buffer, 0.4 µL Standard dNTP Mix (10 mM each), 0.5 µL each primer (10 µM), 0.2 µL Taq Polymerase (5 U/µL), 1 µL template (10 copies/µL), nuclease-free water to 19 µL.
- Reaction B (CleanAmp): 10 µL 2X Universal Buffer, 0.4 µL CleanAmp dNTP Mix (10 mM each), 0.5 µL each primer (10 µM), 0.2 µL standard Taq Polymerase (5 U/µL), 1 µL template (10 copies/µL), nuclease-free water to 19 µL.
- Hot-Start Activation: Place both tubes in a pre-heated thermal cycler at 95°C for 2 minutes.
- Initiation: After 2 min, pause the cycler, quickly add 1 µL of 25 mM MgCl2 (for Reaction B) or water (for Reaction A) to each tube. Close lids and resume cycling.
- Thermal Cycling:
- 95°C for 30 sec
- 60°C for 30 sec
- 72°C for 60 sec
- Cycle to 35 rounds.
- Analysis: Run 10 µL of each product on a 2% agarose gel. Quantify band intensity and presence of non-specific bands.
Protocol 3.2: RPA for Rapid Target Detection Objective: Amplify a target sequence from genomic DNA under isothermal conditions. Materials: TwistAmp Basic kit, target-specific primers (30-35 bases long), molecular-grade water, DNA template. Method:
- Rehydrate Pellet: Add 29.5 µL of rehydration buffer (from kit) to the freeze-dried pellet in a tube. Mix gently.
- Add Components: Add 2.4 µL of each primer (10 µM), 1 µL of template DNA (1-10 ng), and nuclease-free water to a final volume of 47.5 µL.
- Initiate Reaction: Add 2.5 µL of 280 mM magnesium acetate (provided) to the tube lid. Briefly centrifuge to mix and start the reaction.
- Incubate: Place the tube in a constant temperature block or heat bath at 39°C for 20 minutes.
- Termination & Analysis: The reaction can be stopped by adding 50 µL of phenol:chloroform:isoamyl alcohol. Purify the aqueous phase and analyze on an agarose gel or using a fluorescent probe readout.
Visualizing Workflows and Relationships
Diagram 1: Landscape of Alternative Amplification Technologies
Diagram 2: Hot-Start vs Standard dNTP Protocol Workflow
The Scientist's Toolkit: Key Reagent Solutions
Table 3: Essential Materials for Experimental Evaluation
| Item (Example Supplier) | Function in Context | Specific Role in Alternative Workflows |
|---|---|---|
| CleanAmp dNTPs (TriLink) | Thermally-activated nucleotides | Remain inert during setup, activate at >90°C, enabling true hot-start PCR without enzyme modification. |
| TwistAmp Basic Kit (TwistDx) | All-in-one RPA reagents | Contains recombinase, SSB, polymerase, and freeze-dried buffer for rapid, isothermal amplification. |
| Bst 2.0/3.0 Polymerase (NEB) | Strand-displacing DNA polymerase | Essential for LAMP and other isothermal methods; synthesizes DNA while displacing downstream strands. |
| 5-Ethynyl-dUTP (Click Chemistry Tools) | Alkyne-modified nucleotide | Incorporated during PCR, allows post-amplification "click" conjugation of azide-labeled fluorophores or barcodes. |
| FANA-TPs (XNA Triphosphates) | Xeno nucleic acid substrates | Used with engineered polymerases (e.g., Kod-RS) to synthesize nuclease-resistant FANA oligonucleotides. |
| Tandem Duplex Stabilizer (ATCC) | PCR additive | Enhances reverse transcription and qPCR stability, often used with standard dNTPs to improve challenging assays. |
| WarmStart LAMP Kit (NEB) | Optimized LAMP reagent mix | Includes Bst polymerase, buffer with betaine, and colorimetric dye for one-step detection. |
Conclusion
dNTPs are far more than simple reaction components; they are the fundamental currency of PCR, directly dictating the success, accuracy, and scope of amplification. From foundational biochemistry to cutting-edge applications, understanding their optimal use—from concentration and balance to source selection—is non-negotiable for robust experimental and diagnostic outcomes. As PCR technology evolves towards greater sensitivity, multiplexing, and point-of-care use, the demand for highly stable, pure, and specialized dNTP formulations will grow. Future research into novel nucleotide analogs and integrated, stabilized master mixes will further streamline workflows, enhancing reproducibility in biomedical research and unlocking new possibilities in molecular diagnostics and therapeutic development.