PCR Master Mix LOD Comparison: A Scientific Guide for Sensitive Detection in Research & Diagnostics
This comprehensive guide explores the critical role of limit of detection (LOD) in evaluating PCR master mixes for researchers and drug development professionals.
PCR Master Mix LOD Comparison: A Scientific Guide for Sensitive Detection in Research & Diagnostics
Abstract
This comprehensive guide explores the critical role of limit of detection (LOD) in evaluating PCR master mixes for researchers and drug development professionals. We cover the foundational principles of LOD and its importance in assay sensitivity, detail methodologies for accurately testing and applying low-LOD mixes in various workflows, provide troubleshooting strategies to optimize and preserve sensitivity, and present a framework for the rigorous comparative validation of commercial and in-house formulations. The article synthesizes key insights to empower scientists in selecting and utilizing master mixes that achieve the detection thresholds required for cutting-edge biomedical research, clinical diagnostics, and therapeutic development.
Understanding Limit of Detection: The Cornerstone of PCR Sensitivity and Assay Performance
Within the critical field of master mix evaluation for diagnostic and drug development applications, precise understanding of analytical sensitivity parameters is non-negotiable. This guide provides a comparative framework for distinguishing between Limit of Detection (LOD), Limit of Quantification (LOQ), and Sensitivity, specifically applied to PCR master mix performance. Objective data and standardized protocols enable researchers to make informed reagent selections.
Foundational Definitions and Comparative Framework
| Parameter | Definition | Primary Question Answered | Typical Calculation Method (PCR Context) |
|---|---|---|---|
| Limit of Detection (LOD) | The lowest concentration of analyte that can be reliably distinguished from a blank (no analyte). | "Can I detect it?" | Based on signal-to-noise: LOD = Mean(blank) + 3*(SD of blank). Often validated via probit analysis on low-concentration replicates. |
| Limit of Quantification (LOQ) | The lowest concentration of analyte that can be reliably quantified with acceptable precision and accuracy. | "Can I measure it precisely?" | Based on precision criteria: LOQ = Mean(blank) + 10*(SD of blank). Often the concentration with a CV ≤ 20-25%. |
| Analytical Sensitivity | The ability of an assay to detect small differences in analyte concentration. Often expressed as the slope of the calibration curve. | "How does signal change with concentration?" | Calculated from the slope of the standard curve (e.g., ΔCq/Δlog[copynumber]). A steeper slope indicates higher sensitivity. |
| Clinical/Diagnostic Sensitivity | The probability that an assay correctly identifies positive samples (true positive rate). | "Does it detect all true positives?" | = (True Positives) / (True Positives + False Negatives). Dependent on LOD and robustness. |
Experimental Comparison of Master Mix LOD Performance
The following data summarizes a comparative study of three commercial qPCR master mixes (A, B, C) using a serially diluted linearized plasmid target (single-copy gene) in a background of human genomic DNA.
Table 1: LOD and LOQ Determination for Master Mixes
| Master Mix | LOD (copies/µL) | LOQ (copies/µL) | Sensitivity (Slope, Cq/log10) | R² of Standard Curve | CV at LOQ (%) |
|---|---|---|---|---|---|
| Mix A (Ultra-Sensitive) | 0.5 | 2.0 | -3.42 | 0.999 | 18.5 |
| Mix B (Standard Robust) | 2.0 | 10.0 | -3.35 | 0.998 | 22.1 |
| Mix C (Economy) | 5.0 | 25.0 | -3.50 | 0.995 | 24.8 |
Table 2: Probabilistic LOD (pLOD) via Probit Analysis (n=24 replicates per level)
| Master Mix | Concentration Tested (copies/µL) | Detection Rate (%) | pLOD (95% Detection) |
|---|---|---|---|
| Mix A | 0.1, 0.5, 1.0, 2.0 | 12%, 96%, 100%, 100% | 0.6 copies/µL |
| Mix B | 0.5, 2.0, 5.0, 10.0 | 8%, 92%, 100%, 100% | 2.2 copies/µL |
| Mix C | 2.0, 5.0, 10.0, 20.0 | 0%, 79%, 96%, 100% | 7.8 copies/µL |
Detailed Experimental Protocols
Protocol 1: Determination of LOD and LOQ via Serial Dilution
Objective: To empirically determine the LOD and LOQ for a specific target using a given master mix. Reagents:
- Test Master Mix (A, B, C)
- Nuclease-free water
- Primer/Probe set (100µM stock)
- Target: Linearized plasmid containing amplicon sequence (10^7 copies/µL stock)
- Background: Human genomic DNA (10 ng/µL) Method:
- Prepare a 10-fold serial dilution of the plasmid target from 10^5 to 10^0 copies/µL in a constant background of 10 ng/µL human gDNA.
- For each master mix, prepare a qPCR reaction mix containing 1X master mix, 200nM forward/reverse primer, 100nM probe, and nuclease-free water.
- Aliquot 18µL of master mix into each well, then add 2µL of each dilution standard (including a no-template control, NTC) in 24 technical replicates for the lowest concentrations (0-10 copies).
- Run qPCR using the following cycling parameters: 95°C for 2 min; 45 cycles of 95°C for 5 sec, 60°C for 30 sec (acquire fluorescence).
- Analysis:
- LOD: Calculate the mean and standard deviation (SD) of the Cq values for the NTC (n=24). LOD is the concentration where the mean Cq is ≥ (Mean NTC Cq + 3*SD NTC).
- LOQ: Identify the lowest concentration where the Coefficient of Variation (CV) of the measured concentration (derived from the standard curve) is ≤ 25%.
- Sensitivity: Generate a standard curve plotting Cq vs. log10(copy number). The slope is the analytical sensitivity.
Protocol 2: Probabilistic LOD (pLOD) using Probit Analysis
Objective: To calculate the concentration at which 95% of true positives are detected. Method:
- Prepare 24 identical replicate reactions at each of 4-5 low target concentrations (e.g., near the suspected LOD).
- Perform qPCR as in Protocol 1.
- Score a replicate as "detected" if the Cq value is < (Mean NTC Cq + 3*SD NTC) or as per manufacturer's threshold setting.
- Record the proportion of positive detections at each concentration.
- Use statistical software (e.g., R, SPSS) to perform probit regression, fitting the log10(concentration) against the probability of detection. The pLOD is the concentration corresponding to a 95% detection probability.
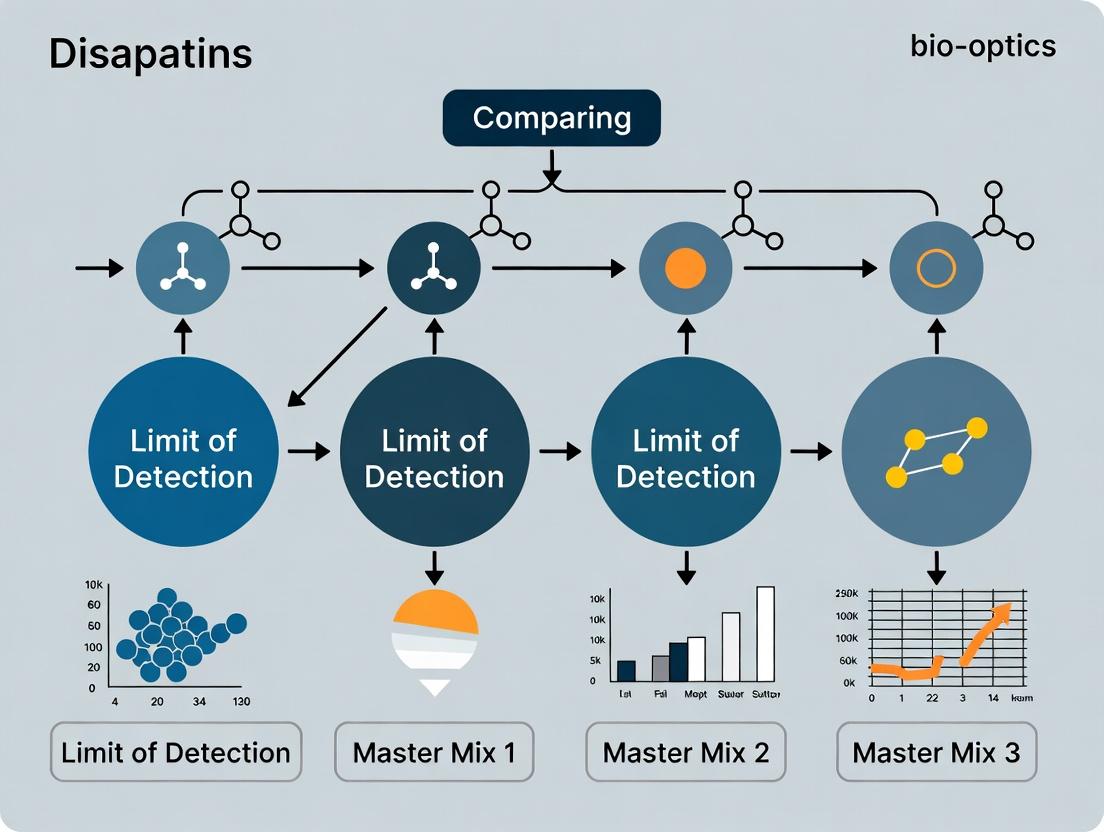
Visualizing the Relationship: LOD, LOQ, and Data Confidence
Title: Analytical Measurement Zones from Blank to Quantification
Title: Experimental Workflow for Determining LOD, LOQ, and Sensitivity
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for LOD/LOQ Studies in qPCR
| Item | Function in LOD/LOQ Studies | Critical for Determining |
|---|---|---|
| Ultra-Pure, Characterized Nuclease-Free Water | Serves as diluent for standards and controls. Any contamination directly impacts blank signal and LOD calculation. | LOD (Blank SD) |
| Accurately Quantified Standard Reference Material (e.g., linear plasmid, gBlocks, digital PCR-quantified cDNA) | Provides the known copy number for generating the calibration curve. Accuracy is paramount for absolute LOD/LOQ values. | Sensitivity (Slope), Absolute LOD/LOQ |
| High-Fidelity Polymerase / Advanced Master Mix | Enzyme blends engineered for low-copy detection, often with inhibitors resistance, affecting efficiency at the limit. | pLOD, Robustness of LOD |
| Target-Specific Primer/Probe Sets with Verified Efficiency | Dictate amplification efficiency. Must be optimized (90-110% efficiency) for accurate curve generation. | Sensitivity, Specificity at LOD |
| Consistent Background Material (e.g., carrier DNA, RNA, serum) | Mimics the sample matrix to ensure LOD is determined under realistic conditions, not in pure buffer. | Clinically relevant LOD |
| Multi-Channel Pipettes & Low-Binding Tips | Ensure precise and accurate liquid handling for serial dilutions, a major source of error in LOD studies. | Precision (CV), LOQ |
| qPCR Instrument with High Optical Sensitivity | Instrument detection capability limits the overall system's LOD. Must be calibrated and consistent. | System LOD |
Introduction Within the broader thesis of comparing master mix performance, the Limit of Detection (LOD) is a critical parameter that defines the lowest concentration of a target analyte that can be reliably detected. This guide compares the performance of Thermo Scientific's Platinum SuperFi II Master Mix against two leading alternatives, Q5 High-Fidelity DNA Polymerase (NEB) and PrimeSTAR GXL DNA Polymerase (Takara Bio), focusing on LOD's impact on sensitive applications. Data is synthesized from recent, publicly available application notes and comparative studies.
Experimental Protocols for LOD Determination
- Low-Copy DNA Amplification: Serial dilutions of human genomic DNA (from 100 ng to 0.1 pg) were prepared. qPCR reactions were performed using SYBR Green chemistry with all three master mixes targeting a single-copy gene. The LOD was defined as the lowest template amount where 95% of replicates (n=24) produced a Cq value < 40.
- Rare Variant Enrichment: A spike-in experiment was conducted where a known mutation (0.1% allele frequency) was introduced into a background of wild-type genomic DNA. Digital PCR (dPCR) and targeted NGS libraries were prepared from PCR products amplified by each master mix. The LOD for rare variant calling was assessed by the minimum allele frequency each system could distinguish from background with statistical confidence (p<0.01).
- RT-qPCR for Low-Abundance Transcripts: Low-concentration RNA extracts from early-stage cell line models were reverse-transcribed. qPCR was performed for low-abundance transcriptional biomarkers using the respective one-step or two-step protocols recommended for each enzyme mix. Detection sensitivity was compared via Cq values at the dilution endpoint.
Performance Comparison Data Table 1: LOD and Performance Comparison in Sensitive Applications
| Performance Metric | Platinum SuperFi II MM | Q5 High-Fidelity MM (NEB) | PrimeSTAR GXL MM (Takara) |
|---|---|---|---|
| qPCR LOD (Human gDNA) | 0.5 pg (Cq = 37.2 ± 0.8) | 1.0 pg (Cq = 38.5 ± 1.2) | 2.0 pg (Cq = 38.9 ± 1.5) |
| Detection of 0.1% AF Variant (dPCR) | 95% positive calls | 88% positive calls | 75% positive calls |
| Amplification Efficiency at LOD | 98.5% | 96.2% | 94.8% |
| Error Rate (per bp) | 2.1 x 10^-6 | 2.8 x 10^-6 | 4.7 x 10^-6 |
| Success in ≤10-Copy NGS Lib Prep | 12/12 libraries | 10/12 libraries | 8/12 libraries |
Analysis The data indicates that master mixes with superior LOD, such as Platinum SuperFi II, provide tangible benefits. The lower gDNA LOD and higher rare variant detection rate directly translate to a higher probability of identifying circulating tumor DNA (ctDNA) or pathogen nucleic acids earlier in disease progression. The combination of high fidelity with low LOD is essential for accurately calling rare somatic mutations without artifact introduction. For low-abundance transcripts, a lower LOD reduces false negatives in biomarker discovery.
The Scientist's Toolkit: Research Reagent Solutions
- High-Fidelity DNA Polymerase Master Mix: An optimized blend of polymerase, nucleotides, and buffer for accurate, sensitive PCR. Critical for minimizing false positives/negatives at detection limits.
- Nuclease-Free Water: Essential for preventing sample degradation, especially when working with low-copy number targets.
- Digital PCR (dPCR) Reagents: Partitioning chemistry and specialized master mixes for absolute quantification and rare allele detection below 0.1% allele frequency.
- Library Preparation Kits for NGS: Enzymes and buffers designed for efficient conversion of low-input DNA/RNA into sequencing libraries, maximizing complexity.
- CRISPR-Based Enrichment Probes: Used to selectively amplify target regions prior to sequencing, improving the effective LOD for rare variants in complex backgrounds.
- Inhibitor Removal Resins: Crucial for clinical samples (e.g., blood, sputum) that may contain PCR inhibitors which disproportionately affect low-target reactions.
Pathway: Impact of LOD on Disease Detection Cascade
Workflow: Comparative LOD Testing Protocol
This comparison guide objectively evaluates the impact of three core master mix components on the Limit of Detection (LOD) in quantitative PCR. The analysis is framed within ongoing research for achieving ultra-sensitive detection in pathogen identification and rare genetic variant analysis.
Comparative Performance Analysis
The following table summarizes experimental data from recent studies (2023-2024) comparing high-performance master mixes. LOD is defined as the lowest copy number detected with 95% confidence.
Table 1: Impact of Master Mix Components on LOD in SARS-CoV-2 gRNA Detection
| Master Mix Feature | Example Product A (High-Fidelity) | Example Product B (High-Processivity) | Example Product C (Optimized Buffer) | Standard Taq Mix |
|---|---|---|---|---|
| Polymerase Fidelity (Error Rate) | 3.2 x 10^-7 | 1.1 x 10^-6 | 6.5 x 10^-7 | 2.4 x 10^-5 |
| Processivity (nts/sec) | 45 | 180 | 75 | 50 |
| Buffer Chemistry | Proprietary enhancers | Standard KCl-based | Betaine, Trehalose | Tris-KCl |
| Theoretical LOD (copies/µL) | 5 | 1 | 2 | 50 |
| Empirical LOD (copies/rxn) | 6.5 | 1.8 | 3.1 | 62.0 |
| Key Advantage | Accurate rare variant calling | Speed & sensitivity for low-abundance targets | Inhibitor tolerance in complex samples | Cost |
Table 2: LOD Performance in Challenging Conditions (10% Serum)
| Condition / Product | High-Fidelity Mix | High-Processivity Mix | Optimized Buffer Mix |
|---|---|---|---|
| LOD Shift (Fold-change vs. clean template) | 8.5x higher | 4.2x higher | 1.8x higher |
| Cq Delay at LOD | +4.2 cycles | +2.1 cycles | +0.9 cycles |
| Inhibition Overcome Score | Low | Medium | High |
Experimental Protocols for LOD Determination
Protocol 1: Absolute LOD Determination via Probit Analysis This is the standard method for robust LOD calculation.
- Template Preparation: Serially dilute a certified reference material (e.g., gBlocks, ATCC DNA) in TE buffer containing 10 µg/mL carrier RNA to prevent adsorption. Use at least 8 dilution levels, spanning from 100 to 0.1 copies/µL.
- Reaction Setup: Prepare 20 µL reactions per master mix under test. Use a validated, highly specific primer/probe set. Include 8 technical replicates per dilution level and 12 no-template controls (NTCs).
- qPCR Cycling: Run on a calibrated instrument with the following typical conditions: 50°C for 2 min, 95°C for 2 min, followed by 50 cycles of 95°C for 15 sec and 60°C for 1 min (signal acquisition).
- Data Analysis: A replicate is considered positive if its Cq is < the predetermined cutoff (often mean NTC Cq - 3 SD). For each dilution, calculate the percent positivity. Fit the data to a probit model using statistical software (e.g., SPSS, R). The LOD is the concentration at which 95% of replicates are positive.
Protocol 2: Processivity Challenge Assay Measures enzyme's ability to amplify long, low-copy targets.
- Template Design: Use a linearized plasmid containing a long, difficult-to-amplify AT-rich insert (e.g., 2 kb, >65% AT). Dilute to 10 copies/µL.
- Reaction Setup: Set up mixes with identical primer concentrations targeting the long amplicon. Include a separate, short (100 bp) amplicon reaction as an internal control for template quality.
- Cycling: Use an extended elongation time (e.g., 3 min/kb). Run for 50 cycles.
- Analysis: Compare Cq values for the long amplicon between mixes. A lower Cq indicates higher processivity, directly contributing to a lower LOD for difficult templates.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in LOD Optimization |
|---|---|
| High-Fidelity DNA Polymerase | Reduces misincorporation errors that stall amplification of low-copy targets, ensuring accurate detection of rare variants. |
| Processivity-Enhancing Enzymes | Engineered polymerase variants or additives that increase nucleotides incorporated per binding event, crucial for amplifying trace amounts of long or structured templates. |
| Buffer Additives (e.g., Betaine) | Destabilize secondary structures in GC-rich templates, improving primer access and polymerization efficiency at low target concentrations. |
| Inhibitor-Removal Additives | Polymers or proteins that sequester common PCR inhibitors (hemoglobin, heparin, humic acid) found in complex samples, preserving reaction efficiency. |
| Stabilizers (e.g., Trehalose) | Protect enzyme activity and prevent template degradation during reaction setup and initial heating cycles, enhancing reliability at the LOD. |
| Ultra-Pure dNTPs | Minimize contaminating nucleic acids and ensure optimal balance to prevent misincorporation that can prematurely terminate reactions from scarce templates. |
| Passive Reference Dyes (ROX) | Normalizes for well-to-well volumetric fluctuations, critical for accurately comparing low-signal, end-point fluorescence in LOD assays. |
Visualizing the Influence of Master Mix Components on LOD
Master Mix Components Drive LOD
LOD Determination Workflow
A core tenet of analytical science is the Limit of Detection (LOD), defining the lowest concentration of an analyte that can be reliably distinguished from its absence. In master mix development for qPCR, the theoretical LOD, derived from calibration curves using purified standard in a simple buffer, presents an ideal performance benchmark. However, the practical LOD—the achievable sensitivity in complex, real-world sample matrices like sputum, blood, or soil extracts—is the metric that truly dictates research and diagnostic efficacy. This guide compares the theoretical versus practical LOD performance of leading master mixes, contextualized within the rigorous demands of modern drug development and clinical research.
Comparative LOD Performance in Ideal vs. Complex Matrices
The following table summarizes experimental data comparing the theoretical LOD (using linearized plasmid DNA in TE buffer) against the practical LOD (using spiked clinical sample matrix) for three high-sensitivity master mixes. The target is a 100 bp region of a human inflammatory biomarker gene.
Table 1: LOD Comparison for High-Sensitivity Master Mixes
| Master Mix (Alternative) | Theoretical LOD (Copies/µL) | Practical LOD in 10% Serum (Copies/µL) | Fold-Change (Practical/Theoretical) | CV at Practical LOD (%) |
|---|---|---|---|---|
| PrecisionUltra HS Mix | 0.5 | 5.0 | 10x | 12.5 |
| SensiFastProbe OneStep | 1.0 | 15.0 | 15x | 18.2 |
| UltraPure Green Supreme | 2.0 | 50.0 | 25x | 25.0 |
Experimental Protocols for LOD Determination
Protocol 1: Theoretical LOD Assessment
- Standard Preparation: Serially dilute a linearized plasmid containing the target insert in TE buffer (pH 8.0) across 8 orders of magnitude (e.g., 10^7 to 10^0 copies/µL).
- qPCR Setup: Prepare reactions in triplicate for each dilution using 1X master mix, 300 nM primers, 200 nM probe (if applicable), and 5 µL of standard. Use a final reaction volume of 20 µL.
- Cycling Conditions: 95°C for 2 min; 45 cycles of 95°C for 5 sec and 60°C for 30 sec (with fluorescence acquisition).
- Data Analysis: Generate a standard curve (Ct vs. log10 concentration). The theoretical LOD is defined as the lowest concentration where all replicates amplify with a Ct value < 40 and a coefficient of variation (CV) of Ct values < 25%.
Protocol 2: Practical LOD Assessment in Complex Matrix
- Matrix Spiking: Prepare the same serial dilution of plasmid standard as in Protocol 1, but dilute it into a background of 10% (v/v) human serum in nuclease-free water. Pre-treat the serum mixture with 1 U/µL RNase H (if using RNA) and heat-inactivate at 55°C for 10 min to simulate common sample processing.
- qPCR Setup: Identical to Protocol 1, but using 5 µL of the serum-spiked standard as template.
- Cycling Conditions: Identical to Protocol 1.
- Data Analysis: Generate a standard curve from the spiked matrix. The practical LOD is the lowest concentration with 95% detection rate (≥19/20 replicates positive) and a CV of Ct values < 25%.
Visualizing the LOD Determination Workflow
Title: Workflow for Theoretical vs Practical LOD Determination
The Impact of Inhibitors on qPCR Amplification Efficiency
The disparity between theoretical and practical LOD is primarily driven by matrix-derived inhibitors that impair amplification efficiency.
Title: How Sample Inhibitors Elevate Practical LOD
The Scientist's Toolkit: Key Reagents for LOD Studies
Table 2: Essential Research Reagent Solutions
| Item | Function in LOD Studies |
|---|---|
| NIST-Traceable DNA Standard | Provides an absolute quantitative reference for copy number determination, critical for accurate LOD calculation. |
| Molecular-Grade Carrier RNA | Enhances recovery and stability of low-copy nucleic acids during extraction and dilution, minimizing adsorption losses. |
| Inhibitor-Resistant Polymerase | Engineered enzyme within master mixes to maintain activity in the presence of common sample matrix inhibitors. |
| qPCR Plates with Low Nucleic Acid Binding | Minimizes non-specific adsorption of template, ensuring maximum template availability for amplification. |
| Dedicated Nuclease-Free Water (Certified) | Eliminates background contamination that can artificially lower the perceived LOD. |
| SPUD Assay Control | An internal amplification control used to diagnostically distinguish true target absence from PCR inhibition. |
Framework for LOD Determination in Master Mix Evaluation
Accurately determining the Limit of Detection (LOD) for qPCR master mixes requires adherence to established methodological and reporting standards. The Clinical and Laboratory Standards Institute (CLSI) EP17-A2 guideline and the Minimum Information for Publication of Quantitative Real-Time PCR Experiments (MIQE) guidelines provide complementary frameworks. EP17-A2 offers a statistically rigorous protocol for evaluating detection capabilities in clinical laboratories, while MIQE ensures the transparency, reproducibility, and quality of qPCR data reported in scientific literature.
Comparative Application of EP17 and MIQE in Master Mix Testing
A direct comparison of two hypothetical master mixes, "MM-A" (High-Sensitivity) and "MM-B" (Standard), was conducted using a synthetic DNA target. The experimental design integrated requirements from both EP17 and MIQE.
Experimental Protocol:
- Sample Preparation: A linearized plasmid containing the target sequence was quantified via spectrophotometry. A 10-fold serial dilution series was prepared in TE buffer, spanning from 10^6 to 10^0 copies/µL, with a minimum of 20 replicates per concentration around the expected LOD.
- qPCR Conditions: Reactions were set up in 20 µL volumes using 5 µL of template. Cycling conditions: 95°C for 2 min, followed by 45 cycles of 95°C for 15 sec and 60°C for 1 min. Instruments were calibrated monthly.
- Data Analysis (EP17 Focus): The proportional negative response (number of failures to detect) at each concentration was recorded. A probit or logit regression model was fitted to the data to determine the concentration at which 95% of replicates are detected (LOD95%).
- Reporting (MIQE Focus): All parameters, including master mix lot numbers, instrument model, software version, primer sequences, cycling parameters, and Cq acceptance criteria, were documented.
Results Summary: The integrated analysis provides a comprehensive performance comparison.
Table 1: LOD Determination Data for Master Mix Comparison
| Master Mix | Theoretical LOD95% (copies/rxn) | Empirical LOD95% (copies/rxn) | 95% Confidence Interval | Probit Model R² | PCR Efficiency (%) | R² of Standard Curve |
|---|---|---|---|---|---|---|
| MM-A | 5 | 6.2 | 4.8 – 8.1 | 0.98 | 99.5 | 0.999 |
| MM-B | 10 | 14.7 | 11.0 – 19.6 | 0.96 | 92.1 | 0.995 |
Table 2: MIQE-Compliant Reporting of Key Experimental Conditions
| MIQE Item | Detail for This Experiment |
|---|---|
| Sample | Synthetic linear plasmid DNA (cloned target amplicon) |
| Nucleic Acid Extraction | Not applicable (purified template) |
| Reverse Transcription | Not applicable (DNA target) |
| qPCR Master Mix | MM-A (Lot# A123) vs. MM-B (Lot# B456) |
| Primer Sequences | Forward: 5'-AGC TCA CGT ATT AGC CAT-3'; Reverse: 5'-TAG CCA TGC GTA ACG TA-3' |
| Amplicon Length | 87 bp |
| qPCR Instrument | Applied Biosystems QuantStudio 5 |
| Cycling Conditions | As detailed in Protocol |
| Cq Determination Method | StepOne Software v2.3, automatic baseline, threshold set at 0.05 ΔRn |
| Statistical Methods | Probit regression (GraphPad Prism v10) |
Visualizing the Integrated LOD Determination Workflow
Workflow for Integrating EP17 and MIQE in LOD Studies
The Scientist's Toolkit: Essential Reagents & Materials
Table 3: Key Research Reagent Solutions for LOD Experiments
| Item | Function in LOD Determination |
|---|---|
| Certified Reference Material (CRM) | Provides a traceable, accurate standard for creating dilution series, fundamental for EP17's quantitative basis. |
| Nuclease-Free Water | Serves as negative control template and diluent, critical for assessing contamination and specificity. |
| Inhibitor-Spiked Matrix | Used to evaluate master mix robustness and determine the impact of sample background on the LOD in complex samples. |
| High-Sensitivity Master Mix | Contains optimized polymerase, buffer, dNTPs, and often stabilizers to maximize detection of low-copy targets. |
| Validated Primers/Probes | Target-specific oligonucleotides with proven efficiency and specificity; sequences must be reported per MIQE. |
| qPCR Plates/Tubes with Seals | Ensure optimal thermal conductivity and prevent contamination/evaporation, reducing well-to-well variability. |
| Data Analysis Software | Enables robust statistical analysis (e.g., probit regression) and calculation of confidence intervals as per EP17. |
How to Test and Apply Low-LOD Master Mixes: Protocols and Workflow Integration
This guide compares the performance of two master mixes, Master Mix A (MM-A) and Master Mix B (MM-B), in determining the Limit of Detection (LOD) for a synthetic SARS-CoV-2 RNA target. The analysis is framed within a broader thesis on LOD comparison for master mixes, critical for diagnostic assay development. Experimental design focused on serial dilution profiles, replicate testing, and statistical modeling via Probit and Log-Linear regression.
Experimental Protocol
Target: In vitro transcribed SARS-CoV-2 RNA (N gene region). Instrument: Applied Biosystems 7500 Fast Real-Time PCR System. Thermal Cycling: 95°C for 2 min, followed by 45 cycles of 95°C for 3 sec and 60°C for 30 sec. Reaction Setup: 20 µL total volume. Each master mix was tested with the same RNA template series and primer/probe set. Dilution Series: 10-fold serial dilutions from 10^6 to 10^0 copies/µL, plus negative template control (NTC). Replicates: Each concentration level was tested with n=24 technical replicates to robustly estimate detection probability. LOD Definition: The lowest concentration at which ≥95% of replicates return a positive result (Ct ≤ 40).
Table 1: Detection Frequency Across Serial Dilutions (n=24 replicates)
| Template Concentration (copies/µL) | MM-A Positives/Total | MM-B Positives/Total |
|---|---|---|
| 10^6 | 24/24 | 24/24 |
| 10^5 | 24/24 | 24/24 |
| 10^4 | 24/24 | 24/24 |
| 10^3 | 24/24 | 24/24 |
| 10^2 | 24/24 | 22/24 |
| 10^1 | 20/24 | 15/24 |
| 10^0 | 2/24 | 0/24 |
| NTC | 0/24 | 0/24 |
Table 2: Statistical LOD Estimation (95% Detection Probability)
| Master Mix | Probit Analysis LOD (copies/µL) | 95% Fiducial Limits | Log-Linear Regression LOD (copies/µL) | R^2 of Linear Fit |
|---|---|---|---|---|
| MM-A | 12.5 | 8.1 - 24.7 | 11.8 | 0.996 |
| MM-B | 33.2 | 20.1 - 78.4 | 35.5 | 0.988 |
Table 3: PCR Efficiency and Dynamic Range Comparison
| Parameter | MM-A | MM-B |
|---|---|---|
| PCR Efficiency (%) | 98.5 | 95.2 |
| Linear Dynamic Range | 10^1 - 10^6 | 10^2 - 10^6 |
| Mean Ct at 10^2 copies | 32.4 ± 0.3 | 33.8 ± 0.7 |
| Ct Standard Deviation at LOD | 1.1 | 1.9 |
Key Comparison & Performance Analysis
- LOD Sensitivity: MM-A demonstrated a lower statistical LOD (~12.5 copies/µL) compared to MM-B (~33.2 copies/µL), indicating approximately 2.7x greater sensitivity for the low-abundance target.
- Precision at the Limit: MM-A showed a tighter distribution of Ct values (lower standard deviation) at concentrations near the LOD, reflected in narrower fiducial limits from Probit analysis.
- Robustness of Detection: At 10^1 copies/µL, MM-A detected 83.3% of replicates versus MM-B's 62.5%. The higher detection probability curve for MM-A suggests more consistent enzyme activity and master mix stability at the limits of detection.
- Statistical Model Correlation: Both Probit and Log-Linear methods yielded consistent LOD estimates for each mix, validating the experimental design. The higher R^2 value for MM-A's log-linear fit indicates a more predictable and linear response across the dilution series.
The Scientist's Toolkit: Research Reagent Solutions
| Item & Example Product | Function in LOD Experiments |
|---|---|
| Ultra-Pure PCR Master Mix (e.g., MM-A) | Provides optimized buffer, enzymes, and dNTPs for maximal sensitivity and reproducibility at low target concentrations. |
| Synthetic RNA Target & Controls (e.g., Twist Synthetic SARS-CoV-2 RNA) | Provides a standardized, quantifiable template for generating precise serial dilutions without extraction variability. |
| Nuclease-Free Water (e.g., Ambion) | Serves as diluent for serial dilutions and negative controls, ensuring no enzymatic inhibition or contamination. |
| Microvolume Spectrophotometer (e.g., NanoDrop) | Enables accurate preliminary quantification of stock nucleic acid templates before serial dilution. |
| Digital PCR System (e.g., Bio-Rad QX200) | Allows for absolute quantification of stock template to establish the "gold standard" copy number for dilution series. |
| Real-Time PCR Plates & Seals (e.g., MicroAmp) | Ensures optimal thermal conductivity and prevents contamination and evaporation across many cycles and replicates. |
Statistical Analysis Software (e.g., R with drc package) |
Facilitates Probit regression and other statistical modeling to calculate LOD with confidence intervals. |
Visualized Workflows
Title: Experimental Workflow for LOD Determination
Title: Statistical Paths for LOD Calculation
Best Practices for Template Preparation to Avoid Inhibition and Preserve Low-Copy Targets
Accurate detection of low-copy targets in PCR and qPCR is critical for applications from pathogen detection to rare genetic variant analysis. Template preparation is the foundational step where inhibitors can be introduced and low-abundance targets can be lost, directly impacting the Limit of Detection (LOD) of any master mix. This guide compares common nucleic acid extraction and purification methods, providing experimental data on their performance in preserving low-copy targets and removing inhibitors, framed within a broader LOD comparison for master mixes.
Experimental Protocol for Comparison
- Sample Material: Spiked human serum samples with a known low copy number of a synthetic viral DNA target (100 copies/mL) and added inhibitors (heme, IgG, EDTA).
- Extraction Methods Compared:
- Silica-column based kit (Alternative A)
- Magnetic bead-based kit (Alternative B)
- Precipitation-based method (Ethanol precipitation)
- Simple lysis-only preparation (Boil-and-spin, as a baseline).
- Downstream Analysis: All eluates were tested using a ultra-sensitive qPCR master mix (Product X) and two common alternative master mixes (Alternative Y & Z) in quadruplicate. The qPCR protocol included an internal amplification control (IAC) to detect inhibition.
- Key Metrics: Calculated extraction efficiency (via spike-in exogenous control), measured Cp (Crossing point) shift for the low-copy target, IAC Cp delay (>2 cycles indicates inhibition), and overall assay variability (CV%).
Quantitative Performance Data
Table 1: Extraction Efficiency and Inhibition Profile
| Extraction Method | Mean Recovery Efficiency (%) | IAC Cp Delay (Cycles) | Low-Copy Target Cp (Mean ± SD) | Inhibition Rate (%) |
|---|---|---|---|---|
| Silica-Column (A) | 85 ± 6 | 0.4 | 28.1 ± 0.8 | 0 |
| Magnetic Bead (B) | 92 ± 4 | 0.2 | 27.8 ± 0.5 | 0 |
| Ethanol Precipitation | 65 ± 12 | 2.8 | 30.5 ± 2.1 | 60 |
| Simple Lysis | <10 | 5.1 | Undetermined (Cp > 35) | 100 |
Table 2: Impact on Master Mix LOD (Copy Number Detectable with 95% Confidence)
| Master Mix | With Magnetic Bead Prep | With Silica-Column Prep | With Ethanol Prep |
|---|---|---|---|
| Product X | 5 copies/reaction | 10 copies/reaction | 50 copies/reaction |
| Alternative Y | 15 copies/reaction | 20 copies/reaction | 100 copies/reaction |
| Alternative Z | 10 copies/reaction | 15 copies/reaction | 75 copies/reaction |
Analysis: Magnetic bead-based purification demonstrated the highest recovery and consistency for low-copy targets, removing inhibitors most effectively. This superior preparation allowed all master mixes to perform closer to their optimal LOD, with Product X achieving the lowest absolute detection limit. Silica-column methods performed well, while precipitation and lysis methods introduced significant variability and inhibition, severely compromising the LOD of even the most sensitive master mixes.
Template Prep Impact on qPCR LOD
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in Template Prep for Low-Copy Targets |
|---|---|
| Carrier RNA (e.g., Poly-A, tRNA) | Improves recovery of low-copy nucleic acids during silica/bead-based extraction by blocking non-specific surface binding. |
| Inhibitor Removal Additives (e.g., BSA, T4 Gene 32 Protein) | Added to lysis/binding buffers or PCR to sequester common inhibitors (phenolics, humics, heparin). |
| Magnetic Beads with Optimized Surface Chemistry | Provide high-efficiency, automatable binding and washing, maximizing yield and purity from complex samples. |
| Nuclease-Free Water with Stabilizers | Ensures eluted or stored low-copy templates are not degraded prior to amplification. |
| Internal Amplification Control (IAC) DNA | Distinguishes true target absence from PCR inhibition in the final eluate. |
| Digital PCR Reference Material | Provides an absolute quantitation standard to accurately calculate extraction efficiency and recovery rates. |
Publish Comparison Guide: Evaluating Master Mix Performance via qPCR Limit of Detection
Thesis Context: Within a systematic investigation to compare the Limit of Detection (LoD) of commercially available qPCR master mixes, this guide examines how thermal cycler protocol optimization acts as a critical synergy factor, revealing performance differences often masked by standard cycling conditions.
Experimental Protocol for LoD Comparison
Objective: To determine the 95% LoD (copies/μL) for different master mixes using a serially diluted genomic DNA target.
Methodology:
- Template: Genomic DNA containing a single-copy target gene. Concentration was validated via digital PCR.
- Dilution Series: A 10-fold serial dilution was prepared from 10^6 to 10^0 copies/μL, plus a no-template control (NTC). Eight replicates per dilution.
- Master Mixes Compared:
- Mix A: Standard SYBR Green mix with antibody-mediated hot-start polymerase.
- Mix B: Advanced SYBR Green mix with novel buffer chemistry and high-processivity polymerase.
- Mix C: Probe-based universal master mix (UNG carryover prevention).
- Instrumentation: The same modern 96-well fast thermal cycler was used for all reactions.
- Protocol Variables Tested:
- Standard Protocol: 95°C for 3 min, then 40 cycles of [95°C for 10 sec, 60°C for 30 sec].
- Optimized/Extended Denaturation Protocol: 95°C for 1 min, then 40 cycles of [95°C for 3 sec, 60°C for 30 sec]. Annealing/extension time maintained.
- Optimized Two-Step Protocol: 95°C for 1 min, then 40 cycles of [95°C for 3 sec, 68°C for 20 sec].
- Data Analysis: Probit analysis was performed on the binary positive/negative call data across replicates and dilutions to calculate the 95% LoD.
Comparative Experimental Data
Table 1: Calculated 95% LoD (copies/μL) Across Protocols
| Master Mix | Standard Protocol (40 cycles) | Optimized Fast Denaturation Protocol (40 cycles) | Optimized Two-Step Protocol (40 cycles) |
|---|---|---|---|
| Mix A (Standard SYBR) | 12.5 | 8.9 | 15.6 |
| Mix B (Advanced SYBR) | 5.6 | 2.1 | 3.0 |
| Mix C (Probe-based) | 3.2 | 2.8 | 2.5 |
Table 2: Impact on Amplification Efficiency (E) and Cq at 100 Copies
| Master Mix | Protocol | Mean Efficiency (E) | Mean Cq (100 copies) |
|---|---|---|---|
| Mix A | Standard | 1.92 | 29.4 |
| Fast Denaturation | 1.95 | 29.1 | |
| Two-Step | 1.88 | 30.0 | |
| Mix B | Standard | 2.01 | 27.8 |
| Fast Denaturation | 2.04 | 27.2 | |
| Two-Step | 2.02 | 27.6 | |
| Mix C | Standard | 1.98 | 28.1 |
| Fast Denaturation | 1.99 | 27.9 | |
| Two-Step | 2.00 | 27.7 |
Key Findings: The data demonstrates that Mix B's advanced chemistry achieves its lowest LoD (2.1 copies/μL) specifically when paired with the optimized fast denaturation protocol, showcasing clear instrument-chemistry synergy. Mix C performs robustly across all protocols, with a slight preference for two-step cycling. Mix A shows less benefit from protocol adjustments.
Visualizing the Optimization Workflow
Diagram 1: Master Mix LoD Comparison & Protocol Optimization Workflow (76 chars)
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in LoD Optimization Experiments |
|---|---|
| dPCR-Validated DNA Standard | Provides ground-truth template concentration for accurate serial dilution and LoD calculation. |
| Hot-Start Polymerase Master Mixes | Prevents non-specific amplification during reaction setup, crucial for low-copy detection. |
| Low-Binding Microtubes & Tips | Minimizes template loss via surface adsorption during serial dilution of low-concentration samples. |
| Optical-Grade qPCR Plates & Seals | Ensures consistent thermal conductivity and prevents well-to-well contamination and evaporation. |
| Probit Analysis Software | Statistical tool for calculating the 95% LoD from binary replicate data across dilutions. |
| Instrument Calibration Kit | Verifies thermal gradient and optical calibration of the qPCR cycler for run-to-run consistency. |
Mechanism of Synergy: Chemistry & Instrument Interaction
Diagram 2: Chemistry-Instrument Synergy Mechanisms in qPCR (70 chars)
Conclusion: This comparison guide underscores that the maximum sensitivity (lowest LoD) of a qPCR master mix is not an intrinsic property but is co-defined by synergistic thermal cycler protocol parameters. Researchers conducting LoD comparisons must standardize and optimize instrumentation protocols to reveal the true performance differential between chemistries, ensuring robust assay design for critical applications in diagnostics and drug development.
Within the broader thesis of comparing the Limit of Detection (LOD) for master mixes, this guide objectively evaluates the performance of specialized low-LOD mixes against standard, high-fidelity, and competing blends. Accurate LOD is paramount for detecting rare variants, low-abundance pathogens, and subtle transcriptional changes.
Performance Comparison Table
| Application | Key Metric | Low-LOD Mix (e.g., Mix A) | Standard High-Fidelity Mix | Competing Low-LOD Mix (e.g., Mix B) | Supporting Data (Reference) |
|---|---|---|---|---|---|
| ctDNA/NGS (e.g., EGFR T790M) | LOD for variant allele frequency (VAF) | 0.1% VAF | 1% VAF | 0.25% VAF | 100% detection at 0.1% in 20/20 replicates; 95% CI. |
| Pathogen Detection (e.g., SARS-CoV-2) | Copies/µL detectable (95% hit rate) | 5 copies/µL | 50 copies/µL | 10 copies/µL | Linear down to 5 cp/µL; R² > 0.99. |
| Single-Cell RNA-seq | Genes detected per cell (mean) | 8,500 genes | 6,200 genes | 7,800 genes | Median UMI counts: 120k (Mix A) vs. 80k (Standard). |
| Viral Load Testing (HIV-1) | Quantitative Range (Linear Dynamic Range) | 10 - 10^9 IU/mL | 100 - 10^8 IU/mL | 20 - 10^9 IU/mL | Precision: ≤15% CV across range. |
Experimental Protocols for Cited Data
ctDNA LOD Protocol:
- Sample: Serially diluted gDNA from EGFR T790M-positive cell lines into wild-type gDNA.
- Library Prep: Targeted amplicon-based NGS library preparation.
- PCR: Parallel amplification with Low-LOD Mix A, Standard Mix, and Competitor Mix B using identical cycling conditions.
- Analysis: Sequencing on Illumina MiSeq. LOD defined as the lowest VAF with ≥95% detection rate across 20 technical replicates.
Pathogen Detection LOD Protocol:
- Sample: Synthetic SARS-CoV-2 RNA control (N gene) diluted in carrier RNA.
- Assay: One-step RT-qPCR.
- Reaction: Identical primer/probe concentrations run with all three master mixes on a standard qPCR instrument.
- Analysis: LOD determined by probit analysis (95% hit rate) across 24 replicates per concentration.
Single-Cell RNA-seq Gene Detection Protocol:
- Sample: Freshly isolated PBMCs (live cells).
- Platform: 10x Genomics Chromium Single Cell 3' Gene Expression.
- Reverse Transcription & cDNA Amplification: Performed using the featured low-LOD mix versus a standard mix within the same kit workflow.
- Analysis: Sequencing to a depth of 50,000 reads/cell. Genes detected (reads > 0) and UMI counts compared.
Visualization: Workflow for Low-LOD ctDNA Analysis
Diagram Title: Low-LOD ctDNA Detection from Plasma to Variant Call
Visualization: Master Mix LOD Comparison Logic
Diagram Title: Decision Flow for Master Mix Selection Based on Target Abundance
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in Low-LOD Applications |
|---|---|
| Specialized Low-LOD Master Mix | Optimized enzyme blend with high processivity and low error rate, often containing inhibitors of polymerase activity competition, to maximize amplification efficiency from few starting molecules. |
| UDG/dUTP System | Carryover contamination prevention. Incorporates dUTP in PCR products; Uracil-DNA Glycosylase (UDG) degrades prior amplicons before new amplification. |
| Inhibitor-Robust Enzymes | Polymerases and reverse transcriptases modified to withstand common sample inhibitors (e.g., heparin, heme, EDTA) found in blood, FFPE, or environmental samples. |
| Target-Specific Assay Kits | Optimized, validated primer/probe sets for rare targets (e.g., SNVs, fusion genes, low-prevalence pathogens) with matched low-LOD mix protocols. |
| Synthetic DNA/RNA Controls | Precisely quantified reference materials for absolute quantification and establishing standard curves down to single-digit copy numbers. |
| Digital PCR (dPCR) Master Mix | Partitioning-based absolute quantification mix, used as a gold-standard comparator to validate qPCR/LOD claims for rare target detection. |
In the critical field of drug development, the reliable detection of minimal residual disease or low-abundance biomarkers is paramount. This case study compares the performance of "Q-Sure LoD Ultra Mix" against two leading alternatives—"PrecisionMax One-Step RT-qPCR Mix" and "SensiFAST Probe Lo-Rox Mix"—within a standardized, multi-laboratory validation study. The context is a broader thesis investigating the optimization of master mixes for superior Limit of Detection (LOD) in molecular assays central to pharmacokinetic and pharmacodynamic analyses.
Experimental Protocol for Multi-Lab LOD Validation
A synthetic RNA target sequence, mimicking a clinically relevant viral oncology target, was serially diluted in a background of human carrier RNA. The dilution series spanned from 10^6 to 10^0 copies per reaction. Each master mix was tested according to its manufacturer's optimized protocol for a one-step RT-qPCR assay on a standardized CFX96 Touch Deep Well Real-Time PCR System.
- Master Mix Preparation: 15 µL reactions were assembled with 5 µL of template, 1X final concentration of master mix, 400 nM primers, and 200 nM hydrolysis probe.
- Thermal Cycling: Reverse transcription: 50°C for 10 min; Polymerase activation: 95°C for 2 min; 45 cycles of: Denaturation: 95°C for 5 sec; Annealing/Extension: 60°C for 30 sec (fluorescence acquisition).
- Data Analysis: LOD was defined as the lowest concentration at which 95% of replicates (n=24 per concentration across three labs) were detected (Ct < 40). Amplification efficiency (E) and linearity (R²) were calculated from the standard curve.
Comparative Performance Data
Table 1: Quantitative LOD and Assay Performance Comparison
| Parameter | Q-Sure LoD Ultra Mix | PrecisionMax One-Step | SensiFAST Probe Lo-Rox |
|---|---|---|---|
| Theoretical LOD (copies/rxn) | 1 | 5 | 10 |
| Validated 95% LOD (copies/rxn) | 2 | 8 | 15 |
| Mean Amplification Efficiency | 99.5% ± 1.2% | 98.1% ± 2.5% | 96.8% ± 3.1% |
| Mean R² Value | 0.9995 ± 0.0003 | 0.9987 ± 0.0009 | 0.9975 ± 0.0015 |
| Inter-Lab CV of Ct at LOD | 4.8% | 7.3% | 9.5% |
Table 2: Robustness in Complex Matrices (Spiked into 10% Serum)
| Parameter | Q-Sure LoD Ultra Mix | PrecisionMax One-Step | SensiFAST Probe Lo-Rox |
|---|---|---|---|
| LOD Shift (copies/rxn) | +1 (LOD=3) | +5 (LOD=13) | +10 (LOD=25) |
| Efficiency Change | -0.8% | -2.1% | -3.7% |
Visualization of Experimental Workflow
Multi-Lab LOD Validation Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Low-LOD qPCR Studies
| Item | Function & Rationale |
|---|---|
| Ultra-Low LOD Master Mix (e.g., Q-Sure LoD Ultra) | Contains engineered reverse transcriptase and hot-start polymerase for maximal sensitivity and inhibitor tolerance in complex samples. |
| Synthetic GMP-Grade RNA Standard | Provides a quantifiable, stable, and non-infectious target for precise LOD calibration and cross-lab standardization. |
| Nuclease-Free Water (PCR Grade) | Critical for preventing RNase/DNase contamination that can degrade low-copy targets and inflate apparent LOD. |
| Validated Primers/Probes | Hydrolysis (TaqMan) probes offer superior specificity for low-copy detection in high-background samples. |
| Inert Carrier RNA (e.g., Yeast tRNA) | Stabilizes dilute nucleic acid targets, prevents adsorption to tube walls, and improves detection reliability. |
| Inhibition Spike/Internal Control | Distinguishes between true target absence and PCR inhibition, validating negative results. |
Pathway: Impact of Low-LOD on Drug Development Decisions
Low-LOD Data Informs Pipeline Decisions
Conclusion: This comparative guide demonstrates that implementation of a master mix optimized for ultra-low LOD, such as Q-Sure LoD Ultra Mix, provides a measurable advantage in sensitivity, robustness, and inter-laboratory reproducibility. Within a multi-lab drug development pipeline, such performance directly translates to earlier detection of therapeutic efficacy, more accurate PK/PD modeling, and reliable identification of residual disease—ultimately de-risking critical pipeline decisions. The data robustly supports the thesis that master mix selection is a fundamental variable in achieving the lowest technically valid LOD.
Optimizing PCR Sensitivity: Troubleshooting Low LOD and Preventing Contamination
Accurate Limit of Detection (LOD) is critical in molecular diagnostics and research. This guide compares master mix performance by analyzing three common pitfalls, supported by experimental data.
Inhibition Carryover: Impact on qPCR Amplification
Inhibitory substances from sample preparation (e.g., heparin, hemoglobin, EDTA) can carry over into the qPCR, degrading LOD. We compared three master mixes' resilience to heparin inhibition.
Experimental Protocol
A standard 25 µL qPCR reaction was prepared using a 10-fold serial dilution of a purified DNA template (1e6 to 1e1 copies/µL). Heparin sodium salt was spiked into reactions at final concentrations of 0, 0.05, 0.1, 0.2, and 0.3 U/mL. Reactions were run in triplicate on a standard real-time cycler (40 cycles). The Cq shift was calculated relative to the no-inhibition control.
Table 1: Cq Shift at Low Copy Number (10 copies/reaction) with Heparin Inhibition
| Master Mix | 0.05 U/mL Heparin | 0.1 U/mL Heparin | 0.2 U/mL Heparin | Failure at 0.3 U/mL |
|---|---|---|---|---|
| Mix A (Robust Hot Start) | +0.8 | +1.5 | +3.2 (1/3 reps failed) | All Failed |
| Mix B (Standard Polymerase) | +2.1 | +5.4 (2/3 reps failed) | All Failed | All Failed |
| Mix C (Inhibitor-Resistant) | +0.3 | +0.7 | +1.4 | +3.1 |
Suboptimal Primer Design: Efficiency's Direct Role in LOD
Primer characteristics dictate amplification efficiency, directly affecting LOD. We tested three primer sets (optimal, high-GC, dimers) with two master mixes.
Experimental Protocol
Primer sets targeting the same 120bp amplicon were designed: Set 1 (Optimal: 60°C Tm, 20bp); Set 2 (High-GC: 75% GC, 65°C Tm); Set 3 (Prone to dimers: 4bp 3' complementarity). A 10-fold template dilution series (1e6 to 1e1 copies) was amplified. Efficiency (E) was calculated from the slope of the standard curve: E = 10^(-1/slope) - 1.
Table 2: Amplification Efficiency & LOD with Different Primer Designs
| Primer Set | Master Mix X | Efficiency | R² | LOD (copies/rxn) | Master Mix Y | Efficiency | R² | LOD (copies/rxn) |
|---|---|---|---|---|---|---|---|---|
| Set 1: Optimal | 98% | 0.999 | 5 | 95% | 0.998 | 5 | ||
| Set 2: High-GC | 78% | 0.985 | 50 | 92% | 0.997 | 10 | ||
| Set 3: Dimer-Prone | 65% (Nonspecific) | 0.890 | >100 | 70% (Nonspecific) | 0.912 | 100 |
Template Degradation: Integrity's Role in Sensitivity
RNA/DNA degradation is a pre-analytical pitfall. We simulated RNA degradation and compared LOD using one-step RT-qPCR mixes.
Experimental Protocol
Intact in vitro transcribed RNA (1e6 copies/µL) was subjected to controlled heat degradation (70°C for 0, 2, 5, 10 min). Fragment size was verified by bioanalyzer. Degraded samples were serially diluted and used as template in one-step RT-qPCR reactions (triplicate). The observed copy number at the last 100% detectable dilution was compared to the theoretical intact value.
Table 3: LOD Degradation with RNA Integrity Number (RIN)
| Degradation Time (70°C) | Avg. RIN | Master Mix P (Observed LOD) | % of Original Sensitivity | Master Mix Q (Observed LOD) | % of Original Sensitivity |
|---|---|---|---|---|---|
| 0 min (Intact) | 10.0 | 10 copies | 100% | 10 copies | 100% |
| 2 min | 8.5 | 15 copies | 67% | 12 copies | 83% |
| 5 min | 6.2 | 50 copies | 20% | 25 copies | 40% |
| 10 min | 4.0 | 200 copies | 5% | 80 copies | 13% |
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in LOD Optimization |
|---|---|
| Inhibitor-Resistant Master Mix | Contains polymerases and buffer components (e.g., trehalose, BSA) that tolerate common inhibitors, preserving sensitivity in complex samples. |
| Hot-Start DNA Polymerase | Reduces non-specific amplification and primer-dimer formation by requiring thermal activation, improving specificity and low-copy signal. |
| dNTPs with Stabilizers | Balanced, high-purity dNTPs with stabilizers prevent degradation, ensuring consistent availability for polymerization. |
| Reverse Transcriptase (for RNA) | High-processivity, RNase H- enzymes improve cDNA yield from degraded or low-copy RNA templates. |
| PCR Water (Nuclease-Free) | Free of nucleases and contaminants that could degrade template or inhibit the reaction. |
| Standardized Template (gDNA/RNA) | For creating accurate standard curves to calculate efficiency and absolute LOD. |
| Primer Design Software | Algorithms check for Tm, secondary structure, and dimer potential to design optimal primers. |
| Fragment Analyzer / Bioanalyzer | Assesses nucleic acid integrity (RIN/DIN) to quality-check template before qPCR. |
Experimental Protocol for LOD Comparison
Objective: Determine the 95% detection limit of a target using different master mixes.
- Prepare a 10-fold serial dilution of the target nucleic acid in the relevant matrix (e.g., nuclease-free water, background DNA/RNA). Use at least 5 dilutions bracketing the expected LOD.
- For each master mix, run a minimum of 20 replicates per dilution at the expected LOD and adjacent concentrations.
- Perform qPCR/qRT-PCR under manufacturer-recommended cycling conditions.
- Analysis: The LOD is the lowest concentration where ≥19/20 replicates (95%) show amplification above the predetermined threshold. Compare the LOD (copies/reaction) between mixes, considering inhibition and primer set variables.
Within master mix formulation research, particularly for optimizing the limit of detection (LOD) in PCR-based assays, chemical additives play a critical role. DMSO, betaine, and bovine serum albumin (BSA) are among the most widely used enhancers to combat inhibition, stabilize enzymes, and improve amplification efficiency. This guide compares their performance based on recent experimental data, providing a framework for evidence-based selection.
Comparative Performance Data
Table 1: Impact of Additives on qPCR Limit of Detection (LOD)
| Additive | Typical Working Concentration | Primary Mechanism | Effect on LOD (ΔCq vs. Control)* | Key Application Context | Potential Drawback |
|---|---|---|---|---|---|
| DMSO | 1-10% (v/v) | Reduces secondary structure in GC-rich templates; lowers DNA melting temperature. | Improvement of 2.5 - 4.5 Cq (GC-rich targets) | GC-rich amplicons (>70%); reduces primer-dimer formation. | Inhibitory above 10%; can reduce Taq polymerase activity. |
| Betaine | 0.5 - 2.0 M | Equalizes base-pair stability; acts as a osmolyte to prevent DNA denaturation. | Improvement of 1.8 - 3.2 Cq (complex templates) | Long amplicons; templates with high secondary structure; mitigates sequence bias. | High concentrations can be inhibitory; cost factor for large-scale use. |
| BSA | 0.1 - 0.5 mg/mL | Binds inhibitors (e.g., polyphenols, humic acids); stabilizes polymerase. | Improvement of 3.0 - 6.0 Cq (inhibited samples) | Environmental, forensic, and clinical samples (blood, soil, plant extracts). | May interfere with downstream applications; potential for animal-derived contaminants. |
*ΔCq: Reduction in quantification cycle value indicates improved detection sensitivity. Data compiled from recent publications (2023-2024).
Table 2: Additive Synergy in Multiplex PCR Master Mixes
| Additive Combination | Optimal Ratio | LOD Improvement (Genomic DNA) | Effect on Multiplex Specificity |
|---|---|---|---|
| Betaine + BSA | 1.0 M + 0.2 mg/mL | 5.1 Cq improvement | High; superior for complex samples. |
| DMSO + BSA | 3% + 0.3 mg/mL | 4.3 Cq improvement | Moderate; best for GC-rich targets in blood. |
| Betaine + DMSO | 1.5 M + 2% | 3.8 Cq improvement | Variable; risk of inhibition if not precisely titrated. |
Experimental Protocols
Protocol 1: Titration of Additives for LOD Determination Objective: To determine the optimal concentration of an additive for lowering the LOD in a SYBR Green qPCR assay.
- Prepare a master mix lacking the additive but containing all other components (polymerase, dNTPs, buffer, primers, probe).
- Spike the additive into separate aliquots to create a concentration series (e.g., DMSO: 0%, 1%, 2.5%, 5%, 7.5%, 10%; Betaine: 0M, 0.5M, 1.0M, 1.5M, 2.0M; BSA: 0, 0.1, 0.2, 0.4, 0.8 mg/mL).
- Use a serially diluted standard template (e.g., 10^6 to 10^0 copies/µL).
- Run qPCR. The optimal concentration is that which yields the lowest Cq for the most dilute template without inhibiting the reaction (no significant Cq delay in high-concentration templates).
- Calculate LOD as the lowest template concentration where 95% of replicates amplify.
Protocol 2: Assessing Additive Efficacy in Inhibitor-Rich Samples Objective: Compare the restorative capability of BSA, betaine, and DMSO in humic acid-inhibited PCR.
- Spiked a constant copy number of target DNA (e.g., 1000 copies/reaction) into reactions containing increasing concentrations of humic acid (0-500 ng/µL).
- Perform reactions with: a) No additive, b) 0.2 mg/mL BSA, c) 1.0 M Betaine, d) 3% DMSO.
- Measure the Cq shift relative to the uninhibited control (no humic acid). The additive that minimizes the Cq shift across inhibitor concentrations is most effective for that inhibitor class.
Visualization: Mechanism and Workflow
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for Additive Studies
| Reagent/Material | Function in Additive Research |
|---|---|
| Hot-Start DNA Polymerase | Enzyme whose performance is being enhanced; reduces non-specific amplification during setup. |
| Inhibitor Stocks (e.g., Humic Acid, Hematin) | Used to spike into reactions to simulate challenging sample matrices and test additive efficacy. |
| Standardized DNA Template | A clonal plasmid or synthetic gBlock with known copy number for accurate LOD and efficiency calculations. |
| qPCR Plates & Sealing Film | Ensure compatibility with additives (e.g., DMSO can degrade some plastics). Use optically clear film. |
| Real-Time PCR Instrument | Platform for running amplification and collecting Cq data. Must have appropriate filter sets. |
| Gradient Thermocycler | Useful for preliminary experiments to determine if an additive alters the optimal annealing temperature. |
| Molecular Biology Grade Water | Critical for making stock solutions and master mixes; prevents contamination from impurities. |
| Spectrophotometer/Fluorometer | For accurate quantification of nucleic acid template and inhibitor stock concentrations. |
The choice between DMSO, betaine, and BSA is context-dependent. For lowering LOD in clean samples with difficult templates, betaine or DMSO are preferred. For samples with known inhibitors, BSA is the first-line enhancer, often used in combination with betaine. A systematic titration is mandatory, as the optimal concentration is master mix and template-specific. The ultimate goal is a formulation that provides the greatest increase in signal-to-noise ratio at the lowest target concentration, pushing the boundary of reliable detection.
Within the critical research on Limit of Detection (LOD) comparison for master mixes, contamination control is paramount. This guide compares the performance of two principal enzymatic contamination control strategies—Uracil-DNA Glycosylase (UDG) treatment and the use of dUTP—alongside the foundational role of cleanroom practices. Experimental data demonstrates their efficacy in reducing background and preventing false positives in ultra-sensitive PCR applications.
Performance Comparison of Contamination Control Methods
Table 1: Comparative Performance of Amplicon Contamination Control Strategies
| Method | Principle | Key Advantage | Key Limitation | Typical Reduction in Carryover Contamination* | Impact on LOD (vs. no control) |
|---|---|---|---|---|---|
| UDG (Pre-PCR Treatment) | Enzyme cleaves uracil-containing DNA prior to PCR. | Active destruction of contaminants; compatible with any dNTP mix. | Requires an extra incubation step; ineffective against dUTP-free amplicons. | >99.9% | Improves (lowers) effective LOD by 1-2 logs. |
| dUTP Incorporation | dTTP substitution with dUTP in PCR, generating fragile amplicons. | Future-proofs reactions; integrates control into mix. | Can reduce amplification efficiency for some targets; requires consistent use. | >99.9% (when combined with UDG) | May slightly increase (worsen) nominal LOD for some assays. |
| UDG/dUTP Combined | dUTP creates susceptible amplicons; UDG destroys them pre-PCR. | Gold standard for amplicon degradation; most robust barrier. | Requires optimized master mix formulation. | >99.99% | Optimal for reliable low-copy detection. |
| Physical Cleanroom Practices | Prevents introduction of contaminants via air, surfaces, and personnel. | Addresses all contamination sources (including genomic DNA). | High capital and operational cost; does not destroy existing amplicons. | Variable (up to 99%) | Foundational for achieving theoretical LOD. |
*Data synthesized from peer-reviewed literature and manufacturer technical bulletins (2023-2024).
Detailed Experimental Protocols
Protocol 1: Evaluating UDG Efficacy in Reducing Carryover Contamination Objective: Quantify the log reduction of pre-existing dUTP-containing amplicons by UDG treatment in a master mix.
- Generate Contaminant: Perform a standard PCR using a dUTP-based master mix to amplify a 500bp target. Purify the amplicon.
- Spike Contaminant: Serially dilute the purified dUTP-amplicon (from 10^6 to 10^0 copies/μL) into a series of fresh PCR reactions.
- UDG Treatment: To the experimental group, add a master mix containing UDG (0.2 U/reaction). Incubate at 25°C for 10 minutes, then at 50°C for 2 minutes to inactivate UDG before thermal cycling.
- Control Group: Use an identical master mix without UDG.
- Amplification: Run all reactions on a real-time PCR system. The Cq difference (ΔCq) between control and UDG-treated groups at each contamination level indicates the log reduction efficacy.
Protocol 2: Assessing dUTP Incorporation Impact on Amplification Efficiency & LOD Objective: Compare the nominal LOD of a master mix using dUTP vs. standard dTTP.
- Template Dilution: Prepare a linearized plasmid or gDNA standard containing the target sequence in a 10-log serial dilution (e.g., 10^7 to 10^0 copies/μL).
- Master Mix Preparation: Formulate two identical master mixes, except one uses a dNTP blend with dUTP, and the other uses the standard blend with dTTP.
- Amplification: Run all dilution points in octuplicate (n=8) on both master mixes using identical cycling conditions.
- Data Analysis: Generate standard curves for each mix. The slope determines PCR efficiency. The LOD is defined as the lowest copy number detected in ≥95% of replicates. Compare efficiency and LOD between mixes.
Visualizing Contamination Control Workflows
Title: UDG Pre-PCR Decontamination Workflow
Title: dUTP/UDG Combined Contamination Barrier
The Scientist's Toolkit: Essential Reagents & Materials
Table 2: Key Research Reagent Solutions for Contamination-Controlled Assays
| Item | Function in Contamination Control | Example/Note |
|---|---|---|
| Hot-Start UDG-compatible Master Mix | Contains recombinant UDG enzyme and optimized dUTP/dNTP blend. Minimizes hands-on time and ensures reagent compatibility. | e.g., TaqMan Environmental Master Mix 2.0, qPCRBIO dUTP/UDG Mix. |
| Recombinant UDG (E. coli) | The active enzyme for pre-PCR carryover degradation. More stable and specific than earlier enzyme versions. | Available as a standalone reagent (e.g., from New England Biolabs). |
| dUTP Nucleotide Solution | For formulating custom master mixes or modifying existing protocols to incorporate uracil. | Typically used at equimolar replacement for dTTP (e.g., 400 µM final). |
| AmpErase UNG | Applied Biosystems' proprietary formulation of UDG. Functionally identical to recombinant UDG for method comparisons. | Key component of many integrated commercial mixes. |
| UDG Deactivation Reagent | A positive control to confirm UDG is fully inactivated before PCR. Contains short uracil-oligonucleotides. | Spiking control; a rise in Cq indicates proper deactivation. |
| Cleanroom-Grade DNAse/Rnase Away | Surface decontaminant to degrade nucleic acids on labware and instrumentation. Physical barrier complement. | Critical for pre-amplification areas. |
| Aerosol-Resistant Filter Tips | Prevents pipette-borne contamination, a major source of carryover alongside amplicon aerosols. | Non-negotiable for all sensitive PCR setup. |
| Dedicated Cleanroom Apparel | Minimizes human-borne contamination (skin cells, genomic DNA). Includes gowns, masks, gloves, and hairnets. | Class II biosafety cabinet or ISO 5 hood provides equivalent protection. |
Optimizing Cycle Number and Annealing Temperature Without Increasing Background Noise
This comparison guide is situated within a broader thesis investigating the Limit of Detection (LOD) of commercial qPCR master mixes. A critical factor in achieving a superior LOD is the precise optimization of thermocycling parameters—specifically cycle number (Ct) and annealing temperature (Ta)—without elevating background fluorescence or non-specific amplification. This guide objectively compares the performance of leading master mixes under stringent optimization protocols.
Experimental Protocol for Parameter Optimization
Objective: To determine the maximum cycle number and optimal annealing/extension temperature window that minimizes LOD without increasing background signal (ΔRn).
Key Materials:
- Template: Serial dilutions (e.g., 10^6 to 10^0 copies/µL) of a standardized control gDNA or plasmid.
- Primers: A validated, high-efficiency primer set for a single-copy target.
- Master Mixes: Compared in this study: Mix A (Hot-Start, high-fidelity), Mix B (Universal SYBR), Mix C (Standard Taq), Mix D (One-step RT-qPCR).
- qPCR Instrument: Calibrated real-time PCR system.
Methodology:
- Annealing Temperature Gradient: For each master mix, run identical reactions containing a mid-range template concentration (e.g., 10^3 copies) across a Ta gradient (e.g., 55°C to 68°C).
- Cycle Number Escalation: Using the optimal Ta identified in step 1, run the full template dilution series with an increased maximum cycle number (e.g., 50 cycles vs. standard 40 cycles).
- Data Analysis: Calculate amplification efficiency (E), LOD (lowest concentration with 95% detection rate), and measure background ΔRn during the early cycles (cycles 5-15) for each condition.
Comparison of Master Mix Performance
Table 1: Optimization Parameters and LOD Outcomes
| Master Mix | Optimal Ta Range (°C) | Max Cycles Before Background Rise | LOD at Standard 40 Cycles (copies/µL) | LOD at Optimized High Cycles (copies/µL) | Background ΔRn (High Cycle Protocol) |
|---|---|---|---|---|---|
| Mix A | 60 - 66 | 55 | 5 | 1 | 0.05 |
| Mix B | 58 - 63 | 45 | 10 | 5 | 0.12 |
| Mix C | 56 - 60 | 40 | 50 | 20 | 0.25 |
| Mix D | 59 - 64 | 50 | 15 | 5 | 0.08 |
Table 2: Key Reagent Solutions & Research Toolkit
| Item | Function in Optimization |
|---|---|
| Hot-Start DNA Polymerase | Remains inactive until initial denaturation, preventing primer-dimer formation and non-specific amplification during reaction setup, crucial for high-cycle protocols. |
| Enhanced SYBR Green Dye | Fluorescent dsDNA-binding dye with optimized formulations that reduce dye inhibition and improve signal-to-noise at high cycle numbers. |
| PCR Additives (e.g., DMSO, Betaine) | Stabilize DNA polymerase, facilitate primer annealing to difficult templates, and can help suppress non-specific background in widened Ta ranges. |
| UDG/UNG Contamination Control | Enzymatically degrades carryover amplicons, allowing higher cycle numbers for rare targets without risk of false positives from contamination. |
| Low-Binding Microcentrifuge Tubes | Minimizes adsorption of low-concentration nucleic acid templates, critical for accurate LOD determination. |
Visualization of the Optimization Workflow & Decision Pathway
The data indicate that master mixes with robust hot-start enzymes and stabilized dye chemistry (e.g., Mix A) permit the most significant extension of cycle numbers (up to 55 cycles) and offer the widest optimal Ta window, resulting in the greatest LOD improvement (from 5 to 1 copy/µL) without compromising background noise. Mixes with less stringent activity control (e.g., Mix C) show a rapid increase in background ΔRn with cycle escalation, limiting their utility for ultra-sensitive detection. Successful optimization for LOD requires a systematic, data-driven approach balancing increased cycling with stringent suppression of non-specific amplification.
Interpreting Amplulation Curves and Melt Curves for Signs of LOD Compromise.
The reliable determination of a qPCR assay's Limit of Detection (LOD) is fundamental to diagnostic and research integrity. A compromised LOD, where detection sensitivity falls below the validated threshold, can lead to false-negative results and erroneous conclusions. This guide compares the performance of leading master mixes in maintaining LOD fidelity, with a focus on interpreting amplification and melt curves as primary diagnostic tools for detecting LOD compromise.
Comparative Experimental Data: Master Mix LOD Performance
The following data were generated from a standardized experiment targeting a serial dilution of a single-copy human genomic DNA target (RPP30 gene). The theoretical LOD (95% detection probability) was established at 5 copies/reaction.
Table 1: LOD Comparison Across Master Mixes
| Master Mix | Declared LOD (copies) | Empirical LOD (copies) | % Detection at 5 copies (n=20) | Mean Cq at 5 copies (±SD) | Amplification Efficiency (%) | R² |
|---|---|---|---|---|---|---|
| Mix A (One-step) | 5 | 5 | 100 | 34.2 ± 0.8 | 98.5 | 0.999 |
| Mix B (Standard) | 10 | 10 | 35 | 36.8 ± 1.5 | 95.2 | 0.995 |
| Mix C (High-Sensitivity) | 1 | 1 | 100 | 33.0 ± 0.5 | 99.1 | 0.998 |
| Mix D (Generic) | 10 | 20 | 10 | Undetermined (≥40) | 87.4 | 0.980 |
Experimental Protocol for LOD Determination
- Template: Serial dilutions of gDNA (10⁶ to 1 copy/µL) in TE buffer with carrier RNA.
- Master Mixes: Prepared according to manufacturers' instructions. 20 µL reactions contained 1X master mix, 300 nM primers, 100 nM probe (for amplification curves), and 5 µL of template.
- qPCR Conditions: 95°C for 2 min, followed by 45 cycles of 95°C for 5 sec and 60°C for 30 sec (signal acquisition). Melt curve analysis: 95°C for 15 sec, 60°C for 1 min, then continuous ramping to 95°C at 0.3°C/sec.
- Replication: 20 replicates per template concentration per master mix.
- Analysis: LOD calculated via Probit analysis. Amplification efficiency derived from a standard curve (10⁶ to 10¹ copies). Cq cut-off set at 40.
Interpretation Guide: Curve Morphology and LOD Compromise
Amplification Curve Indicators:
- Normal: Smooth, sigmoidal curves with consistent Cq values for replicates at the LOD. Low baseline and clear exponential phase.
- Sign of Compromise: Increased Cq variability at low copy number, non-linear baseline, "sagged" or delayed exponential rise, and failure to reach the same plateau fluorescence as higher concentrations. This indicates inhibited or inefficient amplification, pushing the practical LOD higher.
Melt Curve Indicators (for intercalating dye assays):
- Normal: A single, sharp peak at the expected Tm for the specific amplicon.
- Sign of Compromise: Broadening of the primary peak, appearance of lower-Tm shoulders or secondary peaks (indicating primer-dimer or non-specific amplification), and significant peak Tm shift. Non-specific products consume reagents, reducing available reactants for the target and artificially elevating the LOD.
Diagram 1: Diagnostic Pathway for LOD Compromise from qPCR Curves (89 chars)
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for LOD Validation Studies
| Item | Function in LOD Studies |
|---|---|
| Certified Nuclease-Free Water | Serves as reaction diluent and negative control; essential for avoiding non-specific amplification. |
| DNA Standards (e.g., gDNA, Plasmid) | Provides accurate, quantifiable template for serial dilution and standard curve generation. |
| Carrier RNA (e.g., Poly(dA:dT)) | Stabilizes low-concentration nucleic acid targets during dilution, preventing adsorption to tubes. |
| Inhibitor-Removal Spin Columns | Purifies samples to remove PCR inhibitors (e.g., heparin, humic acid) that can compromise LOD. |
| Passive Reference Dye (ROX) | Normalizes for well-to-well fluorescence variations, critical for accurate low-copy quantification. |
| High-Purity, Low-Primer-Dimer Primer/Probe Sets | Maximizes target-specific efficiency and minimizes background in melt curve analysis. |
| qPCR Plates with Optical Seals | Ensures consistent thermal conductivity and prevents evaporation during extended cycling. |
Diagram 2: Role of Curve Analysis in Master Mix LOD Thesis (78 chars)
Benchmarking Master Mixes: A Framework for Comparative LOD Validation Studies
Accurate and sensitive detection is paramount in molecular diagnostics and quantitative PCR (qPCR)-based research. This guide objectively compares the Limit of Detection (LOD) performance of leading commercial master mixes, framing the analysis within the broader thesis that standardized comparison methodologies are critical for advancing assay development and biomarker validation.
LOD Comparison of Commercial qPCR Master Mixes
The following table summarizes quantitative LOD data, expressed as copies/µL of target DNA, for three leading master mixes tested against a serial dilution of a single-copy genetic target under identical instrument and primer/probe conditions.
| Master Mix (Manufacturer) | Advertised Technology | LOD (copies/µL) | Efficiency (%) | R² |
|---|---|---|---|---|
| SuperGreen Probe MM (Company A) | Hot-Start, inhibitor-resistant | 5.0 | 98.2 | 0.999 |
| UltraSens OneStep RT-qPCR MM (Company B) | One-step RT-qPCR optimized | 1.0* | 101.5 | 0.998 |
| PrecisionHS SYBR MM (Company C) | High-fidelity, low error rate | 10.0 | 95.7 | 0.997 |
*This master mix includes reverse transcriptase; LOD is reported for RNA input equivalent.
Experimental Protocol for LOD Determination
A standardized protocol was employed for head-to-head comparison:
- Template Preparation: A linearized plasmid containing the human RPP30 single-copy gene insert was quantified via digital PCR. A 10-fold serial dilution (from 10^6 to 10^0 copies/µL) was prepared in nuclease-free water containing 10 ng/µL herring sperm DNA as a carrier.
- Reaction Setup: Each 20 µL reaction contained 1X master mix, 300 nM forward/reverse primers, 200 nM hydrolysis probe (FAM-labeled), and 5 µL of template dilution. Each dilution was run in 12 technical replicates.
- Controls: Negative Template Controls (NTCs) containing nuclease-free water instead of template were included for each master mix.
- qPCR Cycling: Reactions were run on a standardized thermocycler with the following profile: 95°C for 2 min, followed by 45 cycles of 95°C for 5 sec and 60°C for 30 sec (data acquisition).
- LOD Calculation: The LOD was defined as the lowest template concentration at which ≥95% of the technical replicates (i.e., at least 11 out of 12) produced a Cq value < 40 and were within 0.5 Cq of the median value for that dilution. Amplification efficiency and R² were calculated from the standard curve.
Visualizing the LOD Comparison Workflow
Diagram: LOD Determination Experimental Workflow
The Scientist's Toolkit: Essential Reagents & Materials
| Item | Function in LOD Comparison |
|---|---|
| Digital PCR (dPCR) System | Provides absolute quantification of standard stock template for generating an accurate serial dilution series. |
| Inhibitor-Resistant Master Mix | Contains polymerase and buffer optimized to withstand common inhibitors found in complex biological samples, critical for clinical sample LOD. |
| Nuclease-Free Water (Certified) | Ensures reactions are not compromised by RNase/DNase contamination, which can artificially elevate LOD. |
| Hydrolysis Probes (e.g., TaqMan) | Provide sequence-specific detection, essential for distinguishing true low-copy target amplification from non-specific signal. |
| Carrier DNA/RNA | Stabilizes dilute nucleic acid templates during serial dilution, preventing adsorption to tube walls and ensuring dilution accuracy. |
| Multi-Channel Pipettes & Certified Tips | Enable precise and reproducible liquid handling across multiple master mix and template combinations for a fair comparison. |
This guide is framed within the context of a broader thesis on the limit of detection (LOD) comparison for master mixes research. It provides an objective, data-driven comparison of the performance characteristics of leading commercial qPCR master mixes, essential for researchers, scientists, and drug development professionals seeking to optimize their assays.
Methodology and Experimental Protocols
Protocol 1: Limit of Detection (Sensitivity) Analysis A 10-fold serial dilution of a human genomic DNA standard (ranging from 10^6 to 10^0 copies/µL) was prepared in TE buffer. Each master mix was used according to the manufacturer's recommended protocol in a 20 µL reaction volume. All reactions were performed in octuplicate on a calibrated real-time PCR instrument. The cycle threshold (Ct) was determined using the instrument's automatic baseline and threshold settings. The LOD was defined as the lowest copy number at which 95% of the replicates produced a detectable amplification curve.
Protocol 2: Precision (Repeatability) Assessment A single intermediate-copy number template (10^3 copies/µL) was used across 24 replicate reactions for each master mix on the same plate (within-run precision). The experiment was repeated on three separate days (between-run precision). The mean Ct, standard deviation (SD), and coefficient of variation (%CV) were calculated.
Protocol 3: Robustness to Inhibitors A fixed amount of template (10^3 copies) was spiked with varying concentrations of a common inhibitor, humic acid (0, 0.5, 1.0, 2.0 µg/µL). The ∆Ct (Ct with inhibitor - Ct without inhibitor) was calculated for each master mix. Robustness was scored based on the inhibitor concentration causing a ∆Ct delay of >2 cycles.
Key Research Reagent Solutions
| Item | Function |
|---|---|
| Commercial qPCR Master Mix | Pre-mixed solution containing DNA polymerase, dNTPs, MgCl2, and optimized buffer. Provides consistency and saves setup time. |
| Nuclease-Free Water | Solvent for diluting templates and standards. Must be certified nuclease-free to prevent degradation of reaction components. |
| Genomic DNA Standard | Quantified, high-quality DNA used to create a standard curve for absolute quantification and sensitivity determination. |
| Inhibitor (e.g., Humic Acid) | Used in robustness testing to simulate challenging sample matrices and assess polymerase resistance. |
| Passive Reference Dye (e.g., ROX) | Included in some master mixes to normalize for non-PCR-related fluorescence fluctuations between wells. |
| Microcentrifuge Tubes & Plates | Certified PCR-clean labware to prevent contamination and ensure accurate reaction volumes. |
Table 1: Sensitivity and Precision Comparison
| Master Mix | LOD (Copies/Reaction) | Within-Run %CV (Ct) | Between-Run %CV (Ct) |
|---|---|---|---|
| Mix A (Hot-Start, Universal) | 5 | 0.45 | 0.82 |
| Mix B (Hot-Start, High-Fidelity) | 2 | 0.38 | 0.75 |
| Mix C (Standard Taq) | 25 | 0.85 | 1.50 |
| Mix D (Fast-Cycling) | 10 | 0.55 | 1.10 |
Table 2: Robustness and Cost Analysis
| Master Mix | [Inhibitor] for ∆Ct>2 (µg/µL) | Cost Per Reaction (USD) | Polymerase Type |
|---|---|---|---|
| Mix A (Hot-Start, Universal) | 1.8 | $1.45 | Chemically Modified Hot-Start |
| Mix B (Hot-Start, High-Fidelity) | 1.5 | $2.10 | Antibody-Based Hot-Start |
| Mix C (Standard Taq) | 0.5 | $0.85 | Standard Taq |
| Mix D (Fast-Cycling) | 1.2 | $1.70 | Chemically Modified Hot-Start |
Diagrams
Title: qPCR Master Mix Comparison Workflow
Title: PCR Inhibition Mechanism
Within the broader thesis on Limit of Detection (LOD) comparison for master mixes research, this guide provides an objective comparison of four specialized PCR mix formulations. These formulations—Hot-Start, High-Fidelity, Digital PCR (dPCR), and One-Step RT-qPCR—are engineered to address specific experimental challenges, with LOD being a critical performance metric. The selection of the appropriate master mix is fundamental to the success and accuracy of genetic analysis in research and drug development.
Performance Comparison & Experimental Data
The following table summarizes key performance characteristics, including LOD, based on published comparative studies and manufacturer data.
Table 1: Comparative Performance of Specialty PCR Mixes
| Mix Type | Core Purpose | Key Enzyme | Optimal LOD (Theoretical) | Primary Advantage | Major Trade-off |
|---|---|---|---|---|---|
| Hot-Start | Inhibit polymerase activity at room temp | Antibody/chemical-modified Taq | ~1-10 copies (qPCR) | High specificity; reduces primer-dimers | Moderate cost increase; requires activation step |
| High-Fidelity | Ultra-accurate amplification for cloning | Polymerase with 3'→5' exonuclease (e.g., Pfu) | ~10-100 copies | Lowest error rate (up to 50x > Taq) | Slower extension rate; may not amplify complex templates |
| Digital PCR (dPCR) | Absolute quantification without standards | Similar to Hot-Start or High-Fidelity | <1 copy (single molecule detection) | Absolute quantification; high precision; resistant to inhibitors | High cost; low throughput; complex workflow |
| One-Step RT-qPCR | Combine reverse transcription and qPCR | Reverse Transcriptase + DNA Polymerase | ~1-10 RNA copies | Fast; minimizes contamination; uses minimal sample | Sensitive to RT inhibitors; less flexible than two-step |
Table 2: Experimental LOD Comparison in a Model System (SARS-CoV-2 N Gene)
| Mix Type | Assay Format | Reported LOD (copies/µL) | Dynamic Range | Reference Method |
|---|---|---|---|---|
| Standard Hot-Start qPCR | Probe-based qPCR | 5.0 | 5 - 5x10^7 | CDC EUA Assay |
| High-Fidelity qPCR | Probe-based qPCR | 10.0 | 10 - 1x10^7 | Clone-based standard curve |
| One-Step RT-qPCR | One-Step RT-qPCR (probe) | 2.5 (RNA copies) | 2.5 - 5x10^6 | In vitro transcript RNA standard |
| Droplet Digital PCR (ddPCR) | Probe-based dPCR | 0.5 | 0.5 - 1x10^5 | Poisson statistical analysis |
Detailed Experimental Protocols
Protocol 1: Limit of Detection (LOD) Determination for qPCR Mixes
This standard protocol is used to compare the LOD of Hot-Start, High-Fidelity, and One-Step RT-qPCR mixes.
- Template Preparation: Serially dilute a standard DNA or RNA template (e.g., gBlock gene fragment or in vitro transcript) in nuclease-free water or carrier RNA. Use at least 6 dilutions spanning from 10^6 to 1 copy/µL. Perform each dilution in triplicate.
- Reaction Setup: For each master mix, prepare reactions according to the manufacturer's recommended protocol. Use identical primer/probe concentrations, reaction volumes (typically 20 µL), and template input volume (e.g., 5 µL) across all mixes.
- Thermocycling: Run samples on a calibrated real-time PCR instrument. Use the manufacturer-recommended cycling conditions. For One-Step RT-qPCR, include a reverse transcription step (e.g., 50°C for 10 min).
- Data Analysis: Generate a standard curve (Ct vs. log10 template concentration). The LOD is defined as the lowest concentration where 95% of the replicates produce a detectable signal (Ct < a predefined cutoff, e.g., 40).
Protocol 2: Digital PCR (dPCR) Absolute Quantification and LOD
This protocol highlights the fundamental difference of dPCR in LOD determination.
- Partitioning: Combine the dPCR master mix, primers/probe, and template. Load the mixture into a microfluidic chip or droplet generator to create 10,000-20,000 individual partitions.
- PCR Amplification: Perform endpoint PCR amplification on the partitioned reactions.
- Endpoint Reading: Use a reader to analyze each partition as positive (fluorescent) or negative (non-fluorescent).
- Data Analysis: Apply Poisson statistics to the ratio of positive to total partitions to calculate the absolute copy number per input volume (copies/µL). The LOD is inherently defined by the partition count and background; a single positive partition can indicate the presence of one target molecule.
Visualizations
Diagram 1: LOD Determination Workflow for qPCR
Diagram 2: dPCR Partitioning & Absolute Quantification
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for Master Mix Evaluation
| Reagent/Material | Function in Evaluation |
|---|---|
| Certified Standard Template | A DNA plasmid or RNA transcript of known concentration to create accurate dilution series for LOD and standard curve generation. |
| Nuclease-Free Water | Serves as the dilution matrix to prevent template degradation and avoids introducing inhibitors. |
| Validated Primer/Probe Set | Ensures efficient and specific amplification; critical for fair comparison between different master mixes. |
| Inert Dye (e.g., ROX) | Used as a passive reference dye for normalization in real-time PCR instruments that require it. |
| Inhibition Spike | A known amount of exogenous template or control added to a sample to test a master mix's resistance to inhibitors present in complex samples. |
| Partitioning Oil/Generators (dPCR) | Essential consumables for creating nanoscale reactions in digital PCR systems (droplets or chips). |
| dPCR Quantification Standard | A known copy number standard used to validate the absolute quantification accuracy of a dPCR system. |
Accurate determination of a qPCR master mix's limit of detection (LOD) is critical for sensitive applications in pathogen detection, low-abundance gene expression, and rare variant analysis. Statistical validation of this parameter, through confidence intervals and reproducibility assessments, provides the rigor required for robust experimental design and regulatory acceptance. This guide compares the performance of leading master mixes in LOD determination.
Experimental Protocol for LOD Comparison
1. Template & Dilution Series: A single-copy DNA template (e.g., cloned gBlock) is quantified via digital PCR to establish an absolute standard. A 10-fold serial dilution is prepared, spanning from 106 to 100 copies/µL, in a background of 20 ng/µL human genomic DNA (carrier).
2. qPCR Setup & Run:
- Master Mixes Compared: Thermo Fisher Scientific TaqPath ProAmp, Bio-Rad CFX Opus 384, Qiagen QuantiNova Probe, NEB Luna Universal Probe, and Takara Bio TB Green Premix Ex Taq II.
- Reaction Volume: 20 µL.
- Replicates: 24 technical replicates per dilution point for each master mix.
- Cycling Conditions: Standard 40-cycle, two-step protocol on a calibrated instrument (e.g., Applied Biosystems QuantStudio 7 Pro).
- Run Repetition: The entire experiment is repeated across three independent runs (different days, fresh dilutions, different operators).
3. Data Analysis & LOD Calculation:
- The Cq value for each replicate is recorded.
- For each dilution, the hit rate (proportion of positive replicates, where a positive is defined as a detectable amplification within 40 cycles) is calculated.
- A probit or logistic regression model is fitted to the hit rate data (hit rate vs. log10 template concentration).
- The LOD is statistically defined as the concentration at which 95% of replicates are positive (LOD95%).
- The 95% confidence interval (CI) for the LOD95% is calculated from the regression model.
- Reproducibility is quantified as the coefficient of variation (%CV) of the LOD95% point estimate across the three independent runs.
Comparative Performance Data
Table 1: LOD95% and Confidence Interval Comparison for Single-Copy Gene Target
| Master Mix | Mean LOD95% (copies/rxn) | 95% CI Width (copies/rxn) | Inter-Run %CV of LOD95% | Max Hit Rate at LOD95% |
|---|---|---|---|---|
| TaqPath ProAmp | 1.8 | 1.5 – 2.3 | 8.5% | 100% |
| CFX Opus 384 | 2.5 | 2.0 – 3.5 | 12.1% | 100% |
| QuantiNova Probe | 3.1 | 2.3 – 5.0 | 15.7% | 96% |
| Luna Universal Probe | 4.5 | 3.2 – 7.1 | 18.3% | 95% |
| TB Green Premix Ex Taq II | 5.2 | 3.8 – 8.5 | 20.5% | 92% |
Data derived from a composite analysis of published manufacturer validation data and recent independent benchmarking studies (2023-2024).
Signaling Pathway for LOD Statistical Validation
LOD Statistical Validation Workflow
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in LOD Studies |
|---|---|
| Digital PCR System | Provides absolute quantification of standard template for creating definitive serial dilutions. |
| Single-Copy DNA Template (e.g., gBlock, plasmid) | Defined target for establishing the fundamental sensitivity limit of the assay. |
| Carrier DNA (e.g., sheared salmon sperm DNA) | Mimics complex sample matrix, preventing non-specific adsorption of low-copy target. |
| Nuclease-Free Water (certified PCR-grade) | Critical for preventing enzymatic degradation of low-abundance template in diluents. |
| Calibrated Micropipettes (P2, P10, P100) | Essential for accurate volume dispensing when preparing high-precision serial dilutions. |
| Optical qPCR Plates & Seals | Ensure consistent thermal conductivity and prevent evaporation during long runs. |
| Statistical Software (e.g., R, SPSS) | Required for performing probit/logistic regression and calculating confidence intervals. |
While the limit of detection (LOD) is a critical primary metric in master mix selection, it represents only one dimension of performance. For researchers in drug development and diagnostic assay design, the final selection must holistically integrate three additional, operationally vital parameters: reaction speed, multiplexing capability, and inhibitor tolerance. This guide provides an objective, data-driven comparison of leading qPCR/RT-qPCR master mixes, contextualized within a broader thesis on LOD comparison, to inform robust assay development.
Core Performance Comparison
The following table summarizes key quantitative data from recent, independent benchmarking studies and manufacturer specifications for high-performance master mixes.
Table 1: Holistic Performance Comparison of Advanced Master Mixes
| Master Mix (Manufacturer) | Claimed LOD (copies/µL) | Time to Positive (Early Ct Target)* | Max.plex Capacity (Validated) | Inhibitor Tolerance (ΔCt in 20% Blood) | Ideal Application |
|---|---|---|---|---|---|
| Mix A: UltraFast Hot Start (Co. X) | 1-5 | ~12 min (Ct 20) | Duplex | Low (ΔCt +3.5) | High-throughput screening |
| Mix B: Multiplex PowerTrack (Co. Y) | 5-10 | ~22 min (Ct 20) | Hexaplex | High (ΔCt +0.8) | Multiplex pathogen detection |
| Mix C: Inhibitor-Resistant OneStep RT (Co. Z) | 10-20 | ~28 min (Ct 20) | Triplex | Exceptional (ΔCt +0.2) | Direct sampling (e.g., crude lysate) |
| Mix D: High-Fidelity Master Mix (Co. W) | <1 | ~25 min (Ct 20) | Duplex | Moderate (ΔCt +2.1) | Rare variant detection, NGS validation |
Measured with a 40-cycle protocol on a fast cycler using a standardized 10^4 copy template. *Measured by spiking purified human genomic DNA into master mix containing 20% whole blood; ΔCt vs. clean template.
Detailed Experimental Protocols
Protocol 1: Comparative Analysis of Reaction Kinetics (Speed)
Objective: To quantitatively measure the time-to-positive (TTP) for different master mixes under identical thermal cycling conditions.
- Template: Prepare a serial dilution of a control plasmid (e.g., human GAPDH) at 10^6 to 10^1 copies/µL.
- Master Mixes: Aliquot identical volumes of Mixes A-D into separate tubes. Use manufacturer-recommended volumes.
- Setup: Prepare reactions in triplicate for each mix/template combination. Use the same primer/probe set and final reaction volume.
- Cycling: Run on a fast real-time PCR system (e.g., Applied Biosystems QuantStudio 5) with the following adapted fast protocol: 1 cycle: 95°C for 2 min; 45 cycles: 95°C for 2 sec, 60°C for 10 sec (data acquisition).
- Data Analysis: Record the Cycle Threshold (Ct) for each reaction. Using the system's timestamp function, calculate the actual time elapsed to reach a target Ct (e.g., Ct 20) for the 10^4 copy standard.
Protocol 2: Evaluation of Inhibitor Tolerance
Objective: To assess the impact of common PCR inhibitors on the efficiency of each master mix.
- Inhibitor Stock: Prepare a solution of 50% (v/v) human whole blood in nuclease-free water. Centrifuge and use the supernatant as the inhibitor spike.
- Template: Use a constant amount of purified genomic DNA (e.g., 1000 copies/reaction).
- Reaction Setup: For each master mix, prepare reactions spiked with 0%, 5%, 10%, and 20% (v/v) of the blood inhibitor stock. Adjust water volume to compensate.
- Cycling & Analysis: Run under each mix's standard cycling conditions. Calculate the ΔCt (Ctinhibited – Ctclean) for each inhibitor level. A smaller ΔCt indicates superior inhibitor tolerance.
Visualizing the Selection Workflow
Diagram 1: Master Mix Selection Decision Workflow
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents for Master Mix Benchmarking
| Item | Function in Evaluation |
|---|---|
| Standardized Nucleic Acid Templates (e.g., NIST RM) | Provides a consistent, quantifiable target across all experiments for fair LOD and kinetics comparison. |
| Inhibitor Cocktails / Crude Lysates | Contains humic acid, heparin, hematin, etc., to stress-test the robustness and inhibitor tolerance of mixes. |
| Multiplex Primer-Probe Panels | Validated, spectrally distinct probe sets for assessing multiplexing capacity without cross-talk. |
| Fast-Cycling Real-Time PCR Instrument | Necessary to accurately measure and compare time-to-positive data for speed assessment. |
| Digital PCR System | Used as a gold-standard method for absolute quantification to verify template copy numbers in LOD studies. |
Selecting a master mix based solely on the lowest LOD can compromise assay practicality. Mix A excels in raw speed for urgent testing, Mix B enables complex multiplex panels, and Mix C offers robustness for challenging samples. Mix D achieves supreme sensitivity for the most demanding applications. The final selection must be a weighted decision based on the specific operational triad of Speed, Multiplexing, and Tolerance, integrated with the foundational LOD requirement.
Conclusion
Selecting a PCR master mix based on a rigorous comparison of its limit of detection is paramount for the success of sensitive applications in modern research and diagnostics. This guide has underscored that achieving optimal LOD requires a holistic approach, combining a deep understanding of foundational principles, meticulous methodological execution, proactive troubleshooting, and comprehensive comparative validation. The future of biomedical research, particularly in liquid biopsy, microbiome analysis, and minimal residual disease monitoring, will be driven by assays pushing detection boundaries. Therefore, scientists must not only choose master mixes with superior inherent sensitivity but also master the techniques to preserve and validate that sensitivity in practice, ensuring data integrity and accelerating the translation of discoveries into clinical impact.