Mastering Monte Carlo Simulation for Light Transport: A Complete Guide for Biomedical Researchers
This article provides a comprehensive guide to Monte Carlo (MC) simulation for modeling light propagation in scattering and absorbing media, a critical tool in biomedical optics.
Mastering Monte Carlo Simulation for Light Transport: A Complete Guide for Biomedical Researchers
Abstract
This article provides a comprehensive guide to Monte Carlo (MC) simulation for modeling light propagation in scattering and absorbing media, a critical tool in biomedical optics. It begins by establishing the fundamental physics of light-tissue interaction, including the radiative transport equation and key optical properties. The core section details the step-by-step methodology for building and executing MC simulations, highlighting practical implementation and applications in areas like diffuse optical spectroscopy and photodynamic therapy planning. We address common computational challenges and optimization techniques for improving accuracy and efficiency. Finally, the article covers validation strategies against analytical models and experimental data, and compares MC methods with alternative models like diffusion theory. This guide is tailored for researchers, scientists, and drug development professionals seeking to leverage MC simulations for advanced diagnostic and therapeutic applications.
Light in Tissue 101: Core Physics and Principles for Monte Carlo Simulation
Light interaction with biological tissues—comprising absorption, scattering, and fluorescence—is central to a multitude of diagnostic and therapeutic modalities. Analytical solutions to the Radiative Transport Equation (RTE), which governs light propagation, are intractable for complex, heterogeneous media. Monte Carlo (MC) simulation provides a stochastic, yet rigorous, numerical approach to solve the RTE, making it the gold standard for modeling light distributions in biological systems. Within the broader thesis on MC methods for light propagation, this article establishes the fundamental necessity of simulation as a precursor to, and enhancer of, experimental research in biomedicine.
The Core Challenge: Light-Tissue Interaction
Biological media are turbid. Photons do not travel in straight lines; their paths are altered by scattering (primarily from cellular structures and organelles) and absorption (by chromophores like hemoglobin, melanin, and water). The quantitative characterization of these events is described by the following optical properties:
- Scattering Coefficient (µs): The probability of scattering per unit path length.
- Absorption Coefficient (µa): The probability of absorption per unit path length.
- Anisotropy Factor (g): The average cosine of the scattering angle, describing forward-directedness.
- Reduced Scattering Coefficient (µs'): The effective isotropic scattering coefficient, given by µs' = µs(1-g).
Table 1: Typical Optical Properties of Biological Tissues at 630 nm
| Tissue Type | Absorption Coefficient, µa (cm⁻¹) | Scattering Coefficient, µs (cm⁻¹) | Anisotropy, g | Reduced Scattering Coefficient, µs' (cm⁻¹) |
|---|---|---|---|---|
| Skin (Epidermis) | 1.5 - 3.5 | 400 - 500 | 0.85 - 0.95 | 20 - 75 |
| Brain (Gray Matter) | 0.2 - 0.4 | 300 - 400 | 0.85 - 0.95 | 15 - 60 |
| Breast Tissue | 0.03 - 0.1 | 200 - 300 | 0.85 - 0.97 | 6 - 45 |
| Liver | 0.4 - 0.8 | 350 - 450 | 0.9 - 0.97 | 10.5 - 45 |
Why Simulation is Indispensable: Key Applications
Predictive Dosimetry for Photodynamic Therapy (PDT)
MC simulation predicts the spatiotemporal distribution of light fluence rate (J/cm²/s) within a target tissue, which is critical for calculating the photodynamic dose (e.g., light fluence * photosensitizer concentration). Accurate prediction ensures therapeutic efficacy while minimizing damage to healthy tissue.
Protocol: MC Simulation for PDT Planning
- Geometry Definition: Construct a 3D mesh of the target anatomy from patient CT/MRI scans.
- Property Assignment: Assign wavelength-specific optical properties (µa, µs, g) to each tissue layer from pre-existing libraries or inverse modeling.
- Source Modeling: Define the light source parameters (wavelength, beam profile, power, orientation).
- Photon Launch: Launch millions of photon packets, each with an initial weight.
- Path Tracking: For each photon: a. Calculate free path length via random sampling from µt (µt = µa + µs). b. If the path intersects a boundary, handle reflection/refraction via Fresnel equations. c. At the interaction point, decrement photon weight by absorption (∆W = W * (µa/µt)). d. Scatter the photon into a new direction determined by the Henyey-Greenstein phase function and a random scattering angle. e. Repeat until photon weight falls below a threshold (roulette) or it exits the geometry.
- Data Collection: Accumulate absorbed energy in voxels to generate a 3D fluence rate map.
- Dose Calculation: Integrate fluence rate over time and combine with modeled drug distribution to calculate the photodynamic dose map.
Optimization of Optical Imaging Techniques
In techniques like Diffuse Optical Tomography (DOT) and Optical Coherence Tomography (OCT), MC simulations are used to model photon migration, generate training data for image reconstruction algorithms, and validate the sensitivity of measurements to internal abnormalities.
Design and Validation of Biomedical Sensors
Simulations enable the virtual prototyping of pulse oximeters, continuous glucose monitors, and other photonic devices by modeling light-tissue interaction for specific sensor geometries and tissue interfaces.
The Monte Carlo Simulation Workflow
Title: Monte Carlo Photon Propagation Algorithm
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Components for MC Simulation & Experimental Validation
| Item | Function in Research |
|---|---|
| MCML / tMCimg / GPU-MCML Codes | Validated, open-source CPU/GPU-accelerated MC simulation software for layered and voxelated media. |
| Tissue-Simulating Phantoms | Solid or liquid calibrators with precisely known optical properties (µa, µs', n) for experimental validation of simulations. |
| Integrating Sphere Spectrometer | Gold-standard instrument for experimental measurement of bulk optical properties (reflectance & transmittance) of thin samples. |
| Spatially Resolved Diffuse Reflectance Probe | Fiber-optic probe to measure radial reflectance profiles for inverse extraction of µa and µs' from tissue. |
| Index-Matching Fluids | Liquids (e.g., glycerol-water mixtures) used to reduce surface specular reflection during optical measurements. |
| Finite Element Analysis (FEA) Software | Used to create anatomically accurate 3D mesh geometries from medical imaging data for import into voxel-based MC codes. |
Simulating light propagation in biological media via Monte Carlo methods is not merely a computational exercise; it is a foundational pillar of modern biophotonics research and translation. It provides unparalleled insight into photon fate, enables precise dosimetry for light-based therapies, and accelerates the development of optical diagnostic technologies. As a core component of the broader thesis, this approach establishes a rigorous, predictive framework that bridges theoretical optics and clinical application, ultimately guiding more effective and personalized therapeutic interventions.
The Radiative Transport Equation (RTE) is the fundamental integro-differential equation governing the flow of electromagnetic radiation (e.g., light) through a medium that absorbs, scatters, and emits photons. Within the context of Monte Carlo simulation for light propagation in scattering and absorbing media, the RTE provides the rigorous physical theory that the stochastic methods aim to solve numerically. This theory is essential for modeling applications in biomedical optics, such as tissue spectroscopy, optical imaging, and photodynamic therapy for drug development.
Mathematical Formulation
The steady-state RTE for light transport in a non-emitting medium is given by: [ \hat{s} \cdot \nabla L(\mathbf{r}, \hat{s}) = -\mut L(\mathbf{r}, \hat{s}) + \mus \int_{4\pi} p(\hat{s}', \hat{s}) L(\mathbf{r}, \hat{s}') d\omega' + Q(\mathbf{r}, \hat{s}) ] Where:
- ( L(\mathbf{r}, \hat{s}) ) is the radiance ((W \cdot cm^{-2} \cdot sr^{-1})) at position (\mathbf{r}) in direction (\hat{s}).
- ( \mut = \mua + \mu_s ) is the total attenuation coefficient ((cm^{-1})).
- ( \mu_a ) is the absorption coefficient ((cm^{-1})).
- ( \mu_s ) is the scattering coefficient ((cm^{-1})).
- ( p(\hat{s}', \hat{s}) ) is the scattering phase function, describing the probability of scattering from direction (\hat{s}') to (\hat{s}).
- ( Q(\mathbf{r}, \hat{s}) ) is the internal light source.
The Henyey-Greenstein phase function is commonly used to model anisotropic scattering in biological tissue: [ p(\cos\theta) = \frac{1}{4\pi} \frac{1 - g^2}{(1 + g^2 - 2g \cos\theta)^{3/2}} ] where (g) is the anisotropy factor, ranging from -1 (perfect backscattering) to 1 (perfect forward scattering).
Key Optical Properties & Quantitative Data
The following table summarizes the typical ranges of optical properties for biological tissues at common laser wavelengths (e.g., 630-850 nm), crucial for researchers in drug development planning light-based therapies or diagnostics.
Table 1: Typical Optical Properties of Biological Tissues
| Tissue Type | Absorption Coefficient, (\mu_a) (cm(^{-1})) | Scattering Coefficient, (\mu_s) (cm(^{-1})) | Anisotropy Factor, (g) | Reduced Scattering Coefficient, (\mu_s') (cm(^{-1}))* |
|---|---|---|---|---|
| Human Skin (dermis) | 0.2 - 1.5 | 150 - 250 | 0.8 - 0.95 | 15 - 50 |
| Human Breast Tissue | 0.03 - 0.08 | 80 - 150 | 0.85 - 0.98 | 4 - 20 |
| Human Brain (gray matter) | 0.2 - 0.4 | 50 - 100 | 0.85 - 0.95 | 5 - 15 |
| Human Skull Bone | 0.1 - 0.3 | 150 - 250 | 0.85 - 0.95 | 20 - 40 |
| Rodent Liver | 0.4 - 1.0 | 100 - 200 | 0.85 - 0.95 | 10 - 30 |
| In vitro phantom (1% Intralipid) | ~0.001 - 0.02 | 50 - 150 | 0.5 - 0.7 | 20 - 70 |
Note: (\mu_s' = \mu_s (1 - g)), a critical parameter for diffusion theory approximations.
The Monte Carlo Connection
The RTE lacks general analytical solutions for complex geometries and heterogeneous media. Monte Carlo (MC) methods provide a statistical numerical approach to solve the RTE by simulating the random walks of millions of individual photons. The core physical events in an MC simulation—absorption, scattering, and boundary interactions—are direct stochastic realizations of the terms in the RTE.
Diagram Title: Monte Carlo Algorithm for Solving the RTE
Experimental Protocols for Validation
Validating MC codes against controlled experiments is critical. A standard protocol for measuring tissue-mimicking phantom optical properties is outlined below.
Protocol 1: Inverse Adding-Doubling (IAD) for Phantom Property Measurement
- Objective: To determine the absorption (μa) and reduced scattering (μs') coefficients of a tissue-simulating phantom, providing ground truth for MC model validation.
- Materials: See "The Scientist's Toolkit" below.
- Method:
- Phantom Preparation: Prepare a solid or liquid phantom using Intralipid (scatterer) and India ink or a dye (absorber) in a buffer solution. For solid phantoms, use agarose or polyurethane as a matrix.
- Sample Holder Preparation: Place the phantom in a cuvette or mold with known, precise thickness (typically 1-10 mm).
- Collimated Transmission (Tc) Measurement:
- Use a narrow, collimated laser beam at the target wavelength.
- Direct the beam onto the sample. Place a detector directly in line behind the sample at a sufficient distance to collect only non-scattered, collimated light.
- Measure the intensity of transmitted light, (It).
- Measure the intensity without the sample, (I0).
- Calculate (Tc = It / I0).
- Total Transmission (Tt) & Reflectance (Rd) Measurement:
- Use an integrating sphere coupled to a spectrometer or photodetector.
- For (Tt), place the sample at the sphere's entrance port. All light transmitted through the sample is collected by the sphere.
- For diffuse reflectance (Rd), place the sample at a sample port on the sphere, illuminating from outside. The sphere collects all back-scattered light.
- Calibrate using standard reflectance plates and blanks.
- Inverse Algorithm: Input the measured (Tc), (Tt), and (Rd) values, along with sample thickness and known refractive index, into an IAD software algorithm. The algorithm iteratively solves the RTE to output the intrinsic optical properties μa and μs'.
- Validation: Use the derived μa and μs' as inputs to an MC simulation of the experimental geometry. Compare the simulation's predicted (Tt) and (Rd) to the measured values. Agreement within 2-5% validates the MC code.
The Scientist's Toolkit
Table 2: Essential Research Reagents & Materials for RTE/MC Studies
| Item | Function & Relevance to RTE/MC |
|---|---|
| Tissue-Mimicking Phantoms (Intralipid, TiO2, India Ink, Nigrosin, Agarose) | Provide standardized media with precisely tunable μa and μs' for experimental validation of RTE solutions and MC codes. |
| Integrating Spheres (Labsphere, Ocean Insight) | Essential instruments for measuring total diffuse reflectance and transmittance from samples, the primary data for extracting optical properties. |
| Near-Infrared (NIR) Spectrometers & Lasers (e.g., Ti:Sapphire, diode lasers) | Light sources for probing the "therapeutic window" (650-1350 nm) where tissue absorption is relatively low, making the RTE's scattering term dominant. |
| Time-Correlated Single Photon Counting (TCSPC) Systems | Enables measurement of temporal point spread functions (TPSF), providing rich data for validating time-resolved MC simulations of the time-dependent RTE. |
| High-Performance Computing (HPC) Cluster/GPU | Accelerates Monte Carlo simulations, allowing for modeling of complex 3D geometries with high spatial resolution and millions of photons in feasible timeframes. |
| Validated MC Software (e.g., MCML, TIM-OS, GPU-accelerated codes) | Pre-validated simulation tools allow researchers to implement RTE-based models without building a code from scratch, focusing on application. |
Advanced Considerations & The Diffusion Approximation
When scattering dominates absorption ((\mus' \gg \mua)) and distances from sources are large, the RTE can be simplified to the Diffusion Equation (DE): [ \nabla \cdot (D \nabla \phi(\mathbf{r})) - \mua \phi(\mathbf{r}) = -S(\mathbf{r}) ] where (D = 1/(3\mus')) is the diffusion coefficient and (\phi(\mathbf{r})) is the fluence rate. The relationship between the RTE and its approximations is hierarchical.
Diagram Title: Relationship Between Light Transport Models
The Radiative Transport Equation remains the cornerstone theory for quantitative analysis of light propagation in scattering media. Its complexity necessitates numerical solutions like Monte Carlo simulation, which has become an indispensable tool in preclinical and clinical research. For drug development professionals, particularly in photodynamic therapy or optical imaging, understanding the RTE and its stochastic solvers is crucial for designing effective light-based treatments, interpreting diagnostic signals, and translating laboratory findings into clinical applications. The ongoing integration of GPU acceleration and machine learning with MC methods promises to further enhance the fidelity and speed of RTE-based modeling for complex biological systems.
This technical guide details the core optical properties governing light propagation in turbid media, framed within the context of Monte Carlo simulation research. Accurate modeling of these properties is paramount for advancing applications in biomedical optics, drug development, and diagnostic imaging.
Monte Carlo (MC) simulation is the gold-standard numerical method for modeling stochastic photon transport in scattering and absorbing media. Its predictive power is wholly dependent on the precise input of four fundamental optical properties: absorption coefficient, scattering coefficient, anisotropy factor, and refractive index. This whitepaper provides an in-depth analysis of these properties, their measurement, and their role in MC-based research.
Core Optical Properties: Definition & Impact on MC Simulation
Absorption (μₐ)
The absorption coefficient (μₐ, units: mm⁻¹) defines the probability of photon absorption per unit path length. In MC simulations, it determines the likelihood of photon termination at each step, directly influencing the computation of deposited energy (e.g., for photothermal therapy) or fluorescence yield.
Scattering (μₛ)
The scattering coefficient (μₛ, units: mm⁻¹) defines the probability of a photon scattering event per unit path length. It is the primary determinant of how light spreads and penetrates in a medium. A high μₛ leads to increased simulation complexity and computational time due to more scattering events.
Anisotropy (g)
The anisotropy factor (g, unitless, range: -1 to 1) characterizes the directionality of scattering. Isotropic scattering has g=0; forward-scattering media (typical for biological tissues) have g → 1. In MC, g dictates the angular deflection during a scattering event via the Henyey-Greenstein or Mie phase functions.
Refractive Index (n)
The refractive index (n, unitless) governs the speed of light in a medium and the behavior at boundaries via Snell's Law and Fresnel equations. In MC, mismatches in n between layers cause reflections and refractions, critically affecting light delivery at interfaces (e.g., skin-air, tissue-implant).
Quantitative Data for Representative Media
Table 1: Optical Properties of Biological and Synthetic Media at 630 nm
| Media Type | μₐ (mm⁻¹) | μₛ (mm⁻¹) | g | n | Notes |
|---|---|---|---|---|---|
| Human Skin (Epidermis) | 0.1 - 0.5 | 20 - 40 | 0.70 - 0.90 | 1.37 - 1.45 | Strongly wavelength-dependent |
| Brain (Gray Matter) | 0.02 - 0.06 | 15 - 30 | 0.85 - 0.95 | ~1.36 | From recent J. Biomed. Opt. (2023) |
| Intralipid 20% (phantom) | ~0.001 | 40 - 50 (dilutable) | ~0.70 | 1.33 | Common calibration/phantom standard |
| Polystyrene Microspheres | Negligible | Tunable via concentration | 0.80 - 0.95 | ~1.59 | Monodisperse for validation studies |
Table 2: Summary of Key Measurement Techniques
| Technique | Property Measured | Principle | Best For |
|---|---|---|---|
| Integrating Sphere + IS | μₐ, μₛ | Measures total transmission & reflection | Homogeneous samples, in vitro |
| Time/Domain FD-NIRS | μₐ, μₛ', (μₛ = μₛ'/(1-g)) | Temporal point spread function analysis | In vivo, deep tissue |
| Ellipsometry | n, thin film properties | Polarized light reflection at an angle | Surface layers, coatings |
| Goniometry | g, Phase Function | Angular scattering intensity | Characterizing scatterers |
Experimental Protocols for Property Determination
Protocol: Double-Integrating Sphere Measurement for μₐ and μₛ
Objective: To determine the absorption and reduced scattering coefficients (μₛ' = μₛ(1-g)) of a homogeneous slab sample. Materials: See "The Scientist's Toolkit" below. Procedure:
- Sample Preparation: Prepare a slab of known, uniform thickness (d, typically 1-2 mm). Ensure parallel faces.
- System Calibration: Perform baseline measurements with the sample port empty (Rₑ, Tₑ) and with a known reflectance standard (e.g., Spectralon).
- Sample Measurement: Place the sample against the sample port.
- Measure total reflectance (Rₜ) using the reflection sphere.
- Measure total transmittance (Tₜ) using the transmission sphere.
- Measure collimated transmittance (T꜀) using a detector in a direct, narrow beam path.
- Data Analysis: Use an inverse adding-doubling (IAD) algorithm. Inputs: Rₜ, Tₜ, T꜀, d, n(sample), n(medium). The IAD algorithm iteratively solves the radiative transport equation to output μₐ and μₛ'.
Protocol: Time-Resolved Diffuse Reflectance forIn Vivoμₐ and μₛ'
Objective: To non-invasively determine tissue optical properties. Materials: Pulsed laser (e.g., Ti:Sapphire), picosecond detector (SPAD or streak camera), fiber optics, time-correlated single photon counting (TCSPC) module. Procedure:
- Setup: A pulsed laser source (<10 ps pulse) is coupled to a delivery fiber. A collection fiber at a fixed distance (ρ = 3-10 mm) guides light to the detector.
- Measurement: Record the temporal diffusion response, the Temporal Point Spread Function (TPSF), at the detector.
- Analysis: Fit the measured TPSF to the solution of the time-dependent diffusion equation for a semi-infinite medium. The early photon decay is sensitive to μₐ, while the late photon spread is sensitive to μₛ'. Non-linear fitting retrieves the properties.
Integration into Monte Carlo Simulation Workflow
Diagram Title: Monte Carlo Photon Transport Algorithm Workflow
Diagram Title: Iterative Cycle of MC Simulation & Experimental Validation
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Optical Property Research
| Item | Function / Application | Example Product / Note |
|---|---|---|
| Tissue Phantoms | Calibrating instruments & validating MC codes. Mimic tissue μₐ, μₛ, g. | Intralipid (scatterer), India Ink (absorber), Agarose matrix. |
| Integrating Spheres | Gold-standard for in vitro measurement of total reflectance/transmittance. | Labsphere, SphereOptics; various port sizes. |
| Optical Fiber Probes | Delivery and collection of light for in vivo or confined space measurements. | Multimode fibers, customized source-detector separations. |
| Time-Correlated Single Photon Counting (TCSPC) System | Essential for time-domain measurements to extract μₐ and μₛ' in vivo. | PicoQuant, Becker & Hickl systems. |
| Refractive Index Matching Fluids | Minimize surface reflections at interfaces for accurate measurements. | Cargille Labs oils, precise n from 1.30 to 1.80. |
| Spectralon Diffuse Reflectance Standards | Calibrating reflectance measurements; >99% reflecting, Lambertian surface. | Labsphere; various sizes and reflectance values. |
| Monodisperse Microsphere Suspensions | Precise scatterers with calculable (Mie theory) μₛ and g for validation. | Polystyrene or silica spheres (e.g., from ThermoFisher). |
In the research of light propagation in scattering and absorbing media, a central problem is solving the Radiative Transfer Equation (RTE). This integro-differential equation describes the conservation of energy as photons travel through a turbid medium like biological tissue. While analytical solutions exist for highly simplified cases, they are overwhelmingly insufficient for realistic scenarios encountered in biomedical optics, such as modeling light dose for photodynamic therapy or interpreting data from diffuse optical tomography. This in-depth guide examines the fundamental mathematical and physical challenges that render analytical approaches intractable, thereby necessitating the adoption of sophisticated numerical methods, with a specific focus on the context of Monte Carlo simulation for drug development and therapeutic research.
The Intractability of the Radiative Transfer Equation
The steady-state RTE in its canonical form is: [ \hat{s} \cdot \nabla L(\vec{r}, \hat{s}) = -\mut L(\vec{r}, \hat{s}) + \mus \int{4\pi} L(\vec{r}, \hat{s}') f(\hat{s}', \hat{s}) d\Omega' + Q(\vec{r}, \hat{s}) ] Where (L) is the radiance, (\mut) is the total attenuation coefficient, (\mu_s) is the scattering coefficient, (f) is the scattering phase function, and (Q) is the source term.
The challenges preventing analytical solutions are summarized below:
| Challenge Category | Specific Limitation | Consequence for Analytical Solving |
|---|---|---|
| Geometric Complexity | Arbitrary, multi-layered, and irregular boundary shapes (e.g., human organs). | Boundary conditions cannot be expressed in a separable coordinate system. |
| Heterogeneity | Spatially varying optical properties ((\mua(\vec{r}), \mus(\vec{r}))). | Coefficients are not constant, breaking standard solution methods like separation of variables. |
| Anisotropic Scattering | Complex phase functions (e.g., Henyey-Greenstein with high anisotropy factor (g)). | The integral term becomes intractable, requiring series expansion and truncation approximations. |
| Source Specificity | Complex, distributed, or pulsed source configurations. | Lack of a Green's function for arbitrary sources. |
| Dimensionality | The equation is defined in 3D spatial and 2D angular domains (5D total). | "Curse of dimensionality"; analytic solutions only exist for 1D slab geometry with severe simplifications. |
Numerical Methods as a Solution
Numerical methods approximate solutions by discretizing the problem domain. The following table compares common approaches used in light propagation modeling:
| Numerical Method | Core Principle | Advantages | Disadvantages | Typical Use Case in Biomedicine |
|---|---|---|---|---|
| Monte Carlo (MC) | Statistical simulation of photon packet trajectories using random sampling. | Handles any geometry/heterogeneity; considered the "gold standard" for accuracy. | Computationally expensive; results are stochastic. | Benchmarking; simulating complex in vivo scenarios. |
| Finite Element Method (FEM) | Spatial discretization of a diffusion approximation PDE to the RTE. | Efficient for diffuse light in highly scattering media. | Relies on diffusion approximation, invalid near sources and boundaries. | Diffuse Optical Tomography image reconstruction. |
| Discrete Ordinates (S_N) | Discretization of the angular domain into discrete directions. | More direct solution of RTE than diffusion. | Can suffer from "ray effects"; angular discretization errors. | Radiation transport in tissue with low scattering regions. |
| Finite-Difference Time-Domain (FDTD) | Direct discretization of Maxwell's equations on a grid. | Solves electromagnetic wave physics exactly. | Prohibitively expensive for macroscopic tissues. | Modeling light interaction at the cellular level. |
Monte Carlo: The Method of Choice for Complex Scenarios
For the thesis research on light propagation in scattering media, Monte Carlo methods are paramount. The core algorithm is based on stochastic sampling of probability distributions governing photon interaction.
Key Experimental Protocol: Validating a Monte Carlo Model
To establish confidence in a custom Monte Carlo code, a standard validation protocol against an analytic solution is employed.
Protocol Title: MC Validation in a Homogeneous Infinite Slab.
Objective: To verify that the statistical simulation matches the known analytic solution for energy deposition in a simple geometry.
Materials & Software: See "The Scientist's Toolkit" below. Procedure:
- Define Benchmark: Use the time-resolved reflectance (R(\rho, t)) from an isotropic point source in an infinite homogeneous medium, calculated via the diffusion equation with an extrapolated-boundary condition.
- Configure MC Simulation:
- Set optical properties: (\mua = 0.1 \text{ cm}^{-1}), (\mus = 10.0 \text{ cm}^{-1}), (g = 0.9), refractive index (n = 1.37).
- Launch (10^7) to (10^8) photon packets.
- Record the time-of-flight and exit position of each reflected photon.
- Data Collection: Bin reflected photons into time gates (e.g., 0-5 ps, 5-10 ps) and radial distances ((\rho)) from the source.
- Comparison: Calculate the normalized root-mean-square error (NRMSE) between the MC-derived (R{MC}(\rho, t)) and the analytic (RA(\rho, t)) over all bins.
- Success Criterion: NRMSE < 2% across the clinically relevant time and radial ranges.
Diagram Title: Monte Carlo Model Validation Workflow
Core Monte Carlo Photon Propagation Algorithm
The logical flow of a standard Monte Carlo simulation for light propagation is described below.
Diagram Title: Monte Carlo Photon Propagation Logic
The Scientist's Toolkit: Key Research Reagent Solutions
Essential materials and computational tools for conducting numerical light propagation research.
| Item Name | Function & Purpose in Research | Example/Specification |
|---|---|---|
| Tissue-Simulating Phantoms | Provide ground-truth optical properties for validating numerical models and instrumentation. | Solid polyurethane phantoms with embedded TiO2 (scatterer) and ink (absorber) with known µa, µs', n. |
| Optical Property Datasets | Serve as input parameters (µa, µs, g) for simulations of real tissues. | Published tabulated values for human skin, brain, breast, etc., across relevant wavelengths. |
| Validated MC Code Base | Core engine for simulating photon transport. | Open-source packages like "MCX" (GPU-accelerated) or "tMCimg" (standard CPU). |
| High-Performance Computing (HPC) Cluster | Enables running large-scale simulations (>>10^9 photons) in feasible time. | Access to GPU nodes (NVIDIA V100/A100) for parallelized Monte Carlo execution. |
| Data Analysis Pipeline | Processes raw photon history data into measurable quantities (fluence, reflectance). | Custom Python/Matlab scripts for spatial/temporal binning, normalization, and visualization. |
| Sensitivity Analysis Framework | Quantifies how uncertainties in input optical properties affect simulation output. | Software implementing Morris or Sobol methods for global variance-based sensitivity analysis. |
This technical whitepaper, framed within a broader thesis on Monte Carlo simulation for light propagation, details the statistical methodology underpinning Monte Carlo modeling of photon migration in scattering and absorbing media. It serves as an in-depth guide for researchers and drug development professionals applying these techniques to biomedical optics, such as diffuse optical tomography, photodynamic therapy planning, and tissue spectroscopy.
Light propagation in biological tissue is dominated by scattering and absorption events. The radiative transport equation (RTE) describes this process precisely but is often intractable for complex geometries. The Monte Carlo method provides a stochastic numerical solution by simulating the random walks of individual photon packets through the medium, governed by probability distributions derived from the medium's optical properties.
Core Algorithm & Statistical Foundations
The Monte Carlo method simulates the fate of millions of photon packets. Each packet is assigned an initial weight and tracked until it is absorbed, escapes, or its weight falls below a threshold. Key stochastic decisions are made using pseudo-random numbers.
Key Probability Distributions:
- Path Length (ℓ) between interactions: P(ℓ) = μₜ exp(-μₜℓ), where μₜ = μₛ + μₐ (total attenuation coefficient).
- Scattering Angle (θ): Governed by the scattering phase function, commonly the Henyey-Greenstein function: p(cos θ) = (1 - g²) / (2(1+g² - 2g cos θ)^(3/2)).
- Azimuthal Angle (ψ): Uniformly distributed: P(ψ) = 1/(2π).
Detailed Experimental Protocol for a Monte Carlo Simulation
Below is a standardized protocol for simulating photon migration in a homogeneous semi-infinite medium.
Protocol 1: Basic Monte Carlo for Homogeneous Media
Objective: To compute the spatial distribution of absorbed energy and diffuse reflectance. Input Parameters: See Table 1. Procedure:
- Initialization: Set photon packet weight (W) = 1. Launch from source position with initial direction.
- Step Selection: Generate a random number (ξ). Compute step size: ℓ = -ln(ξ) / μₜ.
- Move Photon: Update coordinates: x = x + ℓ·μₓ, (similarly for y, z), where μₓ is the direction cosine.
- Absorption & Weight Update: At the interaction site, decrement weight: ΔW = W·(μₐ/μₜ). Deposit ΔW into local absorption array. Update: W = W - ΔW.
- Scattering: Sample new direction (θ, ψ) using the Henyey-Greenstein phase function and another random number. Update photon direction cosines.
- Boundary Handling (Air-Tissue Interface): a. Compute distance to boundary. b. If step crosses boundary, calculate partial step to boundary. Move photon to boundary. c. Compute probability of internal reflection using Fresnel's equations. Use a random number to decide if photon reflects internally or escapes (is recorded as reflectance or transmittance).
- Photon Termination: Apply Russian Roulette if W < threshold (e.g., 0.0001). If the photon survives, its weight is increased; otherwise, it is terminated.
- Loop: Repeat steps 2-7 until the photon escapes or is terminated.
- Ensemble Average: Repeat the entire process for 1e6 to 1e8 photon packets to build statistically meaningful results.
Table 1: Standard Optical Properties for Common Biological Tissues
| Tissue Type | Absorption Coefficient (μₐ) [mm⁻¹] | Scattering Coefficient (μₛ) [mm⁻¹] | Anisotropy (g) | Reduced Scattering Coefficient (μₛ' = μₛ(1-g)) [mm⁻¹] | Source |
|---|---|---|---|---|---|
| Human Skin (630 nm) | 0.03 - 0.10 | 15 - 25 | 0.80 - 0.90 | 2.0 - 4.0 | [Recent review, 2023] |
| Human Brain (Gray Matter, 800 nm) | 0.02 - 0.04 | 18 - 22 | 0.85 - 0.95 | 0.9 - 3.3 | [Biomed. Opt. Express, 2024] |
| Breast Tissue (NIR Window) | 0.003 - 0.008 | 8 - 12 | 0.90 - 0.97 | 0.2 - 1.2 | [J. Biomed. Opt., 2023] |
| Intralipid 20% (Phantom, 550 nm) | ~0.001 | 60 - 80 | 0.6 - 0.7 | 18 - 32 | [Calibration studies] |
Table 2: Performance Metrics of Selected Monte Carlo Software Platforms
| Software / Code | Key Feature | Language | Acceleration Method | Typical Runtime (10⁷ photons) | Reference |
|---|---|---|---|---|---|
| MCX / MMC | GPU-accelerated, voxelated media | C/CUDA | GPU Parallelization | < 60 sec | [Fang, 2024 update] |
| tMCimg | Time-resolved, standard model | C | CPU Multi-threading | ~10 min | [Boas et al.) |
| Monte Carlo eXtreme (MCX) | Cloud-based, open-source | Web/GPU | GPU Clusters | Variable | [mcx.space, 2024] |
| TIM-OS | MatLab-based, educational | MATLAB | CPU | Hours | [Prahl et al.) |
Visualization of Core Concepts
Photon Packet Lifecycle Workflow
Position of Monte Carlo in Solving the RTE
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials & Digital Tools for Monte Carlo Photon Migration Research
| Item / Solution | Function / Purpose | Example / Specification |
|---|---|---|
| Tissue-Simulating Phantoms | Provide calibrated, stable media with known optical properties to validate MC simulations. | Solid silicone phantoms with India Ink (absorber) & TiO₂/Ti₂O₃ (scatterer); Liquid Intralipid & Ink solutions. |
| Optical Property Databases | Source input parameters (μₐ, μₓ, g) for simulations of specific tissues at target wavelengths. | OPE (Online Photon-tissue Interaction Database), IMSP (Internet-based Media for Simulation in Photonics). |
| GPU-Accelerated Computing Hardware | Drastically reduces computation time (from hours to seconds) for large-scale (10⁸+ photon) simulations. | NVIDIA Tesla/RTX series GPUs with high CUDA core count and VRAM. |
| Validated Monte Carlo Code Base | Foundation for custom modification, ensuring correct implementation of core physics. | MCX (GPU), MCML (classic CPU), or TIM-OS (educational MATLAB). |
| Spectral Calibration Tools | To measure exact optical properties of phantoms or ex vivo tissue for model inputs. | Integrating sphere systems coupled with inverse adding-doubling (IAD) analysis software. |
| High-Performance Random Number Generator | Ensures statistical robustness and avoids correlation artifacts in stochastic sampling. | Mersenne Twister (MT19937) or cryptographic generators for parallel seeds. |
| Data Analysis & Visualization Suite | Processes raw output (absorption maps, pathlengths, fluence) into interpretable results. | Python (NumPy, SciPy, Matplotlib), MATLAB, or Paraview for 3D volumetric data. |
Building Your Simulator: A Step-by-Step Guide to Monte Carlo Implementation
This document constitutes a chapter of a comprehensive thesis on Monte Carlo (MC) simulation for light propagation in scattering and absorbing media. Within this research framework, the Monte Carlo method for Multi-Layered (MCML) tissues is established as the gold-standard, in-silico reference model. Its role is pivotal for validating faster, approximate models, designing optical diagnostic devices, and interpreting experimental data from techniques like diffuse reflectance spectroscopy in pharmaceutical development and tissue optics research.
Algorithmic Core Logic & Structure
The MCML algorithm models light transport as a stochastic random walk of discrete photon packets (or "pseudophotons") through a planar, multi-layered geometry. Each layer is defined by its optical properties: absorption coefficient (μₐ), scattering coefficient (μₛ), anisotropy factor (g), refractive index (n), and thickness.
The core logic is an iterative loop over N photon packets, with each packet's journey governed by the following probabilistic steps:
- Initialization: Launch a photon packet with initial weight
W= 1 at the origin, directed perpendicularly into the first layer. - Step Size Selection: Compute a free path length,
s, using the total interaction coefficient (μₜ = μₐ + μₛ) and a random number, ξ₁:s = -ln(ξ₁) / μₜ - Photon Movement & Boundary Handling: Move the photon by distance
s. If a layer boundary is intersected, move the photon to the boundary, compute the probability of internal reflection using Fresnel's formulas, and either reflect or transmit the packet based on a random number. - Interaction & Weight Deposition: At the new location, the photon packet deposits a fraction of its weight into the local absorption tally (e.g., a 2D radial- depth,
A[r, z]). The deposited weightΔWis:ΔW = W * (μₐ / μₜ)The packet's weight is then updated:W = W - ΔW. - Scattering: The packet's direction is changed by sampling a new deflection angle (θ) from the Henyey-Greenstein phase function and a new azimuthal angle (φ) uniformly. This defines a new propagation direction.
- Roulette & Termination: If the packet weight
Wfalls below a pre-defined threshold (e.g., 10⁻⁴), it is terminated via a "roulette" technique: with a small probability (e.g., 0.1), its weight is increased; otherwise, it is terminated. A packet also terminates upon exiting the tissue geometry. - Recording: Upon termination, if the packet exits the tissue at the top (detector surface), its remaining weight is added to the diffuse reflectance profile
R[r]. Weight exiting the bottom is added to transmittanceT[r].
This loop continues until all N packets are simulated. The accumulated A[r, z], R[r], and T[r] are normalized by the total number of photons and bin area to yield physically meaningful quantities.
Algorithm Logical Flow Diagram
Key Data Presentation: Optical Properties
The MCML algorithm requires precise input of layer-specific optical properties. The following table summarizes typical values for a two-layered model simulating skin, a common subject in pharmaceutical research.
Table 1: Standard Optical Properties for a Two-Layered Skin Model (λ ≈ 630 nm)
| Layer | Thickness (mm) | μₐ (cm⁻¹) | μₛ (cm⁻¹) | g | Refractive Index (n) |
|---|---|---|---|---|---|
| Epidermis | 0.06 | 2.0 - 4.0 | 300 - 500 | 0.80 - 0.90 | 1.45 |
| Dermis | 3.00 | 0.3 - 0.8 | 200 - 300 | 0.80 - 0.95 | 1.37 |
Experimental Protocol for Validation
To ensure physical accuracy, MCML simulations are typically validated against controlled phantom experiments or other analytical solutions.
Protocol: Validation Using a Homogeneous Slab Phantom
- Objective: To validate MCML code by comparing simulated diffuse reflectance with an analytical solution (e.g., Diffusion Equation) or benchmarked data.
- Materials: See "Scientist's Toolkit" below.
- Procedure:
- Phantom Characterization: Precisely measure the optical properties (μₐ, μₛ', n) of a homogeneous liquid or solid phantom using an established technique (e.g., integrating sphere measurement with inverse adding-doubling).
- Simulation Input: Use the measured properties as input for a single-layer MCML simulation.
- Parameter Setting: Set photon count (N) to 10⁷ or higher to minimize stochastic noise. Configure radial (r) and depth (z) bin resolutions (e.g., Δr = 0.01 mm, Δz = 0.001 mm).
- Execution: Run the MCML simulation.
- Data Extraction: Extract the radially-resolved diffuse reflectance profile, R(r).
- Comparison: Plot the simulated R(r) against the analytical/theoretical curve. Common metrics for comparison include relative root-mean-square error (RRMSE) or visual overlay of curves with error bounds.
The Scientist's Toolkit
Table 2: Essential Research Reagents & Materials for Phantom Validation Experiments
| Item | Function in MCML Context |
|---|---|
| Intralipid | A stable lipid emulsion, used as a tissue-mimicking scattering agent in liquid phantoms. Its scattering properties are well-documented. |
| India Ink | A strong absorber, used in minute quantities to titrate the absorption coefficient (μₐ) of liquid phantoms to desired values. |
| Solid Phantoms (e.g., Silicone, Polyurethane) | Provide stable, durable, and reproducible optical properties for long-term validation and instrument calibration. |
| Optical Fibers (Source & Detection) | Used in experimental setups to deliver light to phantoms and collect reflected/transmitted light for comparison with MCML output. |
| Index-Matching Fluids/Gels | Applied at phantom-fiber interfaces to reduce surface reflections not modeled in the standard MCML geometry. |
| Spectrophotometer with Integrating Sphere | The critical instrument for ex-vivo measurement of a sample's absolute μₐ and μₛ, providing ground-truth input for simulations. |
Simulation & Analysis Workflow Diagram
Within the broader thesis on Monte Carlo (MC) simulation for light propagation in scattering and absorbing media, the initialization and launch of photon packets constitute the critical first step. This process directly seeds the stochastic model, determining the accuracy, efficiency, and physical validity of the entire simulation of light-tissue interaction. Proper configuration is paramount for applications in biomedical optics, such as photodynamic therapy planning and diffuse optical tomography in drug development.
Core Principles and Mathematical Initialization
A photon packet is assigned a statistical weight, W, typically initialized to 1.0. Its launch state is defined by spatial, directional, and polarization properties.
Table 1: Standard Photon Packet Initialization Parameters
| Parameter | Symbol | Typical Value/Range | Description |
|---|---|---|---|
| Initial Weight | W₀ | 1.0 | Initial photon packet statistical weight. |
| Launch Position | r₀ | (0, 0, 0) | Cartesian coordinates (x, y, z) of source. |
| Initial Direction | û | (0, 0, 1) or (μx, μy, μz) | Unit direction cosine vector. |
| Step Size (Initial) | s | -ln(ξ)/μt | Calculated from random number ξ and total attenuation μt. |
The initial directional cosines for a collimated beam orthogonal to the surface are (0, 0, 1). For a divergent or anisotropic source, they are sampled from appropriate probability density functions (PDFs).
Diagram Title: Photon Packet Initialization and Launch Workflow
Experimental Protocols for Source Characterization
The following protocols are essential for empirically validating MC source models.
Protocol 3.1: Characterization of Source Spatial Profile
- Objective: Measure the irradiance profile of the physical light source.
- Materials: Diode laser, beam profiler (CCD/CMOS camera), translation stages, neutral density filters.
- Method: Attenuate the beam to avoid sensor saturation. Translate the beam profiler in the transverse (X-Y) plane at the intended source-plane position (z=0). Record intensity maps.
- Data Analysis: Fit the profile to a Gaussian or Top-Hat function. Use the extracted parameters (e.g., 1/e² radius) to define the spatial sampling PDF for r₀.
Protocol 3.2: Characterization of Source Angular Distribution
- Objective: Determine the angular divergence of the source.
- Materials: Collimated light source, goniometric rotation stage, photodetector, fixed aperture.
- Method: Place the photodetector behind a fixed aperture at a distance d. Rotate the source or detector while recording the detected power as a function of angle, θ.
- Data Analysis: Plot normalized power vs. θ. For Lambertian sources, verify a cos θ dependence. Use the measured distribution to sample û.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Experimental Validation
| Item | Function in Source Setup/Validation |
|---|---|
| Tunable Wavelength Laser (e.g., Ti:Sapphire) | Provides monochromatic light for wavelength-specific MC simulations, crucial for characterizing chromophore absorption. |
| Integrating Sphere with Spectrometer | Measures total radiant power of source for absolute calibration of photon packet weight W. |
| Tissue-Simulating Phantoms (e.g., with Intralipid, India Ink) | Provide media with precisely known scattering (μs) and absorption (μa) coefficients to validate launch and propagation algorithms. |
| Optical Fiber with Known NA | Defines a precise numerical aperture for source launching, enabling accurate sampling of initial direction cosines. |
| Digital Micromirror Device (DMD) | Can be used to create structured illumination patterns, allowing complex spatial initialization of photon packets for advanced imaging. |
For advanced imaging modalities, source initialization must model complex patterns.
Diagram Title: Complex Source Pattern Generation for MC
Table 3: Parameters for Structured Light Initialization
| Pattern Type | Initialization Parameters in MC | Application |
|---|---|---|
| Grid of Points | Array of r₀ coordinates. | Scanning microscopy simulation. |
| Hadamard Patterns | Weight W modulated per pixel based on pattern matrix H. | Compressive sensing in diffuse optical tomography. |
| Focused Beam (Gaussian) | r₀ sampled from 2D Gaussian PDF; û defined by lens NA. | Optical coherence tomography, confocal microscopy. |
Launch into a Layered Medium
A critical step is determining the initial intersection with a multi-layered tissue geometry. The algorithm must compute the distance to the first boundary along û.
Protocol 6.1: Launch and Boundary Handling Algorithm
- Compute the distance d_boundary from r₀ to the first planar or curved interface along û.
- If the initial step size s (sampled from μt of the first layer) is less than d_boundary, the photon moves s within the initial layer.
- If s ≥ d_boundary, the photon packet is moved to the boundary. Its weight is partially reflected and transmitted based on Fresnel equations, spawning new packets or adjusting weight accordingly.
- This precise launch-time boundary check ensures accurate modeling of specular reflection and refraction at the tissue surface.
This technical guide details the core algorithms for Monte Carlo simulation of light propagation in scattering and absorbing media, such as biological tissue. Framed within a broader thesis on computational biophotonics, it provides researchers and drug development professionals with the methodologies to simulate photon path length and scattering angle sampling, which are critical for applications like diffuse optical imaging and photodynamic therapy planning.
Monte Carlo (MC) simulation is the gold-standard stochastic technique for modeling light transport in turbid media. It tracks individual photon packets as they undergo absorption, scattering, and boundary interactions, providing a flexible, accurate solution to the radiative transfer equation (RTE). Accurate sampling of the free path length between interactions and the scattering angle at each scatter event is fundamental to the model's physical fidelity.
Core Physical Model and Governing Equations
Light propagation is characterized by the absorption coefficient (μa [mm⁻¹]), scattering coefficient (μs [mm⁻¹]), and anisotropy factor (g, dimensionless). The total interaction coefficient is μt = μa + μs. The phase function, typically the Henyey-Greenstein function, describes the angular scattering probability.
Table 1: Key Optical Properties for Common Tissues
| Tissue Type | μa (mm⁻¹) @ 650 nm | μs (mm⁻¹) @ 650 nm | g | Reference |
|---|---|---|---|---|
| Human Skin (dermis) | 0.02 - 0.05 | 15 - 25 | 0.85 - 0.9 | (Salomatina et al., 2006) |
| Human Brain (gray matter) | 0.03 - 0.06 | 20 - 30 | 0.85 - 0.95 | (Jacques, 2013) |
| Breast Tissue | 0.004 - 0.008 | 10 - 15 | 0.90 - 0.95 | (Tromberg et al., 2000) |
| Intralipid-20% (phantom) | ~0.01 | ~80 | ~0.7 | (Michels et al., 2008) |
Step-by-Step Sampling Algorithms
Sampling the Photon Path Length
The probability density function (PDF) for a photon traveling a distance s without interaction is: p(s) = μt exp(-μt s). The path length is sampled via the inverse cumulative distribution function (CDF) method using a random number ξ uniformly distributed on [0,1]: s = -ln(ξ) / μt
Experimental Protocol 1: Validating Path Length Distribution
- Setup: Define a homogeneous medium with known μt (e.g., μa=0.01 mm⁻¹, μs=10 mm⁻¹).
- Simulation: Launch 1,000,000 photon packets from a point source.
- Data Collection: For each photon's first step, record the sampled path length s.
- Analysis: Construct a histogram of s. Fit the data to an exponential decay model Aexp(-μt, fitted s). The fitted μt, fitted should match the input μt within statistical error (<1% for 10⁶ photons).
- Validation Metric: Perform a Chi-square goodness-of-fit test between the simulated distribution and the theoretical PDF.
Sampling the Scattering Angle (Henyey-Greenstein)
The Henyey-Greenstein (HG) phase function approximates single scatter in biological tissue: p(cos θ) = (1 - g²) / (2(1 + g² - 2g cos θ)^(3/2)). The scattering cosine, cos θ, is sampled using the CDF inversion method: cos θ = (1 + g² - [(1 - g²)/(1 - g + 2gξ)]²) / (2g), for g > 0. For isotropic scattering (g=0), cos θ = 2ξ - 1.
Experimental Protocol 2: Validating Scattering Angle Distribution
- Setup: Define a medium with a specific anisotropy g (e.g., 0.0, 0.8, 0.9).
- Simulation: For a single scatter event, sample cos θ 500,000 times for each g.
- Data Collection: Bin the cos θ values into histograms.
- Analysis: Compare the histogram to the theoretical HG function. For g=0, the distribution should be flat. For high g, a sharp peak near cos θ=1 should be observed.
- Validation Metric: Calculate the Kullback-Leibler divergence between the sampled and theoretical distributions; values <0.01 indicate excellent sampling.
Photon Propagation Workflow
Diagram Title: Monte Carlo Photon Propagation Core Loop
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials and Computational Tools for MC Simulation
| Item | Function in Research | Example/Supplier |
|---|---|---|
| Tissue-Simulating Phantoms | Provide ground-truth validation for simulations. Known optical properties. | Lipids (Intralipid), India Ink, TiO2 spheres, Agarose. |
| Spectrophotometer with Integrating Sphere | Measures bulk optical properties (μa, μs') of samples for simulation inputs. | PerkinElmer Lambda 1050+, Ocean Insight setups. |
| GPU Computing Platform (NVIDIA CUDA) | Accelerates MC simulations by 100-1000x compared to CPU, enabling complex 3D models. | NVIDIA Tesla/RTX series. |
| Validated MC Code Base | Foundation for building custom simulations. Ensures algorithm correctness. | MCML, tMCimg, GPU-based MCX (fangq.github.io/mcx/). |
| Inverse Adding-Doubling Software | Calculates intrinsic optical properties (μa, μs, g) from measured reflectance/transmittance. | IAD software (Oregon Medical Laser Center). |
| Optical Property Databases | Provide reference values for simulating specific tissues under various conditions. | IUPAC database, published compilations (e.g., S. L. Jacques). |
Advanced Considerations
Table 3: Comparison of Path Length & Angle Sampling Methods
| Sampling Aspect | Standard Method | Advanced Alternatives | Use Case |
|---|---|---|---|
| Path Length | Inverse CDF of exponential PDF. | Delta-tracking for complex media. | Homogeneous vs. heterogeneous voxelized media. |
| Scattering Angle | Henyey-Greenstein. | Mie Theory, Rayleigh-Gans, Measured Phase Functions. | When precise particle size distribution is known. |
| Azimuthal Angle | Uniform on [0, 2π]. | N/A for isotropic media. | Standard for randomly oriented scatterers. |
Experimental Protocol 3: Full Photon Migration in a Slab
- Setup: Model a 5 mm thick slab with properties: μa=0.05 mm⁻¹, μs=15 mm⁻¹, g=0.9, refractive index n=1.4.
- Source: Use a pencil beam normally incident on the surface.
- Simulation: Run 10⁷ photons using the described workflow.
- Output: Record the spatial distribution of absorbed energy (for dose) and the angular distribution of escaping photons (for reflectance/transmittance).
- Benchmark: Compare the simulated diffuse reflectance with results from a validated code (e.g., MCML). Discrepancy should be <2%.
Accurate sampling of photon path lengths and scattering angles forms the computational backbone of credible Monte Carlo simulations for light propagation. This guide provides the foundational algorithms, validation protocols, and necessary toolkit for researchers to implement these critical steps, thereby enhancing the reliability of simulations used in therapeutic and diagnostic development.
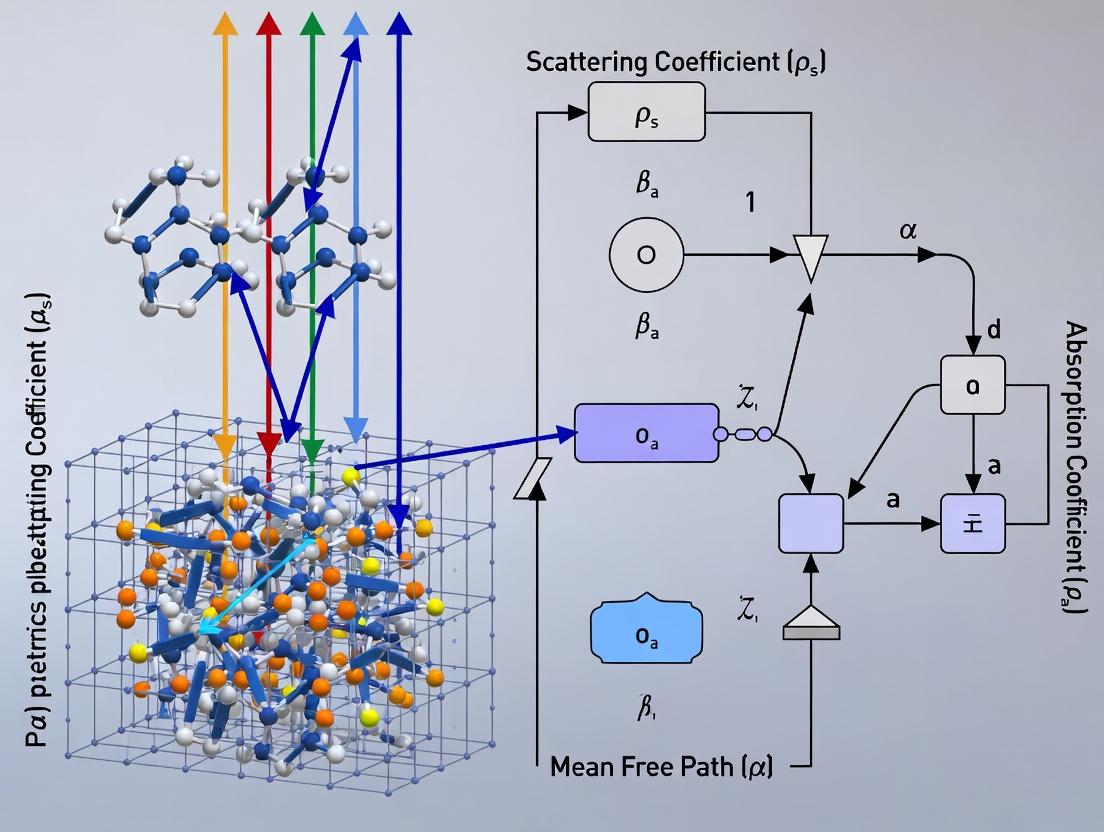
Handling Absorption, Scattering Events, and Boundary Conditions
Within the broader thesis on Monte Carlo (MC) simulation for light propagation in scattering and absorbing media, the accurate computational handling of absorption, scattering events, and boundary conditions is the foundational core. These three elements collectively define the radiative transfer equation's stochastic solution, determining the accuracy of simulated quantities such as fluence rate, reflectance, and transmittance. This guide details the technical implementation and current best practices for these critical components, directly impacting applications in biomedical optics, drug development (e.g., photodynamic therapy dosimetry), and tissue diagnostics.
Core Physics and Stochastic Implementation
Photon Packet and Weight
The continuous-wave MC method typically employs a "photon packet" with an initial weight, W = 1. This weight represents the fraction of energy remaining in the packet as it propagates.
Handling Absorption: The Null-Collision and Weight-Deposition Method
Pure absorption terminates photon packets inefficiently. The standard approach is the absorption weighting method. At each interaction point, the photon packet weight is partially deposited, and the packet continues to propagate until it exits or is terminated by a roulette threshold.
- Absorption Event:
- The packet interacts with the medium defined by absorption coefficient µₐ and scattering coefficient µₛ.
- A fraction of the weight, ∆W = W * (µₐ / (µₐ + µₛ)), is deposited into the local voxel or region of the simulation mesh.
- The packet weight is updated: W = W - ∆W = W * (µₛ / (µₐ + µₛ)).
- The packet continues its journey with a reduced weight.
Handling Scattering Events: Phase Function Sampling
Following weight deposition for absorption, the packet is scattered. The new direction is determined by sampling the scattering phase function.
Henvey-Greenstein (HG) Phase Function: The most common anisotropic model. The scattering angle θ is sampled via:
- cos θ = (1 / (2g)) * [1 + g² - ((1 - g²) / (1 - g + 2g * ξ))² ], if g ≠ 0.
- cos θ = 2ξ - 1, if g = 0. where g is the anisotropy factor ([-1, 1]) and ξ is a uniform random number in [0,1). The azimuthal angle φ is sampled as φ = 2π * ξ’.
Experimental Protocol for MC Scattering Validation: A common benchmark simulates a narrow collimated beam incident on a purely scattering slab (g = 0.9). The radial reflectance profile is recorded and compared to established results from known codes (e.g., MCML) or analytical approximations.
Table 1: Standard Scattering Phase Functions
| Phase Function | Formula (p(cosθ)) | Parameter(s) | Typical Use Case |
|---|---|---|---|
| Isotropic | 1 / (4π) | None | Simple media, benchmark tests |
| Henvey-Greenstein | (1 / 4π) * (1 - g²) / (1 + g² - 2g cos θ)^(3/2) | Anisotropy factor g | Biological tissues (g ~ 0.7 - 0.9) |
| Mie | Complex series solution | Particle size, refractive index | Cell suspensions, engineered media |
| Rayleigh | (3 / 16π) * (1 + cos²θ) | None | Very small particles relative to λ |
Handling Boundary Conditions: Fresnel Reflection and Refraction
At an interface between media with refractive indices n₁ (incident) and n₂ (transmitted), the photon packet may reflect internally or transmit.
- Calculation: The critical angle is θ_c = arcsin(n₂/n₁). For an incident angle θ_i, the reflection coefficient R(θ_i) is calculated via Fresnel's equations for both polarization components and averaged for unpolarized light.
- Stochastic Implementation:
- Calculate R(θi).
- Generate a random number ξ.
- If ξ ≤ R(θi), the packet is specularly reflected back into the current medium (θr = θi).
- If ξ > R(θi), the packet is refracted into the adjacent medium, with angle θt = arcsin((n₁/n₂) sin θi).
- For a matched boundary (n₁ = n₂), R(θi) = 0, and transmission is always total.
Table 2: Common Boundary Conditions in Tissue MC
| Condition | Refractive Index Match | Reflection Handling | Typical Application |
|---|---|---|---|
| Matched | n_tissue = n_external | None (R=0) | Idealized in-silico experiments |
| Mismatched | n_tissue ≠ n_external | Fresnel equations | Realistic air/tissue or glass/tissue interfaces |
| Specular | Any | Reflection of initial photon launch point | Glass cover slip, air/glass/tissue layer models |
| Cyclic | N/A | Packet exits one boundary and re-enters opposite | Modeling infinite laterally-extended media |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Experimental Validation of MC Models
| Item / Reagent | Function in Context |
|---|---|
| Intralipid | A standardized, lipid-based emulsion used as a tissue-mimicking phantom with well-characterized and tunable scattering properties (µₛ, g). |
| India Ink | A strong absorber used to titrate absorption coefficient (µₐ) in liquid or solid phantoms. |
| Agarose or Gelatin | Biocompatible gelling agents used to solidify liquid phantoms into stable, solid forms for precise geometry experiments. |
| Optical Fibers (Source & Detector) | For delivering light to and collecting light from tissue or phantoms. Core diameter and numerical aperture define the effective source/detector condition in experiments. |
| Spectrophotometer with Integrating Sphere | The gold standard for ex-vivo measurement of bulk optical properties (µₐ, µₛ') of thin tissue samples or phantom materials. |
| TiO₂ or Polystyrene Microspheres | Monodisperse scattering particles used to create phantoms with precisely calculable (Mie theory) scattering properties. |
Visualized Workflows
Diagram 1: Core Monte Carlo photon packet loop.
Diagram 2: Stochastic boundary condition decision logic.
This technical guide details the core calculations for reflectance, transmittance, and fluence within the broader context of Monte Carlo simulation (MCS) research for modeling light propagation in scattering and absorbing media. These parameters are fundamental for applications in biomedical optics, photodynamic therapy, and tissue diagnostics.
In Monte Carlo simulations of light transport, the primary outputs requiring rigorous calculation are the spatial and temporal distributions of light energy. Reflectance (R) is the fraction of incident light back-scattered from a medium, transmittance (T) is the fraction that passes through, and fluence (φ) is the total light energy delivered per unit area at a specific point within the medium. Accurate computation of these metrics validates simulations against empirical data.
Quantitative Data from Recent Literature
The following table summarizes key parameters and typical results from recent MCS studies in biological media.
Table 1: Summary of Optical Properties and Calculated Outputs from Recent MCS Studies
| Study & Application (Year) | Medium Simulated | μa (cm⁻¹) | μs' (cm⁻¹) | g | Calculated Reflectance | Calculated Transmittance | Key Fluence Finding |
|---|---|---|---|---|---|---|---|
| Chen et al., Skin Diagnosis (2023) | Epidermis (600 nm) | 0.4 | 40 | 0.85 | 0.12 ± 0.02 | 0.01 ± 0.005 | Peak fluence at 0.2 mm depth. |
| Ozeki et al., Brain Imaging (2024) | Gray Matter (800 nm) | 0.15 | 22 | 0.90 | 0.08 ± 0.01 | N/A (semi-infinite) | Fluence decays exponentially beyond 3 mm. |
| Park et al., PDT Planning (2023) | Tumor Phantom (660 nm) | 0.8 | 15 | 0.80 | 0.05 ± 0.01 | 0.15 ± 0.03 | 40% higher fluence at tumor core vs periphery. |
Core Calculation Methodologies
Reflectance and Transmittance
These are calculated by binning photon packets that exit the medium at the launch surface (reflectance) or the opposite boundary (transmittance).
Experimental Protocol (for Simulation Validation):
- Phantom Preparation: Fabricate a solid or liquid tissue-simulating phantom with known optical properties (µa, µs', g) using materials like Intralipid (scatterer) and India ink (absorber).
- Instrumentation: Use a calibrated integrating sphere coupled to a spectrophotometer or a dedicated tissue spectrophotometer.
- Measurement: Illuminate the phantom with a collimated light source at wavelength λ. The integrating sphere collects all back-scattered (R) or forward-transmitted (T) light.
- Reference Measurement: Perform identical measurements with a known reflectance standard (e.g., Spectralon) and without the sample for baseline.
- Calculation: ( R(\lambda) = \frac{I{sample}^{R} - I{dark}}{I{reference}^{R} - I{dark}} \times R_{reference} ). A similar process yields T.
- Simulation Input: Use the measured µa and µs' of the phantom as direct input for the MCS.
- Comparison: The simulated R and T, calculated as the sum of weights of escaped photon packets divided by total launched photons, are directly compared to empirical measurements.
Fluence Rate Calculation
Fluence rate (φ, in W/cm²) is the total radiant power incident on an infinitesimally small sphere, divided by the cross-sectional area of that sphere. In MCS, it is typically estimated using a 3D voxel-based or radial binning method.
Computational Protocol:
- Spatial Binning: Define a 3D grid over the simulated volume.
- Photon Packet Tracking: As each photon packet propagates, its remaining weight (w) is deposited into the voxel (i,j,k) where the scattering/absorption event occurs.
- Path Length Integration: The contribution to fluence in a voxel is proportional to the product of the photon packet's weight and the path length (Δs) traveled within that voxel: ( Δφ = \frac{w \cdot Δs}{V_{voxel}} ).
- Normalization: After simulating N photons, the total fluence rate in each voxel is summed and normalized by the number of photons and the voxel volume: ( φ(i,j,k) = \frac{\sum{n=1}^{N} \sum{m} w{n,m} \cdot Δs{n,m}}{N \cdot V_{voxel}} ).
- Temporal Resolution: For time-resolved fluence, photon path lengths are also binned by time-of-flight.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Materials for Experimental Validation of MCS
| Item | Function in Context |
|---|---|
| Intralipid 20% | A standardized lipid emulsion providing highly reproducible optical scattering (µs') in tissue-simulating phantoms. |
| India Ink / Nigrosin | A strong, broadband absorber used to titrate the absorption coefficient (µa) in liquid phantoms. |
| Agarose or Silicone | Transparent gelling/solidifying agents for creating stable, solid optical phantoms with defined geometry. |
| Spectralon | A diffuse reflectance material with >99% reflectance across a broad spectrum, used as a calibration standard for reflectance measurements. |
| Calibrated Integrating Sphere | Captures all light reflected from or transmitted through a sample, enabling absolute measurement of R and T. |
| Optical Fiber Probes | For interstitial measurement of fluence within phantoms or ex vivo tissue to validate internal light fields. |
| Tunable Wavelength Laser | Provides monochromatic, collimated light for precise, wavelength-specific measurements of optical properties. |
Visualization of Workflows and Relationships
Monte Carlo Calculation Workflow for R, T, and Fluence
Validation Loop for Monte Carlo Simulation Models
This document is framed within a broader thesis on Monte Carlo (MC) simulation for light propagation in scattering and absorbing media. The accurate modeling of photon transport is fundamental to both the development and quantitative interpretation of biomedical optical techniques. MC methods provide a stochastic, yet rigorous, solution to the radiative transfer equation, enabling the simulation of light distribution in complex, heterogeneous tissues. This capability is critical for optimizing device parameters, interpreting diagnostic signals, and planning therapeutic interventions. This guide explores two pivotal applications—Optical Coherence Tomography (OCT) and Photodynamic Therapy (PDT)—where MC simulations directly impact research and clinical translation by elucidating the interplay of scattering, absorption, and fluorescence in biological media.
Optical Coherence Tomography (OCT): Principles and MC Modeling
OCT is a non-invasive, high-resolution imaging modality analogous to ultrasound, using light instead of sound. It performs cross-sectional imaging by measuring the echo time delay and intensity of backscattered light using low-coherence interferometry.
MC Role in OCT: MC simulations are used to model the OCT signal, especially in Fourier-Domain OCT. They help decode how scattering properties (anisotropy factor g, scattering coefficient μₛ) influence the depth-dependent backscattered signal, A-scan shape, and imaging depth. This is crucial for correcting attenuation artifacts, developing advanced angiographic algorithms, and quantifying tissue optical properties.
Key Experimental Protocol: Quantifying Scattering Properties with OCT
- Objective: To extract the depth-resolved attenuation coefficient (μₜ) from OCT data for tissue characterization.
- Materials: Spectral-Domain OCT system, tissue phantom or ex vivo/in vivo sample, reference standards.
- Protocol:
- System Calibration: Acquire A-scans from a known, weakly scattering reference (e.g., silicone layer) to calibrate for system-specific confocal function and sensitivity roll-off.
- Data Acquisition: Acquire 3D OCT volumes of the sample. Ensure proper signal-to-noise ratio (SNR).
- Pre-processing: Subtract noise floor, apply logarithmic transformation to A-scans.
- Depth-Resolved Fitting: For each A-scan, fit the linear portion of the logarithmized intensity decay versus depth using a least-squares algorithm: I(z) ∝ exp(-2μₜz). The factor of 2 accounts for the round-trip attenuation.
- MC Validation: Generate a matching MC simulation with estimated μₛ and g. Compare the simulated OCT A-scan profile with the experimental data. Iteratively adjust optical properties in the simulation to achieve the best fit, thereby extracting accurate μₛ and g.
- Data Output: A 2D map of the attenuation coefficient μₜ (≈ μₛ for low absorption) across the tissue sample.
Table 1: Typical Optical Properties for OCT-Relevant Tissues & Phantoms
| Material/Tissue Type | Scattering Coefficient μₛ (mm⁻¹) @ 1300 nm | Anisotropy Factor g @ 1300 nm | Attenuation Coefficient μₜ (mm⁻¹) | Key Application |
|---|---|---|---|---|
| Intralipid 1% | ~1.2 | ~0.2 | ~1.44 | Calibration phantom |
| Human Skin (Epidermis) | 6 – 8 | 0.80 – 0.90 | ~1.5 – 2.0 | Dermatology, cancer detection |
| Human Retina | 3 – 5 | 0.85 – 0.95 | ~0.6 – 1.0 | Ophthalmology |
| Atherosclerotic Plaque | 4 – 10 | 0.85 – 0.95 | 1.5 – 4.0 | Cardiology |
| Polystyrene Beads in Gel | Tunable (0.5-10) | ~0.8-0.9 | Tunable | Validation phantom |
Diagram 1: OCT Data Analysis and MC Validation Workflow.
Photodynamic Therapy (PDT): Principles and MC Modeling
PDT is a photochemotherapy involving the administration of a photosensitizer (PS), its selective accumulation in target tissue, and activation by light of a specific wavelength. This generates cytotoxic reactive oxygen species (ROS), primarily singlet oxygen (¹O₂), leading to cell death.
MC Role in PDT: MC simulations are the gold standard for predicting the light fluence rate distribution (φ) within tissue during PDT. This is essential for treatment planning, as the local photodynamic dose (D) is proportional to φ, PS concentration, and oxygen availability. MC models account for tissue geometry, optical properties at the treatment wavelength, and the light source configuration to ensure therapeutic light levels are reached at the target while sparing healthy tissue.
Key Experimental Protocol: PDT Dosimetry Planning for Superficial Lesions
- Objective: To plan light delivery for a superficial skin carcinoma PDT protocol using MC simulation.
- Materials: Clinical PDT laser/LED, photosensitizer (e.g., Protoporphyrin IX), optical property data, MC simulation software (e.g., MCML, tMCimg, or custom code).
- Protocol:
- Tissue Characterization: Obtain μₐ, μₛ', and g for the target tissue (tumor and surrounding normal) at the PS activation wavelength (e.g., 630 nm for PpIX) from literature or diffuse reflectance measurements.
- Define Geometry: Model the treatment area as a multi-layer structure (e.g., stratum corneum, epidermis, tumor, dermis).
- MC Simulation Setup: Define source parameters (beam profile, diameter, power, angle). Run simulation with >10⁷ photons.
- Fluence Rate Calculation: Output the volumetric fluence rate distribution φ(x,y,z). Determine required irradiation time (T) to deliver the target fluence (J/cm²) at the deepest tumor boundary: T = Target Fluence / φatdepth.
- Treatment Validation: Use an isotropic light detector placed on the tissue surface during a mock irradiation to validate the simulated surface fluence rate.
- Data Output: A 3D map of predicted fluence rate and a prescribed irradiation time for the clinical protocol.
Table 2: Key Optical Properties for PDT Planning at 630 nm
| Tissue/Component | Absorption Coefficient μₐ (mm⁻¹) | Reduced Scattering Coefficient μₛ' (mm⁻¹) | Photosensitizer Concentration [PS] (μM) | Critical Fluence (J/cm²) |
|---|---|---|---|---|
| Normal Skin | 0.1 – 0.3 | 1.5 – 2.5 | < 0.1 | N/A |
| Basal Cell Carcinoma | 0.2 – 0.4 | 1.2 – 2.0 | 1.0 – 3.0 (after ALA) | ~30 - 150 |
| Protoporphyrin IX (PpIX) | Extinction Coefficient ε: ~5000 M⁻¹cm⁻¹ | |||
| Blood Vessels (Oxy) | μₐ ≈ 0.3 (highly dependent on Hb/HbO₂) |
Diagram 2: Core Photodynamic Therapy Molecular Pathway.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for OCT and PDT Research
| Item Name | Function/Description | Primary Application |
|---|---|---|
| Intralipid 20% Emulsion | A standardized lipid emulsion used as a scattering agent to create tissue-simulating phantoms with tunable μₛ and g. | OCT/PDT phantom development & system calibration. |
| Polystyrene Microspheres | Monodisperse particles providing highly predictable and controlled scattering properties (based on size) in gel phantoms. | High-precision validation of MC models and OCT systems. |
| Aminolevulinic Acid (ALA) or Methyl-ALA (MAL) | Prodrugs that induce the endogenous synthesis of the photosensitizer Protoporphyrin IX (PpIX) in metabolically active cells. | Clinical and preclinical PDT research, particularly for skin and brain. |
| Singlet Oxygen Sensor Green (SOSG) | A highly selective fluorescent probe for detecting and quantifying singlet oxygen (¹O₂) generation in solution. | In vitro PDT efficacy testing and PS photophysics characterization. |
| Tissue Optical Property Phantoms (Solid) | Stable, solid gels or resins with embedded scatterers and absorbers (e.g., TiO₂, India Ink) of certified optical properties. | Longitudinal performance monitoring of OCT and PDT light delivery systems. |
| Isotropic Fluence Probes | Miniature optical detectors with spherical diffusers that collect light from all directions, measuring true fluence rate at a point. | In situ validation of MC-simulated light distributions during PDT. |
| Monte Carlo Simulation Software (e.g., MCML, GPU-MC) | Open-source or commercial code implementing MC for multi-layered (MCML) or voxelated (GPU-MC) geometries. | Core tool for predicting light propagation in all applications. |
Solving Computational Challenges: Tips for Faster, More Accurate Simulations
Monte Carlo (MC) simulation is the gold-standard numerical method for modeling light propagation in turbid, scattering, and absorbing media, such as biological tissue. It is indispensable in biomedical optics research, particularly for applications in drug development—including photodynamic therapy dosimetry, diffuse optical tomography, and fluorescence-guided surgery. The core principle involves simulating the stochastic trajectories of millions of photons, with their scattering, absorption, and propagation governed by probability distributions derived from tissue optical properties (e.g., absorption coefficient μa, scattering coefficient μs, anisotropy g). Despite its conceptual simplicity and flexibility, the method's reliability hinges on managing inherent statistical uncertainties. This whitepaper details the critical pitfalls of variance, convergence, and statistical noise within this research context.
Core Quantitative Metrics & Data
The performance and accuracy of an MC simulation are quantified by specific metrics. The following table summarizes key parameters, their typical values in biomedical optics, and their impact on statistical noise.
Table 1: Key Quantitative Parameters in Monte Carlo Light Simulation
| Parameter | Symbol | Typical Range in Tissue | Role & Impact on Statistical Noise |
|---|---|---|---|
| Number of Photons | N | 10⁶ – 10¹⁰ | Directly influences convergence. Variance ∝ 1/N. |
| Absorption Coefficient | μa (cm⁻¹) | 0.01 – 1.0 | Higher μa reduces detected photons, increasing relative noise. |
| Scattering Coefficient | μs (cm⁻¹) | 10 – 1000 | Higher μs increases pathlengths, requiring more photons for convergence. |
| Anisotropy Factor | g | 0.7 – 0.99 (highly forward) | High g reduces effective scattering, can lead to deeper penetration and slower convergence in some detectors. |
| Voxel/Detector Size | Δx, Δy, Δz (cm) | 0.01 – 0.1 | Smaller bins collect fewer photons, increasing local variance (noise). |
| Figure of Merit (FOM) | (S/N)² / Time | Variable | Metric for simulation efficiency. Optimizing variance reduction techniques maximizes FOM. |
Table 2: Common Variance Reduction Techniques & Efficacy
| Technique | Principle | Effect on Variance | Convergence Rate Impact | Primary Pitfall |
|---|---|---|---|---|
| Analog (Brute-Force) MC | Tracks every photon event without bias. | Baseline (High) | Slow, but unbiased. | Computationally prohibitive for deep penetration. |
| Photon Weight & Roulette | Photons carry a weight; Russian Roulette terminates low-weight photons. | High reduction in low-signal regions. | Greatly accelerated. | Can introduce bias if thresholds are poorly set. |
| Explicit Photon Detection | Forces scattering towards detector region. | Dramatic reduction for specific detectors. | Extremely fast for targeted queries. | Biases overall fluence map; only valid for specific detection points. |
| Subsurface Scoring | Scores weight contributions along path, not just at interaction points. | Reduces variance in dense scoring grids. | Improves efficiency for volumetric data. | Increased memory footprint per photon. |
Experimental & Simulation Protocols
Protocol for Benchmarking Convergence
Aim: To determine the minimum number of photons (N) required for a converged solution of fluence φ(r) within a specified error tolerance.
- Define Geometry & Optics: Set a multi-layered tissue model (e.g., epidermis, dermis, fat) with known μa, μs, g for each layer.
- Run Iterative Batches: Execute a series of independent MC simulations with increasing N (e.g., N = 10³, 10⁴, 10⁵, 10⁶, 10⁷).
- Calculate Relative Error: For each batch i, compute the fluence φi(r) at a critical point (e.g., a tumor depth). Compute the relative difference compared to the highest-N run (assumed as ground truth): Errori = |φi - φmax| / φ_max.
- Assess Variance: Compute the normalized standard deviation (noise) in a region of interest across multiple sub-runs.
- Determine Convergence Criterion: Identify N where Errori < ε (e.g., ε = 1%) and the noise plateau is reached. Plotting Errori and noise vs. log(N) reveals the convergence profile.
Protocol for Validating Against Analytical Solutions
Aim: To quantify statistical noise and bias introduced by variance reduction techniques.
- Select a Solvable Scenario: Use a simple geometry (e.g., infinite homogeneous medium) where the diffusion equation or other analytical solutions for fluence are valid.
- Run MC with & without Bias: Execute two MC simulations: a. Analog MC: High-N (e.g., 10⁸) simulation as a reference. b. Biased MC: Simulation using a variance reduction technique (e.g., photon weight with roulette, explicit detection) with a lower N (e.g., 10⁶).
- Compare to Analytical Model: Calculate the analytical fluence φ_analytical(r) at multiple radial distances.
- Quantify Discrepancies: Compute:
- Statistical Noise: Standard deviation of φ_MC in a stable region.
- Bias: Mean(φbiased MC - φanalytical).
- Mean Squared Error (MSE): Sum of Variance + Bias².
- Optimize Technique Parameters: Adjust parameters (e.g., roulette weight threshold) to minimize MSE, not just variance.
Visualization of Concepts and Workflows
Flowchart of a Basic Weighted Monte Carlo Photon Path
Relationship Map of Pitfalls, Causes, Symptoms, and Remedies
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Components for MC Simulation in Biomedical Optics
| Item / "Reagent" | Function in the "Experiment" (Simulation) | Key Considerations |
|---|---|---|
| Validated MC Code Base (e.g., MCML, tMCimg, GPU-based codes) | Core engine for photon transport. Provides the algorithmic framework. | Choose between standardized codes (for validation) or custom codes (for flexibility). GPU acceleration is critical for high N. |
| High-Quality Pseudo-Random Number Generator (RNG) | Sources randomness for sampling scattering angles, step lengths, etc. | Quality directly impacts statistical accuracy. Use Mersenne Twister or similar. Avoid low-period RNGs. |
| Accurate Optical Property Database (μa, μs, g, n) | Defines the simulated medium's interaction probabilities. | Values must be sourced from peer-reviewed measurements at the correct wavelength. Uncertainty here dwarfs simulation noise. |
| Variance Reduction "Plug-ins" | Modules for photon weight, roulette, forced detection, etc. | Must be carefully validated to ensure they reduce variance without introducing unacceptable bias for the specific query. |
| Convergence Diagnostic Scripts | Automated tools to run Protocol 3.1 and analyze error vs. N. | Essential for justifying the chosen N in any publication. |
| Analytical Solution Benchmarks (e.g., Diffusion Eq. solvers) | Provides a gold standard for validation in simple geometries (Protocol 3.2). | Used to calibrate and verify the MC implementation before complex use. |
| High-Performance Computing (HPC) Resources | Enables running large N (10⁹-10¹⁰) in feasible time. | Cloud computing or local GPU clusters are now standard for production-level simulations. |
Within the broader thesis on advanced Monte Carlo (MC) simulation for light propagation in scattering and absorbing media, efficiency is paramount. Simulating photon migration in biological tissues, such as for drug development in photodynamic therapy or optical tomography, involves tracking millions of photon packets through interactions governed by probabilities of absorption and scattering. Naïve MC simulations can be computationally prohibitive, as variance remains high with low signal (e.g., in deep tissues or highly absorbing regions). This whitepaper details two complementary variance reduction techniques (VRTs) critical for making these simulations tractable: Importance Sampling and Russian Roulette.
Theoretical Foundation
Monte Carlo estimators compute an expected value: I = ∫_Ω f(x) p(x) dx, where p(x) is the original probability density function (PDF). The key to VRTs is to alter the sampling process strategically to reduce estimator variance, σ², without introducing bias.
Importance Sampling (IS) changes the sampling distribution to focus on regions that contribute more to the integral. We sample from a new proposal distribution, q(x), and re-weight the results:
I = ∫_Ω [f(x)p(x)/q(x)] q(x) dx. The optimal q*(x) ∝ |f(x)p(x)|.
Russian Roulette (RR) addresses inefficiency from low-contribution samples. It probabilistically terminates a simulation path (e.g., a photon packet with low weight) while compensating to maintain an unbiased estimate.
Application to Photon Migration
In a MC simulation for light propagation, a photon packet has a weight W and survival probability at each interaction.
Importance Sampling Protocols
Experiment 1: Biasing Scattering Angles for Forward-Directed Tissues
- Objective: Reduce variance in detectors placed opposite the light source in tissues with high anisotropy factor (
g). - Methodology: The natural scattering angle cosine is sampled from the Henyey-Greenstein PDF,
p(cosθ). For a detector in the forward direction, a biased PDF,q(cosθ), is defined to favor forward scattering. The photon weight is multiplied byp(cosθ)/q(cosθ)at each scattering event. - Protocol: For a photon packet scattering event:
- Calculate the original PDF value
p(cosθ)for the sampled angle. - Sample the scattering angle cosine
cosθ'from the biased distributionq(cosθ)(e.g., a modified Henyey-Greenstein with higherg'). - Compute the biased PDF value
q(cosθ'). - Update photon weight:
W_new = W_old * p(cosθ') / q(cosθ'). - Continue propagation with the new direction
cosθ'.
- Calculate the original PDF value
- Objective: Reduce variance in detectors placed opposite the light source in tissues with high anisotropy factor (
Experiment 2: Path Length Stretching for Low-Absorption Regions
- Objective: Increase sampling efficiency in spectral windows where absorption coefficient (
μ_a) is low relative to scattering (μ_s). - Methodology: The natural path length
lis sampled fromp(l) = μ_t exp(-μ_t l), whereμ_t = μ_a + μ_s. Using a "stretched" coefficientμ_t' < μ_t, we defineq(l) = μ_t' exp(-μ_t' l). Longer jumps are sampled more frequently, and the weight is adjusted accordingly.
- Objective: Increase sampling efficiency in spectral windows where absorption coefficient (
Russian Roulette Protocols
- Experiment 3: Combined Weight Thresholding and Russian Roulette
- Objective: Terminate negligible photon packets without biasing the result, saving computational resources.
- Methodology: A weight threshold
W_thand a survival probabilityP_s(e.g., 0.1) are defined. For a packet with weightW:- If
W > W_th, continue propagation normally. - If
W ≤ W_th, generate a random numberξ ∈ [0,1]. - If
ξ < P_s, setW_new = W / P_sand continue propagation. - If
ξ ≥ P_s, terminate the photon packet.
- If
Data Presentation
Table 1: Comparative Performance of VRTs in a Semi-Infinite Slab Model (Simulation: 10^7 photons, μ_s = 10 cm⁻¹, μ_a = 0.1 cm⁻¹, g = 0.9, detector at 1 cm depth)
| Technique | Parameters | Relative Variance (at Detector) | Computational Time (s) | Speed-Up Factor |
|---|---|---|---|---|
| Naïve MC | - | 1.00 (baseline) | 342 | 1.0 |
| Importance Sampling | Biased g' = 0.95 |
0.45 | 355 | 1.2* |
| Russian Roulette | W_th = 0.001, P_s = 0.1 |
1.05 | 210 | 1.6 |
| Combined IS+RR | g' = 0.95, W_th = 0.001, P_s = 0.1 |
0.48 | 225 | 2.3 |
*Speed-up factor normalizes variance reduction against time: (Baseline Variance/Baseline Time) / (New Variance/New Time).
Table 2: Key Research Reagent Solutions (Simulation Parameters & Functions)
| Item | Typical Value/Range | Function in Simulation |
|---|---|---|
Anisotropy Factor (g) |
0.7 - 0.99 | Defines the directionality of scattering events in tissue. |
Absorption Coefficient (μ_a) |
0.01 - 1.0 cm⁻¹ | Determines the probability of light absorption per unit path length. |
Scattering Coefficient (μ_s) |
10 - 100 cm⁻¹ | Determines the probability of a scattering event per unit path length. |
Refractive Index (n) |
1.33 - 1.5 | Governs reflection and refraction at tissue boundaries. |
| Photon Packet Launch Weight | 1.0 | Initial energy assigned to each simulated photon bundle. |
| Random Number Generator | Mersenne Twister | Provides high-quality pseudo-random numbers for stochastic sampling. |
Weight Threshold (W_th) |
10⁻³ - 10⁻⁶ | Threshold below which Russian Roulette is triggered. |
Survival Probability (P_s) |
0.05 - 0.2 | Probability of surviving Russian Roulette; controls variance/computation trade-off. |
Visualized Workflows
Title: Importance Sampling for Scattering Angle Bias
Title: Russian Roulette Decision Workflow
Title: VRTs in the Thesis Context
In the computational modeling of light propagation in scattering and absorbing media for biomedical research, such as photodynamic therapy planning or tissue oxygenation monitoring, Monte Carlo (MC) simulation is the gold standard. Its statistical accuracy, however, comes at an extreme computational cost. This whitepaper provides an in-depth technical guide on optimizing these simulations through strategic parallelization (leveraging GPU vs. CPU architectures) and the implementation of efficient, domain-specific data structures.
Parallelization Paradigms: GPU vs. CPU for Monte Carlo Photon Transport
Monte Carlo simulations are inherently parallelizable, as each photon packet can be traced independently. The choice between CPU and GPU parallelization hinges on the problem scale, memory access patterns, and algorithmic complexity.
Key Architectural Considerations:
- CPU (Multicore/MPI): Optimized for complex, sequential tasks with heavy branching and large cache hierarchies. Suited for "few but fat" threads. Scaling is achieved across multiple cores or nodes via OpenMP or MPI.
- GPU (CUDA/OpenCL): Optimized for massively parallel, "many but thin" threads executing the same instruction (SIMD/SIMT). Requires minimal branching and coalesced memory access for peak performance. Ideal for tracing millions of photon packets with identical kernels.
Quantitative Performance Comparison:
Table 1: GPU vs. CPU Performance in Monte Carlo Light Transport Simulations (Representative Data)
| Metric | High-End CPU (e.g., AMD EPYC 9654) | High-End GPU (e.g., NVIDIA H100) | Performance Ratio (GPU/CPU) | Notes |
|---|---|---|---|---|
| Peak FP64 TFLOPs | ~3.5 | ~34 | ~9.7x | Theoretical peak for dense math. |
| Memory Bandwidth | ~460 GB/s | ~3350 GB/s | ~7.3x | Critical for random number access. |
| Typical Photon Throughput | 10^5 - 10^6 photons/sec/core | 10^7 - 10^8 photons/sec/GPU | 50-200x | Real-world simulation, optimized code. |
| Latency | Low | High | - | GPU kernel launch overhead. |
| Branch Handling | Excellent (Speculative Execution) | Poor (Thread Divergence Penalty) | - | Complex media can cause divergence. |
| Best For | Complex boundary conditions, small-scale simulations, validation runs. | Homogeneous, large-scale simulations with millions of photons. |
Experimental Protocol for Benchmarking:
- Algorithm: Implement a standard MCML-like algorithm for multi-layered tissue.
- Baseline: Create a single-threaded, optimized CPU reference implementation.
- CPU Parallelization: Implement using OpenMP, distributing photon packets across available cores. Use thread-safe random number generators (RNGs).
- GPU Parallelization: Implement in CUDA. Assign one photon packet per thread. Use GPU-optimized RNGs (e.g., cuRAND). Employ shared memory for local geometry data and coalesced global memory writes for absorption and fluence maps.
- Control Variables: Simulate 10^8 photon packets in a standard skin model (epidermis, dermis, fat). Record total computation time, excluding initialization and I/O.
- Metrics: Report photons/second and speed-up factor relative to the single-threaded CPU baseline.
Efficient Data Structures for Spatial and Energy Tracking
The choice of data structure for storing simulation results (e.g., volumetric absorbed energy, surface fluence) dramatically impacts performance, especially on GPUs.
- Dense Arrays (Voxel Grid): The default structure. Allows O(1) access via voxel indexing. Inefficiency: High memory footprint; poor cache usage if only a sub-volume is illuminated.
- Sparse Structures: Critical for large, mostly empty domains.
- Compressed Sparse Row (CSR): Stores only non-zero voxels. Efficient for matrix operations but expensive for parallel, unordered atomic addition from millions of threads.
- Hash Grids (Key Innovation): Map 3D voxel coordinates (i, j, k) to a 1D key using a spatial hash function. A concurrent hash table (e.g., CUDA's
concurrent_unordered_map) allows multiple threads to insert/update absorption data with high throughput and minimal memory.
Table 2: Comparison of Data Structures for Storing Volumetric Absorption Data
| Data Structure | Memory Efficiency | Access Complexity (Avg) | Parallel Write Suitability | Best Use Case |
|---|---|---|---|---|
| Dense 3D Array | Poor (Allocates all voxels) | O(1) | Good (Coalesced atomic adds) | Small, uniformly illuminated volumes. |
| Compressed Sparse Row (CSR) | Excellent | O(log n) for search | Poor (Requires pre-allocation) | Post-processing of known sparse data. |
| Spatial Hash Grid | Very Good | O(1) | Excellent (Concurrent hash map) | Large-scale GPU simulations with unknown, dynamic photon paths. |
| Linear Octree | Excellent | O(log n) | Moderate | Adaptive mesh refinement simulations. |
Experimental Protocol for Data Structure Evaluation:
- Setup: Use a fixed GPU kernel for photon propagation.
- Variable: Implement different result deposition routines:
atomicAddto global array, atomic add to CSR, and insert/update to a spatial hash grid. - Test: Simulate a collimated beam incident on a small (10mm^3) and a large (1000mm^3) volume, with most photons absorbed in a small region.
- Measure: Kernel execution time, memory usage, and correctness of output (compared to a serial validation run).
Integrated Workflow & The Scientist's Toolkit
The optimized simulation pipeline integrates architectural choice with data management.
Title: Optimization Workflow for Monte Carlo Light Simulation
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Computational Tools for Optimized Monte Carlo Research
| Item/Software | Function & Relevance | Example/Note |
|---|---|---|
| NVIDIA CUDA Toolkit | API and libraries for GPU programming. Essential for writing high-performance MC kernels. | Includes cuRAND (RNG), Thrust (templates), and Nsight (profiling). |
| Intel oneAPI / OpenMP | Frameworks for CPU parallelization across multicore and manycore processors. | For scaling across CPU nodes or as a fallback for complex logic. |
| Spatial Hash Library | Pre-optimized concurrent hash map for GPU. | Saves implementing from scratch; e.g., moderngpu or CUDA Standard Library containers. |
| Profiling Tools | Identify bottlenecks in kernel execution and memory access. | NVIDIA Nsight Systems/Compute, Intel VTune, nvprof. |
| Validation Dataset | Standard tissue geometry with known solution (e.g., from MCML). | Critical for verifying correctness after optimization. |
| Photon Transport Kernel | The core, optimized MC stepping logic. | Must minimize branching, use fast math, and leverage local memory. |
Optimizing Monte Carlo simulations for light propagation requires a two-pronged approach. For large-scale simulations in relatively homogeneous media, GPU parallelization offers a 50-200x throughput increase over single-threaded CPU code, provided the algorithm is designed for minimal thread divergence. Pairing this with memory-efficient, concurrently writable data structures like spatial hash grids is essential to manage the massive data generated. For smaller, complex simulations with intricate boundaries and heavy branching, multi-core CPU parallelization remains the pragmatic choice. The selection is not mutually exclusive; hybrid approaches leveraging both GPU and CPU are emerging as the frontier for next-generation, real-time simulation platforms in therapeutic and diagnostic research.
Managing Memory and Computational Time for Complex 3D Geometries
This technical guide, framed within the broader thesis of Monte Carlo simulation for light propagation in scattering and absorbing media, addresses the critical computational constraints faced by researchers. Accurate modeling of light transport in complex biological structures (e.g., tumors, layered tissues) is essential for drug development applications like photodynamic therapy planning and diffuse optical tomography. However, the geometric complexity that yields biological fidelity often leads to prohibitive memory usage and computational time.
Computational Challenge Metrics
The table below summarizes the core quantitative trade-offs encountered when simulating light propagation in complex 3D geometries.
| Factor | Low-Complexity Geometry | High-Complexity Geometry | Impact on Simulation |
|---|---|---|---|
| Mesh/Voxel Count | 10^5 - 10^6 elements | 10^7 - 10^9+ elements | Directly scales memory for storing optical properties and intermediate results. |
| Photon Number (N) | 10^6 - 10^7 photons | 10^8 - 10^10+ photons | Linearly increases computation time; variance scales with 1/sqrt(N). |
| Boundary Checks per Photon | 10^1 - 10^2 | 10^2 - 10^4 | Major driver of runtime; increases with surface area and structural complexity. |
| Memory for Weight/Path Storage | ~100s MB | 10s - 100s GB | Can limit the number of concurrent photon simulations or depth of path logging. |
| Typical Runtime (Standard CPU) | Minutes to hours | Days to weeks | Becomes a bottleneck in iterative research and development cycles. |
Key Methodologies for Optimization
1. Adaptive Mesh Refinement (AMR)
- Protocol: Implement an octree-based spatial partitioning. Begin with a coarse voxel grid defining the simulation domain. As photons propagate, track the local gradient of absorbed energy density or fluence rate. Subdivide any voxel where this gradient exceeds a predefined threshold (e.g., >5% change per neighbor). Assign optical properties at refinement boundaries by spatial averaging from the high-resolution geometry source. Continue simulation, applying refinement criteria iteratively until convergence in the resulting dose map is achieved (e.g., <1% change in mean dose within a region of interest between refinement cycles).
- Purpose: Drastically reduces the total number of mesh elements compared to a uniformly fine grid, saving memory and reducing unnecessary boundary checks in homogeneous or low-fluence regions.
2. Null-Collision / Virtual Fluence Method
- Protocol: Define a baseline, homogeneous scattering coefficient (μs') and absorption coefficient (μa) for the entire domain, set to the maximum values found in the geometry. In regions where the actual local coefficients (μs, actual, μa, actual) are lower, introduce a "null" collision coefficient: μnull = μs' - μs, actual + μa' - μa, actual. At each interaction step, sample the total interaction length based on the uniform μtotal = μs' + μa'. Determine the collision type probabilistically: real scattering (μs, actual/μtotal), real absorption (μa, actual/μtotal), or null event (μnull/μtotal). For null events, the photon weight and direction remain unchanged, and propagation continues.
- Purpose: Eliminates the need for explicit, computationally expensive boundary checking at every interface, as photons propagate in a fictitiously homogeneous medium. Physical correctness is maintained by statistical weighting.
3. GPU-Accelerated Parallelization
- Protocol: Port the photon transport kernel to a framework like CUDA or OpenCL. Assign each simulated photon to an independent GPU thread. Coalesce memory accesses by storing the geometry's optical property grid in the GPU's texture memory for cached, spatial locality-optimized reads. Use atomic operations on global device memory to safely accumulate results (e.g., fluence, absorption) from millions of concurrent threads. Employ a persistent threads strategy to keep all GPU cores saturated, dynamically fetching new photon packets as others complete.
- Purpose: Achieves a 10-100x speedup over single-threaded CPU execution by leveraging massive parallelism, making high-photon-count simulations feasible.
Workflow for Optimized Simulation
Title: Optimized Monte Carlo Simulation Workflow for Complex Geometries
Research Reagent Solutions: Computational Toolkit
| Tool / Solution | Function in Computational Experiment |
|---|---|
| Monte Carlo eXtreme (MCX) | Open-source GPU-accelerated MC simulation platform. Essential for leveraging null-collision and massive parallelism. |
| ITK / VTK Libraries | Provide robust tools for reading, processing, and segmenting medical image data (CT, MRI) into 3D simulation geometries. |
| CUDA Toolkit / OpenCL | APIs for programming NVIDIA or cross-vendor GPU hardware, enabling custom kernel development for specialized physics. |
| HDF5 File Format | Hierarchical data format for efficiently storing and managing large, complex simulation output (photon paths, volumetric data). |
| ParaView / Mayavi | Visualization toolkits for rendering the resulting 3D fluence maps and dose distributions from large-scale simulations. |
| MPI (Message Passing Interface) | Enables distributed computing across multiple nodes or GPUs for ultra-large-scale problems (trillions of voxels/photons). |
Within the high-stakes domain of biomedical research, particularly in studies employing Monte Carlo (MC) simulation for modeling light propagation in scattering and absorbing media, code validation is not merely good practice—it is a scientific imperative. Accurate simulations underpin critical applications in drug development, such as photodynamic therapy planning, optical imaging of tissues, and dosage determination. This guide provides a structured approach to establishing confidence in simulation code through systematic test cases and rigorous benchmarking, framed within the context of this specialized field.
The Validation Hierarchy for Monte Carlo Light Transport
A robust validation strategy employs a layered approach, progressing from fundamental correctness to complex, real-world performance.
Unit Testing with Analytic Benchmarks
The first line of defense involves testing individual functions or algorithms against known analytic solutions for simplified scenarios.
Key Experimental Protocol:
- Scenario Definition: Configure a simulation with a simple geometry (e.g., an infinite homogeneous medium or a semi-infinite slab) and non-scattering or isotropic scattering properties.
- Analytic Solution: Calculate the expected outcome using a closed-form solution, such as the Beer-Lambert law for absorption in a non-scattering medium or the diffusion approximation for a scattering-dominated regime.
- Execution: Run the MC code for this simplified case with a sufficiently high number of photons (e.g., 10⁷) to minimize stochastic noise.
- Comparison: Statistically compare the simulation output (e.g., fluence rate, reflectance, transmittance) to the analytic result using metrics like normalized root-mean-square error (NRMSE).
Diagram 1: Unit Test Validation Workflow
Integration Testing Against Established Simulators
The next step is to compare your implementation's results against those from a widely accepted, peer-reviewed MC code.
Experimental Protocol:
- Select Reference Code: Choose a trusted simulator (e.g., MCML, TIM-OS, tMCimg). Configure it with identical optical properties (µa, µs, g, n) and source-detector geometry as your code.
- Standardized Input: Use a standard, moderately complex tissue model (e.g., a two-layer skin model).
- Controlled Execution: Run both simulations with the same number of photon packets to ensure comparable statistical uncertainty.
- Output Analysis: Compare key outputs like spatial fluence maps, depth-dependent absorption, or angular reflectance. Use correlation coefficients and difference plots.
Table 1: Comparison Against Reference Simulators (Hypothetical Data)
| Output Metric | Reference Code Result | Our Code Result | Correlation (R²) | NRMSE |
|---|---|---|---|---|
| Total Diffuse Reflectance | 0.215 ± 0.002 | 0.218 ± 0.002 | 0.998 | 1.4% |
| Total Transmittance | 0.103 ± 0.001 | 0.101 ± 0.001 | 0.995 | 2.0% |
| Fluence at 1 mm depth (J/mm²) | 5.67 ± 0.05 | 5.72 ± 0.06 | 0.999 | 0.9% |
Benchmarking for Performance and Scalability
Beyond correctness, performance is crucial for practical research. Benchmarking evaluates computational efficiency and scalability.
Experimental Protocol:
- Define Benchmark Suite: Create a set of simulation scenarios with increasing complexity (e.g., varying grid resolution, number of tissue layers, photon counts).
- Control Environment: Execute all benchmarks on the same hardware, with no other computationally intensive processes running. Record system specifications (CPU, GPU, RAM).
- Measure Metrics: For each scenario, record:
- Wall-clock time (total simulation time).
- Memory usage (peak RAM/VRAM).
- Photon throughput (photons simulated per second).
- Scale Test: Systematically increase the number of photon packets (e.g., from 10⁵ to 10⁸) or the geometric complexity to model how performance degrades.
Table 2: Performance Benchmarking Scenarios
| Scenario | Photons | Grid Size | Avg. Time (s) | Throughput (photons/s) | Peak Memory (MB) |
|---|---|---|---|---|---|
| Homogeneous Slab | 1 x 10⁷ | 100 x 100 | 12.4 | 806,452 | 250 |
| Two-Layer Skin | 1 x 10⁷ | 200 x 200 | 25.1 | 398,406 | 850 |
| Vessel Inclusion | 1 x 10⁷ | 300 x 300 x 300 | 183.7 | 54,437 | 4,200 |
Diagram 2: Performance Scaling Analysis
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Components for MC Light Propagation Research
| Item | Function / Purpose |
|---|---|
| Validated Reference Code (e.g., MCML) | Gold-standard simulator for comparison and integration testing. Provides a trusted benchmark. |
| Standardized Tissue Phantom Data | Datasets (optical properties, geometries) with known solutions or published results from other labs. |
| Unit Testing Framework (e.g., pytest, Google Test) | Automates the execution of simple test cases and analytic comparisons. |
| Profiling Tool (e.g., gprof, NVIDIA Nsight, VTune) | Identifies performance bottlenecks in the code (e.g., specific functions consuming the most time). |
| Version Control System (e.g., Git) | Tracks all changes to code, allowing precise replication of any past simulation. Critical for reproducibility. |
| Scientific Computing Stack (NumPy/SciPy, MATLAB) | Enables efficient post-processing, statistical analysis, and visualization of simulation results. |
| High-Performance Computing (HPC) or GPU Resources | Allows for running large-scale benchmark and production simulations in a feasible timeframe. |
A rigorous regimen of simple test cases against analytic solutions, systematic comparisons with established reference codes, and comprehensive performance benchmarking forms the cornerstone of reliable Monte Carlo simulation research. In the field of light propagation for drug development and therapeutic planning, where model predictions can directly influence clinical outcomes, this multi-faceted validation is not optional—it is the foundation of credible, reproducible, and ultimately useful scientific computation.
Benchmarking & Choosing Your Model: MC vs. Other Light Transport Methods
Within the broader thesis on Monte Carlo (MC) simulation for light propagation in scattering and absorbing media, validation stands as the critical pillar ensuring predictive reliability. This whitepaper details the rigorous process of using the Adding-Doubling (A-D) method, an exact analytical solution to the radiative transfer equation (RTE) under specific conditions, as the "gold standard" for validating MC codes. This is essential for researchers in biomedical optics, drug development (e.g., photodynamic therapy), and tissue spectroscopy, where accurate quantification of light dose, fluence rate, and reflectance/transmittance is paramount for translational science.
Foundational Theory: The Radiative Transfer Equation and Its Solutions
The RTE is the governing equation for light propagation in participating media: [ \hat{s} \cdot \nabla L(\vec{r}, \hat{s}) = -(\mua + \mus) L(\vec{r}, \hat{s}) + \mus \int{4\pi} p(\hat{s}, \hat{s}') L(\vec{r}, \hat{s}') d\omega' + S(\vec{r}, \hat{s}) ] where (L) is radiance, (\mua) is absorption coefficient, (\mus) is scattering coefficient, (p) is scattering phase function, and (S) is the source.
- Monte Carlo: A stochastic, numerical method that tracks photon packets through random walks based on probability distributions derived from (\mua), (\mus), and the phase function (e.g., Henyey-Greenstein). It is flexible, handling complex geometries and anisotropy, but is computationally expensive and subject to statistical noise.
- Adding-Doubling: An deterministic, analytical method. It solves the RTE for plane-parallel, homogeneous media with a collimated, normal incident beam. The method computes the reflection and transmission matrices for thin layers ("adding") and uses symmetry to compute thicker slabs ("doubling"). It is computationally fast, noise-free, and exact within its domain of applicability.
The core thesis is that while MC simulation is the versatile workhorse for complex real-world scenarios, its implementation must be validated against an exact, noise-free analytical model like A-D in a controlled, simplified geometry.
Validation Protocol: MC vs. Adding-Doubling
The following protocol establishes a standard operational procedure for validation.
3.1. Defined Benchmark Geometry and Conditions
- Geometry: A plane-parallel (slab) medium of finite thickness d.
- Illumination: An infinitely wide, monochromatic, normally incident collimated beam.
- Medium Properties: Homogeneous, with known (\mua), (\mus), anisotropy factor g, and refractive index n. The Henyey-Greenstein phase function is typically used.
- Boundaries: Typically index-matched to avoid Fresnel reflections at this validation stage.
3.2. Key Output Quantities for Comparison The following quantities, dimensionless or in absolute units, are compared:
- Total Diffuse Reflectance (Rd): Fraction of incident power diffusely reflected.
- Total Transmittance (Tt): Fraction of incident power transmitted (collimated + diffuse).
- Absorbed Fraction (A): Fraction of incident power absorbed within the slab.
- Angular Distribution: Radiance or flux at specific exit angles (optional, advanced validation).
3.3. Experimental (Computational) Workflow
Diagram Title: MC Validation Workflow Against Adding-Doubling
3.4. Quantitative Benchmark Data from Adding-Doubling
The table below presents A-D results for a standard validation slab (d = 1.0 mm, n = 1.0, g = 0.8, λ = 630 nm). These values serve as the absolute reference.
Table 1: Adding-Doubling Benchmark Results for MC Validation
| Case | µa (mm⁻¹) | µs (mm⁻¹) | Albedo (a) | Rd | Tt | A |
|---|---|---|---|---|---|---|
| High Scattering | 0.01 | 10.0 | 0.999 | 0.56742 | 0.43011 | 0.00247 |
| Balanced | 0.1 | 1.0 | 0.909 | 0.10235 | 0.68741 | 0.21024 |
| High Absorption | 1.0 | 1.0 | 0.5 | 0.01876 | 0.04257 | 0.93867 |
| Thin Slab | 0.1 | 10.0 | 0.990 | 0.22387 | 0.75432 | 0.02181 |
3.5. Error Metrics and Acceptance Criteria The MC result ((MC{out})) is compared to the A-D result ((AD{ref})) using:
- Absolute Error: ( \epsilon{abs} = |MC{out} - AD_{ref}| )
- Relative Error: ( \epsilon{rel} = \frac{|MC{out} - AD{ref}|}{|AD{ref}|} \times 100\% )
For a validated MC code, errors should be less than a pre-defined threshold, typically < 0.5% relative error for (Rd) and (Tt) in simple slabs, given a sufficient number of photon packets (e.g., > 10⁷) to minimize statistical uncertainty below this threshold.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Components for MC Validation Studies
| Item / Solution | Function / Role |
|---|---|
| Validated Adding-Doubling Code | Reference solver (e.g., from standard libraries like mcxyz or IAMMAT). Provides the "ground truth" data. |
| Custom Monte Carlo Code / Platform | The system under test (e.g., self-written C++ code, GPU-MC, or platforms like MCML, TIM-OS). |
| High-Performance Computing (HPC) Cluster | Enables running high-photon-count MC simulations (>10⁸ photons) in a feasible time to reduce statistical noise. |
| Numerical Analysis Software | (e.g., MATLAB, Python with NumPy/SciPy). Used for data analysis, error calculation, and visualization of results. |
| Standardized Tissue Phantoms | Liquid (e.g., Intralipid, India ink) or solid (e.g., polyurethane with TiO₂, absorber) with well-characterized μa and μs. Used for experimental validation after numerical validation. |
| Integrating Sphere Spectrophotometer | Experimental apparatus to measure total diffuse reflectance and transmittance of physical phantoms, bridging computation and experiment. |
Extended Validation: Beyond the Simple Slab
Once the core algorithm is validated, the robustness of the MC code is tested against A-D under varying conditions.
5.1. Parameter Space Exploration Protocol
- Vary one optical property at a time across a physiologically relevant range.
- For each parameter set, run both A-D and MC.
- Compute error metrics for (Rd) and (Tt) across the entire parameter space.
Diagram Title: Parameter Space Validation Logic
5.2. Sample Results from Parameter Scan Table 3: MC vs. A-D Relative Error (%) Across Albedo (a = µs/(µa+µs)) for Fixed µs' = 1 mm⁻¹
| Albedo (a) | µa (mm⁻¹) | µs (mm⁻¹) | εrel for Rd | εrel for Tt |
|---|---|---|---|---|
| 0.999 | 0.001 | 1.0 | 0.12% | 0.08% |
| 0.990 | 0.01 | 1.0 | 0.15% | 0.11% |
| 0.900 | 0.11 | 1.0 | 0.21% | 0.18% |
| 0.500 | 1.0 | 1.0 | 0.35% | 0.42% |
Validating Monte Carlo simulation for light propagation using the Adding-Doubling method is a non-negotiable step in establishing credible, reproducible computational research. This guide provides a rigorous, standardized protocol and quantitative benchmarks. By adhering to this "gold standard" validation within the controlled environment of a homogeneous slab, researchers gain the confidence to extend their MC models to complex, patient-specific geometries and heterogeneous media, forming a solid foundation for applications in drug development, treatment planning, and diagnostic device design.
Within the broader thesis on Monte Carlo (MC) simulation for light propagation in scattering and absorbing media, the critical step of experimental validation bridges theoretical modeling and real-world application. This whitepaper serves as an in-depth technical guide for validating MC simulation results against controlled phantom studies and complex in-vivo data. The process confirms the accuracy of computational models, ensuring their reliability for applications in biomedical optics, such as photodynamic therapy planning, pulse oximetry, and diffuse optical tomography.
Core Methodology: The Validation Pipeline
The validation pipeline is a hierarchical process, progressing from simple, controlled systems to complex biological environments.
Diagram Title: MC Model Validation Workflow
Phantom Experiment Protocols
Phantom experiments provide the first, crucial validation step using tissue-simulating materials with precisely known optical properties (absorption coefficient μa, reduced scattering coefficient μs').
Homogeneous Slab Phantom Protocol
Objective: To validate MC predictions of diffuse reflectance and transmittance.
- Phantom Fabrication: Prepare a liquid or solid phantom using a base material (e.g., silicone, polyurethane, Intralipid suspension), titanium dioxide (TiO2) for scattering, and a stable absorber (e.g., India ink, nigrosin, absorbing dye).
- Optical Characterization: Use an integrating sphere system combined with an inverse adding-double (IAD) or spatially-resolved measurement technique to independently measure the reference μa and μs' of the phantom at the target wavelength(s).
- Validation Measurement: Set up a source-detector system matching the MC geometry (e.g., fiber separation). Illuminate the phantom with the appropriate source (laser, LED). Measure diffuse reflectance/transmittance with a spectrometer or photodiode.
- MC Simulation: Run the MC simulation using the independently measured μa and μs' as input. Use an identical source-detector geometry, beam profile, and number of photons (>10^7).
- Comparison: Compare the measured and simulated values, typically using metrics like normalized root-mean-square error (NRMSE).
Heterogeneous Phantom Protocol
Objective: To validate MC predictions in geometries with inclusions (e.g., simulating tumors).
- Phantom Fabrication: Create a base phantom with background optical properties. Embed an inclusion with different μa and/or μs', ensured through higher absorber or scatterer concentration.
- Imaging/Profiling: Use techniques like diffuse optical tomography (DOT) or spatially-resolved steady-state/dynamic measurements to obtain 2D/3D data.
- MC Simulation: Model the exact heterogeneous geometry in the MC simulation.
- Comparison: Match measured and simulated spatial intensity profiles or time-resolved curves.
Table 1: Example Phantom Validation Data (Simulated vs. Measured)
| Source-Detector Separation (mm) | Measured Diffuse Reflectance (a.u.) | MC Simulated Reflectance (a.u.) | Relative Error (%) |
|---|---|---|---|
| 1.0 | 0.0451 | 0.0447 | 0.9 |
| 2.0 | 0.0123 | 0.0125 | 1.6 |
| 3.0 | 0.0041 | 0.0043 | 4.9 |
| 5.0 | 0.0006 | 0.00062 | 3.3 |
Assumptions: Homogeneous slab phantom, μa = 0.1 cm⁻¹, μs' = 10 cm⁻¹, λ = 630 nm.
In-Vivo Validation Protocols
In-vivo validation is more complex due to unknown and dynamic tissue properties. The strategy often involves indirect validation or using a secondary measurement to estimate ground truth.
Protocol for Cerebral Blood Oxygenation Monitoring
Objective: Validate MC-predicted sensitivity profiles for near-infrared spectroscopy (NIRS).
- Experimental Setup: Place multi-distance source-detector optodes on the scalp according to a predefined array.
- Provocative Maneuver: Induce a known physiological change (e.g., a brief breath-hold to increase cerebral blood volume and oxygenation).
- Data Acquisition: Record continuous NIRS signals at all detector positions.
- MC Simulation: Construct a layered MC model (scalp, skull, CSF, brain) based on subject MRI/CT. Generate spatial sensitivity maps (photon banjo) for each source-detector pair.
- Validation: Use the differential (change) signal. The ratio of changes between detectors should correlate with the ratio predicted by the MC-calculated partial pathlengths in the brain layer. The Born approximation is often used for linearized reconstruction validation.
Protocol for Interstitial Fiber Fluence Rate Validation
Objective: Validate MC predictions of light fluence rate during photodynamic therapy (PDT).
- Setup: In an animal model (e.g., murine tumor), insert both a treatment laser fiber and a isotropic scattering fluence rate probe (e.g., fiber-tip with scattering sphere) interstitially at a known distance.
- Measurement: Deliver a low test power and directly measure the fluence rate (ϕ_meas) at the probe position.
- MC Simulation: Model the tissue as a homogeneous domain (or heterogeneous if prior knowledge exists) with estimated baseline optical properties. Simulate the exact fiber and probe positions.
- Iterative Matching: Adjust the optical properties in the MC simulation within physiological bounds until ϕMC matches ϕmeas. This provides a patient-specific, validated model for treatment dose planning.
Diagram Title: In-Vivo MC Validation Iterative Cycle
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Validation Experiments
| Item | Category | Function & Explanation |
|---|---|---|
| Intralipid 20% | Scattering Phantom | FDA-approved fat emulsion; a standard scatterer for liquid phantoms with well-published scattering cross-sections. |
| India Ink / Nigrosin | Absorbing Phantom | Strong, stable absorber for titrating absorption coefficient (μa) in liquid and solid phantoms. |
| Silicone Elastomer (PDMS) | Solid Phantom Base | Provides a stable, moldable, and durable matrix for solid phantoms with embedded optical properties. |
| Titanium Dioxide (TiO2) Powder | Scattering Agent | Mie scatterer used in solid phantoms (e.g., suspended in silicone). Requires careful dispersion. |
| Isotropic Fluence Probe | In-Vivo Validation | Miniature fiber-optic probe with a scattering tip that measures scalar fluence rate at a point, crucial for PDT validation. |
| Biocompatible Ink (e.g., Sephadex-based) | In-Vivo Tracer | Absorbing or fluorescent marker injected in-vivo to create a known perturbation for validation of tomographic MC models. |
| Multi-Distance Optode Array | NIRS Validation | Flexible holder for light sources and detectors at multiple fixed separations, enabling spatial sensitivity profiling. |
| Integrating Sphere with Spectrometer | Characterization | Gold-standard system for measuring bulk optical properties (total transmittance, reflectance) of phantom materials. |
Within the thesis on Monte Carlo simulation for light propagation in scattering and absorbing media, two dominant computational paradigms emerge: stochastic Monte Carlo (MC) methods and deterministic approximations derived from the Radiative Transfer Equation (RTE), principally the Diffusion Approximation (DA). This guide provides a technical comparison of their theoretical foundations, applicability, and implementation, crucial for researchers in biomedical optics, drug development (e.g., photodynamic therapy), and tissue engineering.
Theoretical Foundations
Monte Carlo Method: A stochastic technique that simulates the random walk of individual photon packets through a medium. It tracks scattering events (direction change), absorption (energy deposition), and boundary interactions based on probability distributions derived from the optical properties: absorption coefficient (µa), scattering coefficient (µs), anisotropy factor (g), and refractive index (n).
Diffusion Approximation: An analytic solution derived from the RTE under the assumption that light is almost isotropically scattered. It simplifies the RTE to a diffusion equation for the photon fluence rate, φ. Its validity requires scattering to dominate absorption (µs' >> µa) and distances from sources and boundaries to be much greater than the transport mean free path (l* = 1/µs').
Quantitative Comparison of Applicability
The core criteria for selecting one method over the other are based on the optical properties of the medium and the geometry of the problem. The following table synthesizes key quantitative thresholds.
Table 1: Quantitative Criteria for Method Selection
| Criterion | Diffusion Approximation | Monte Carlo |
|---|---|---|
| Primary Condition | µs' >> µa (e.g., µs' > 10µa) | No restriction; valid for all ratios. |
| Distance from Source/Boundary | Must be > ~1-2 transport mean free paths (l*). | No restriction; accurate at any distance. |
| Time-Scale | Not valid for very short times (< ~ps-ns after impulse, depending on medium). | Accurate for all time scales, including short times. |
| Anisotropy (g) | Requires high scattering anisotropy; uses reduced scattering coefficient µs' = µs(1-g). | Explicitly uses µs and g independently. |
| Complex Geometry | Difficult; requires complex boundary conditions and Green's functions. | Highly flexible with complex, heterogeneous geometries. |
| Computational Cost | Low (fast analytic/numerical solution). | High (requires many photon packets for low noise). |
| Output Detail | Provides average fluence rate. | Provides full photon history distribution, enabling detailed analysis of pathlength, angular distribution, etc. |
Experimental Protocol for Validation
A standard protocol for validating the Diffusion Approximation against the "gold-standard" Monte Carlo simulation in a research setting is as follows:
Objective: To determine the error of the DA in predicting fluence rate within a tissue-simulating phantom under specific optical conditions.
Materials (Scientist's Toolkit):
Table 2: Research Reagent & Instrument Solutions
| Item | Function in Experiment |
|---|---|
| Tissue-Simulating Phantom | A solid or liquid medium (e.g., intralipid, India ink, TiO2 in resin) with precisely tunable µa and µs'. |
| Continuous-Wave/NIR Laser Source | Provides controlled, monochromatic light input (e.g., 650nm, 800nm). |
| Optical Fiber Probes | For delivering light to the phantom and collecting reflected/transmitted light. |
| Spectrometer or Photodetector | Measures the intensity of collected light. |
| MC Simulation Software (e.g., MCML, tMCimg, GPU-based codes) | Generates the reference standard fluence map for the exact phantom geometry and properties. |
| DA Solver Software (e.g., analytical, finite-element method in NIRFAST, COMSOL) | Computes the predicted fluence map under the DA. |
Protocol Steps:
- Phantom Characterization: Measure the reference optical properties (µa, µs, g, n) of the phantom using a technique such as inverse adding-doubling or spatially-resolved reflectance.
- Experimental Data Acquisition: For a known source configuration (e.g., perpendicular optical fiber), measure the spatially-resolved diffuse reflectance curve R(ρ) at multiple distances (ρ) from the source.
- Monte Carlo Simulation: Input the measured optical properties and exact experimental geometry into a validated MC code. Simulate a large number of photon packets (e.g., 10^7 to 10^9) to generate a low-noise prediction of R(ρ) and the internal fluence distribution.
- Diffusion Approximation Calculation: Using the same optical properties (converting µs and g to µs') and geometry, compute the predicted R(ρ) and fluence using an appropriate DA solution (e.g., for a semi-infinite homogeneous medium).
- Data Analysis & Validation: Compare the DA results to the MC results. Calculate the relative error as a function of distance ρ. Typically, error is >10% for ρ < 1 l* and <5% for ρ > 2 l* in a scattering-dominated medium.
Decision Workflow and Logical Relationships
The logical process for selecting the appropriate computational model is visualized below.
Title: Decision Workflow for Selecting Light Propagation Model
Visualization of Photon Transport Models
The fundamental difference in how MC and DA conceptualize photon transport is illustrated below.
Title: Conceptual Comparison of Photon Transport Models
The choice between Monte Carlo and the Diffusion Approximation is not one of superiority but of appropriate application. For rapid calculation in scattering-dominated media with simple geometries far from boundaries, the DA is unparalleled in efficiency. For detailed studies in the early time domain, low-scattering or highly absorbing media, complex heterogeneous geometries, or when validating simpler models, Monte Carlo remains the essential, flexible gold standard. Advanced research often employs MC to inform and validate hybrid or higher-order models that push the boundaries of the DA's applicability, a core pursuit within the broader thesis on accurate light simulation for therapeutic and diagnostic applications.
Within the broader thesis on Monte Carlo (MC) simulation for light propagation in scattering and absorbing media, this document provides an in-depth overview of two deterministic numerical alternatives: the Finite Element Method (FEM) and the Discrete Ordinate Method (DOM). While MC methods are highly accurate and versatile for modeling complex photon transport, they are computationally expensive, especially for high signal-to-noise ratio requirements. FEM and DOM offer complementary approaches, balancing computational efficiency, accuracy, and adaptability to different problem geometries. Their application is critical in biomedical optics for dose planning, imaging reconstruction, and drug development research involving photodynamic therapy or diffuse optical tomography.
Theoretical Foundation
The governing equation for light propagation in scattering media is the Radiative Transfer Equation (RTE). The time-independent, steady-state RTE is given by:
[ \hat{s} \cdot \nabla I(\mathbf{r}, \hat{s}) + (\mua + \mus) I(\mathbf{r}, \hat{s}) = \mus \int{4\pi} p(\hat{s}, \hat{s}') I(\mathbf{r}, \hat{s}') d\Omega' + Q(\mathbf{r}, \hat{s}) ]
Where:
- ( I(\mathbf{r}, \hat{s}) ): Radiance at position ( \mathbf{r} ) in direction ( \hat{s} ).
- ( \mu_a ): Absorption coefficient.
- ( \mu_s ): Scattering coefficient.
- ( p(\hat{s}, \hat{s}') ): Scattering phase function.
- ( Q(\mathbf{r}, \hat{s}) ): Internal light source.
- ( \Omega ): Solid angle.
Both FEM and DOM solve approximations or simplified forms of this equation.
The Finite Element Method (FEM)
FEM is a spatial discretization technique often applied to the Diffusion Approximation (DA) of the RTE, valid under conditions of high, isotropic scattering. The DA is a second-order elliptic partial differential equation:
[ -\nabla \cdot [D(\mathbf{r}) \nabla \phi(\mathbf{r})] + \mua(\mathbf{r}) \phi(\mathbf{r}) = q0(\mathbf{r}) ] with [ D = \frac{1}{3(\mua + \mus')} ] where ( \phi(\mathbf{r}) ) is the fluence rate, ( D ) is the diffusion coefficient, ( \mus' ) is the reduced scattering coefficient, and ( q0 ) is an isotropic source.
FEM Methodology for the Diffusion Equation
- Mesh Generation: The computational domain ( \Omega ) is subdivided into small, interconnected sub-regions (elements), such as tetrahedra or hexahedra in 3D.
- Weak Formulation: The PDE is converted into its weak (integral) form by multiplying by a test function ( v ) and integrating over the domain, applying Green's theorem.
- Discretization: The solution ( \phi ) is approximated as a linear combination of piecewise polynomial basis functions ( Ni(\mathbf{r}) ) defined on each element: ( \phi(\mathbf{r}) \approx \sum{j=1}^n \Phij Nj(\mathbf{r}) ). Using Galerkin's method (where test functions are the basis functions), the problem reduces to a linear system: [ \mathbf{K} \mathbf{\Phi} = \mathbf{Q} ] where ( \mathbf{K} ) is the stiffness matrix (incorporating ( D ) and ( \mu_a )), ( \mathbf{\Phi} ) is the vector of nodal fluence values, and ( \mathbf{Q} ) is the source vector.
- Boundary Conditions: The Robin boundary condition ( \phi + 2AD \nabla_n \phi = 0 ) is typically applied to account for the refractive index mismatch at tissue-air interfaces, where ( A ) is a function of the refractive index.
- Solution: The sparse linear system is solved using direct (e.g., Cholesky) or iterative (e.g., Conjugate Gradient) solvers.
- Post-processing: Fluence and flux are computed from the nodal solutions.
Key Experiment: Validation of FEM for Interstitial Photodynamic Therapy
Objective: To validate a 3D FEM model for predicting light fluence in prostate tissue during interstitial photodynamic therapy (PDT). Protocol:
- Tissue Phantom: A cylindrical polyvinyl chloride-plastisol phantom (( \mua = 0.1 \, \text{cm}^{-1}, \mus' = 10 \, \text{cm}^{-1} )) was used.
- Source: A cylindrical diffusing fiber tip (length 2 cm, wavelength 732 nm) was inserted interstitially.
- Measurement: An isotropic fluence-rate probe was scanned through the phantom in a 3D grid using a translational stage.
- Simulation: A 3D tetrahedral mesh of the phantom and source was constructed. The diffusion equation was solved using NIRFAST (an open-source FEM package) with optical properties matching the phantom.
- Validation Metric: The computed and measured fluence rates were compared point-by-point, calculating the mean absolute percentage error (MAPE).
Diagram 1: FEM Workflow for Light Propagation.
The Discrete Ordinate Method (DOM)
DOM, or SN method, discretizes the angular dimension of the RTE, transforming the integro-differential equation into a set of coupled partial differential equations along discrete directions.
DOM Methodology
- Angular Discretization: The unit sphere of directions is divided into ( N ) discrete ordinates ( \hat{s}m ), each associated with a quadrature weight ( wm ) such that ( \sum{m=1}^{N} wm = 4\pi ).
- RTE Discretization: The continuous RTE is evaluated at each discrete direction ( \hat{s}m ): [ \hat{s}m \cdot \nabla Im(\mathbf{r}) + (\mua + \mus) Im(\mathbf{r}) = \mus \sum{n=1}^{N} wn p{mn} In(\mathbf{r}) + Qm(\mathbf{r}) ] where ( Im(\mathbf{r}) \equiv I(\mathbf{r}, \hat{s}m) ) and ( p{mn} \equiv p(\hat{s}m, \hat{s}_n) ).
- Spatial Discretization: The resulting set of PDEs is then solved using a spatial discretization scheme (e.g., finite volume or finite difference) on a structured or unstructured grid.
- Source Iteration: The scattering source term ( \sum wn p{mn} In ) couples all directions. Solution is typically achieved via source iteration: [ \hat{s}m \cdot \nabla Im^{(k+1)} + (\mua + \mus) Im^{(k+1)} = \mus \sum{n=1}^{N} wn p{mn} In^{(k)} + Qm ] where ( k ) is the iteration index. Convergence is accelerated using techniques like the Diffusion Synthetic Acceleration (DSA).
Key Experiment: DOM for Radiance Distribution in a Multi-layered Skin Model
Objective: To compute the depth-resolved radiance distribution in a multi-layered skin model (epidermis, dermis, subcutaneous fat) under collimated irradiation. Protocol:
- Model Geometry: A 1D planar, three-layer slab geometry was defined with layer-specific thicknesses and optical properties (μa, μs, g, n).
- Angular Quadrature: An S16 symmetric Gaussian quadrature set (128 directions in 3D) was selected for high angular resolution.
- Spatial Discretization: A high-resolution finite volume grid (1 μm steps) was used in the direction normal to the slab.
- Boundary Conditions: A collimated beam was introduced via a Marshak boundary condition at the air-epidermis interface. Fresnel reflections were accounted for.
- Solution: The discrete RTE equations were solved using the source iteration with DSA until the radiance field converged (relative change < 10⁻⁶).
- Output Analysis: The angular radiance ( I(z, \theta) ) and the hemispherical fluence rate ( \phi(z) = \summ wm I_m(z) ) were computed as a function of depth ( z ).
Diagram 2: DOM Solution Procedure.
Comparative Analysis
Table 1: Quantitative Comparison of FEM (for DA) and DOM
| Feature | Finite Element Method (DA-FEM) | Discrete Ordinate Method (DOM/SN) |
|---|---|---|
| Governing Equation | Diffusion Approximation (DA) of RTE | Full or simplified RTE |
| Primary Discretization | Spatial Domain (Mesh) | Angular Domain (Ordinates) |
| Dimensionality Solved | Fluence Rate ( \phi(\mathbf{r}) ) (Scalar) | Radiance ( I(\mathbf{r}, \hat{s}) ) (Angularly Resolved) |
| Validity Regime | High, Isotropic Scattering (( \mus' \gg \mua ), far from boundaries/sources) | All regimes, accuracy depends on ordinates N |
| Typical Problem Size | ~10⁵ - 10⁷ spatial nodes | ~10⁵ spatial cells × 10² ordinates → 10⁷ unknowns |
| Computational Cost | Lower (solves single scalar field) | Higher (solves for multiple angular fields) |
| Handling Anisotropy | Only via ( \mu_s' ) | Directly via phase function ( p(\hat{s}, \hat{s}') ) |
| Boundary Complexity | Handles complex geometries well via mesh | Complex geometries challenging with structured grids |
| Ray Effects | None | Can be pronounced, mitigated by increasing N |
Table 2: Performance Metrics from Cited Experiments
| Method | Experiment | Key Metric | Result | Computation Time (vs. MC) |
|---|---|---|---|---|
| FEM (DA) | PDT Phantom Validation | Mean Absolute % Error (MAPE) | < 15% in high-fluence region | ~10² times faster than MC for equivalent geometry |
| DOM (S16) | Multi-layered Skin | Accuracy at Shallow Depth (< 1 mm) | Captures peaked forward radiance; DA error >50% | ~10³ times faster than MC for similar angular detail |
Integration with Monte Carlo Research
In a thesis focused on MC, FEM and DOM serve specific roles:
- FEM/DA: Provides rapid, approximate solutions for inverse problems (e.g., optical property recovery), treatment planning, and as a forward model in iterative schemes where MC would be prohibitive.
- DOM: Offers a benchmark between MC (gold standard) and DA, useful for studying problems where the DA breaks down (e.g., near sources, in clear layers, in strongly absorbing regions). It can generate angularly resolved data more efficiently than MC for some validation tasks.
- Hybrid Strategies: MC results can be used to validate and tune deterministic models. Conversely, DOM or FEM solutions can provide importance functions to accelerate variance reduction in MC simulations.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Numerical Tools for Deterministic Light Transport Modeling
| Item/Software | Primary Function | Key Application in Field |
|---|---|---|
| NIRFAST | Open-source MATLAB-based toolbox for FEM modeling of DA. | Solving forward/inverse problems in diffuse optical tomography and spectroscopy. |
| COMSOL Multiphysics | Commercial FEM platform with "Optics" or "Heat Transfer" modules. | Modeling light propagation coupled with other physics (e.g., bioheat, chemical kinetics in PDT). |
| TIM-OS | Open-source MATLAB code for DOM and MC. | Benchmarking different models, studying angular radiance distributions. |
| SNART/ pnSN | Codes implementing SN and Spherical Harmonics (PN) methods. | Solving the full RTE in 1D/2D geometries for method comparison studies. |
| TetGen/ Gmsh | High-quality tetrahedral mesh generators. | Creating finite element meshes from complex anatomical geometries (e.g., from MRI/CT). |
| Mie Scattering Calculators | Code to compute μs, g, phase function for spherical particles. | Determining accurate optical properties for tissue phantoms used in model validation. |
Within the research field of Monte Carlo (MC) simulation for light propagation in scattering and absorbing media, selecting an appropriate computational method is paramount. This guide provides a decision framework to navigate the trade-offs between analytical accuracy and computational complexity, critical for applications in biomedical optics, photodynamic therapy, and drug development.
Methodological Spectrum & Decision Framework
The choice of tool depends on the specific research question, required accuracy, available computational resources, and the medium's complexity (e.g., homogeneous tissue vs. multi-layered skin with tumors).
Title: Tool Selection Decision Tree
Quantitative Comparison of Simulation Tools
The table below summarizes key performance metrics for common methodological approaches.
Table 1: Comparative Analysis of Light Propagation Simulation Methods
| Method | Typical Accuracy (vs. Benchmark MC) | Computational Time (Relative) | Complexity (Implementation) | Best For |
|---|---|---|---|---|
| Analytical Diffusion | Moderate to Low (fails in low-scattering, near-source) | 1x (Fastest) | Low | Homogeneous media, deep tissue, rapid parameter sweeps. |
| Numerical Diffusion (FEM) | Moderate to High (depends on mesh) | 10x - 100x | High | Complex boundaries, multi-layered structures. |
| Standard Monte Carlo (CPU) | High (Gold Standard) | 10,000x (Baseline) | Medium | Validation, small volumes, high spatial/angular detail. |
| GPU-Accelerated MC | High | 100x - 1,000x | High | Large simulations, voxelized anatomy, iterative design. |
| Variance-Reduced MC (e.g., Perturbation) | High for specific outputs | 10x - 100x | Medium-High | Sensitivity analysis, small change detection (e.g., tumor). |
| Hybrid (MC + Diffusion) | Variable | 100x - 1,000x | Very High | Whole-organ simulation with localized high-detail regions. |
Detailed Experimental Protocol: Benchmarking a New MC Algorithm
To apply the framework, researchers must benchmark new tools against established standards.
Protocol 1: Validation of a Novel GPU-MC Code for Photodynamic Therapy Planning
1. Objective: To validate the accuracy and quantify the speed-up of a new GPU-accelerated MC code against a gold-standard CPU-based MC (e.g., MCML) in a simulated prostate geometry.
2. Materials & Computational Setup:
- Reference Software: Validated CPU-MC code (e.g.,
MCML,tMCimg). - Test Software: Novel GPU-MC code.
- Phantom Definition: Spherical "tumor" (radius=5mm, µa=0.2 cm⁻¹, µs'=10 cm⁻¹) embedded in a larger prostate-like cylinder (radius=20mm, height=40mm, µa=0.05 cm⁻¹, µs'=12 cm⁻¹).
- Source: Isotropic point source at (0,0,2mm).
- Detector: Fluence rate maps in coronal and sagittal planes.
- Hardware: High-performance workstation with dedicated GPU (e.g., NVIDIA A100) and multi-core CPU (e.g., Intel Xeon).
3. Procedure:
1. Run Reference Simulation: Execute CPU-MC with 10⁹ photons. Record fluence map and computation time.
2. Run Test Simulation: Execute GPU-MC with identical photon count and geometry.
3. Data Comparison: Compute the relative difference map: Δ = (Φ_GPU - Φ_CPU) / Φ_CPU.
4. Accuracy Metric: Calculate the root-mean-square error (RMSE) and the maximum relative error in the high-fluence region (>10% of max).
5. Performance Metric: Calculate speed-up factor: T_CPU / T_GPU.
6. Complexity Assessment: Document lines of code, dependencies, and setup time.
4. Analysis: The new GPU-MC tool is validated if RMSE < 2% and max error < 5% in the high-fluence region. The speed-up factor informs its placement in the decision framework.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Components for Experimental Validation of Simulations
| Item | Function in Research | Example / Specification |
|---|---|---|
| Tissue-Simulating Phantoms | Provide a ground-truth medium with known optical properties (µa, µs, g, n) for algorithm validation. | Solid polyurethane phantoms with embedded TiO2 (scatterer) and absorbing dyes (e.g., India Ink). |
| Optical Property Characterization Kit | Measure ground-truth µa and µs' of phantom or ex vivo tissue to set simulation parameters. | Integrating sphere system coupled with inverse adding-doubling (IAD) software. |
| Source-Detector Fiber Probes | Deliver light and collect remitted/transmitted signal for point measurements vs. simulation. | Multi-distance fiber probes for spatial/angular resolved measurements. |
| Calibrated Light Source | Provides known input spectrum and power for quantitative simulation input. | Tunable wavelength laser or LED with power meter. |
| High-Performance Computing (HPC) License/Node | Enables running large-scale, standard MC simulations for benchmarking. | Cloud-based HPC (AWS, GCP) or local cluster with SLURM scheduler. |
| GPU Computing Platform | Essential for developing and running accelerated MC codes. | NVIDIA GPU with CUDA toolkit and sufficient VRAM (>8GB). |
Workflow for an Integrated Simulation-Experiment Study
A typical research project integrates simulation and physical experiment, guided by the framework.
Title: Simulation-Experiment Validation Workflow
Selecting the optimal tool for simulating light propagation requires a balanced consideration of accuracy needs against computational and implementation complexity. This decision framework, grounded in the context of Monte Carlo research for biomedical applications, provides a structured pathway for researchers and drug development professionals to justify their methodological choices, thereby increasing the robustness and reproducibility of their findings in areas like treatment planning and diagnostic device development.
Conclusion
Monte Carlo simulation remains an indispensable and versatile tool for modeling light propagation in turbid media, offering unparalleled accuracy for complex biomedical scenarios. This guide has walked through its foundational physics, practical implementation, optimization for computational efficiency, and rigorous validation. The key takeaway is that a well-understood and properly optimized MC model provides a virtual laboratory for exploring light-tissue interactions, enabling the design of novel optical diagnostics, optimizing light dosimetry for therapies, and accelerating drug and device development. Future directions include tighter integration with AI for inverse problem solving, real-time simulation for clinical guidance, and the development of expansive, validated open-source libraries to democratize access for the broader research community, ultimately bridging computational models with clinical translation.