ICS vs. QuantiFERON: A Comprehensive Technical Guide for T-Cell Response Detection in Research & Drug Development
This detailed guide provides a comparative analysis of Intracellular Cytokine Staining (ICS) and QuantiFERON assays for T-cell response detection, tailored for researchers and drug development professionals.
ICS vs. QuantiFERON: A Comprehensive Technical Guide for T-Cell Response Detection in Research & Drug Development
Abstract
This detailed guide provides a comparative analysis of Intracellular Cytokine Staining (ICS) and QuantiFERON assays for T-cell response detection, tailored for researchers and drug development professionals. It explores the fundamental principles behind each technology, details practical methodologies and applications in immunology and vaccine research, addresses common troubleshooting and optimization challenges, and offers a critical validation and comparative analysis. The article synthesizes current best practices to inform assay selection, data interpretation, and application-specific optimization in biomedical research.
Understanding the Core Technologies: Principles of Flow Cytometry (ICS) vs. IFN-γ Release (QuantiFERON)
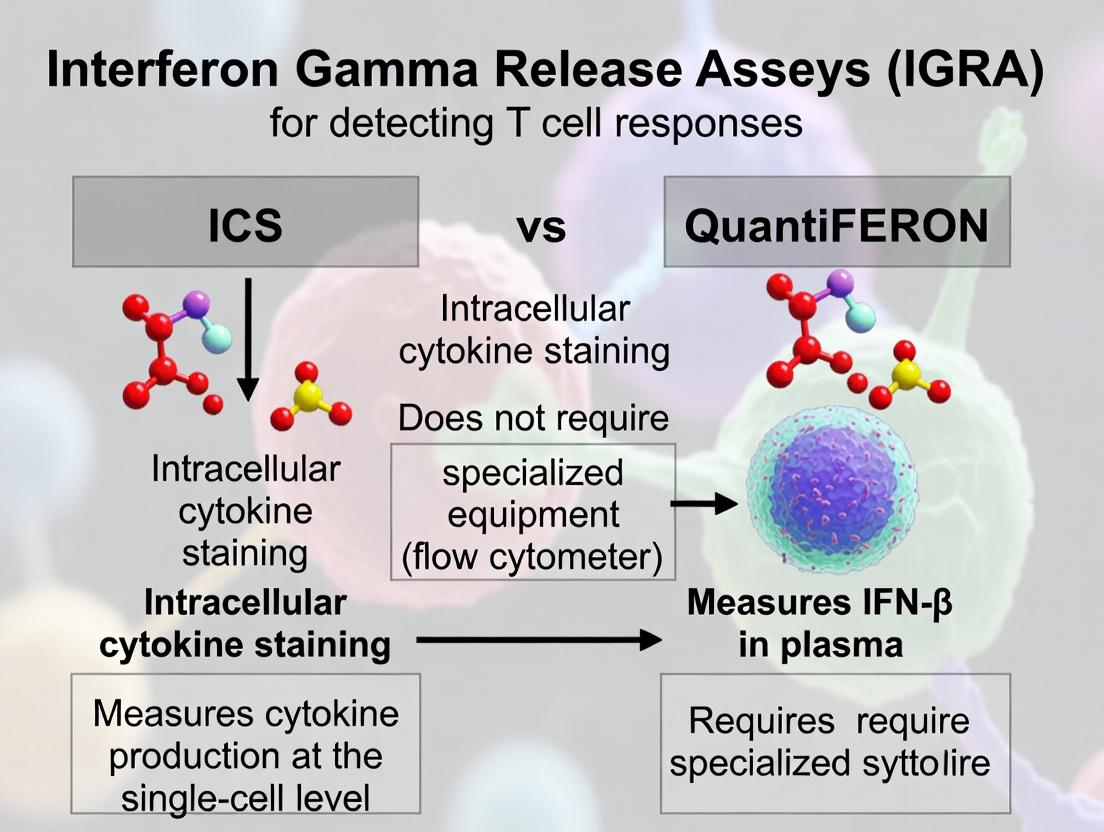
Within immunological research and clinical diagnostics for cell-mediated immunity, Intracellular Cytokine Staining (ICS) and QuantiFERON assays are pivotal technologies. Framed within the broader thesis of comparing methodologies for T-cell response detection, this guide provides an objective comparison of these two principal players. ICS is a flow cytometry-based technique that detects cytokine production at the single-cell level, while QuantiFERON is an ELISA-based platform measuring cytokine release in supernatants, primarily for diagnoses like latent tuberculosis infection (LTBI). This article compares their performance, experimental protocols, and applications for researchers and drug development professionals.
Intracellular Cytokine Staining (ICS): A functional assay that involves stimulating T-cells, inhibiting cytokine secretion, staining for surface markers, permeabilizing cells, and staining for intracellular cytokines. Analysis by flow cytometry provides data on the frequency, phenotype, and function of antigen-specific T-cells.
QuantiFERON Assays: A family of standardized in vitro blood tests. For QFT-Plus (TB), whole blood is incubated with TB-specific antigens (TB1 and TB2 tubes). Antigen-responsive T-cells release IFN-γ, which is quantified by ELISA. Other variants target CMV (QFT-CMV) or SARS-CoV-2 (QFT-SARS-CoV-2).
Experimental Protocols
Detailed ICS Protocol
- Cell Preparation: Isolate PBMCs from heparinized blood via density gradient centrifugation.
- Stimulation: Incubate cells (typically 5-10 x 10^5 cells/well) with antigen (peptide pools, proteins) or positive control (PMA/ionomycin) in the presence of a protein transport inhibitor (e.g., Brefeldin A) for 4-18 hours at 37°C, 5% CO2.
- Surface Staining: Wash cells, stain with fluorescently conjugated antibodies against surface markers (e.g., CD3, CD4, CD8, CD45RA, CCR7).
- Fixation & Permeabilization: Use a commercial fix/permeabilization kit (e.g., BD Cytofix/Cytoperm).
- Intracellular Staining: Stain with antibodies against cytokines (e.g., IFN-γ, IL-2, TNF-α).
- Acquisition & Analysis: Acquire on a flow cytometer. Analyze using software (e.g., FlowJo) to gate on live, single cells, identify T-cell subsets, and determine the percentage of cytokine-positive cells within subsets.
Detailed QuantiFERON-TB Plus Protocol
- Blood Collection: Draw blood directly into four specialized tubes: Nil (background control), Mitogen (positive control), TB1 (contains CD4+ T-cell stimulating peptides), and TB2 (contains peptides stimulating both CD4+ and CD8+ T-cells).
- Incubation: Incubate tubes for 16-24 hours at 37°C.
- Plasma Harvest: Centrifuge tubes and collect plasma supernatant.
- ELISA: Add plasma to the ELISA plate pre-coated with anti-IFN-γ antibody. Incubate, wash, add conjugate, incubate, wash, add substrate, and stop reaction.
- Quantification: Measure optical density (OD). The QFT software calculates the IFN-γ concentration (IU/mL) for each antigen tube after subtracting the Nil value. Results are interpreted as Positive, Negative, or Indeterminate based on cut-offs.
Performance Comparison & Experimental Data
The following table summarizes key performance characteristics based on published studies.
Table 1: Comparison of ICS and QuantiFERON Assay Performance
| Feature | Intracellular Cytokine Staining (ICS) | QuantiFERON (QFT-Plus exemplified) |
|---|---|---|
| Primary Readout | Frequency of cytokine+ T-cells (%), polyfunctionality | Concentration of IFN-γ in plasma (IU/mL) |
| Key Output Data | Phenotype (memory subsets), multifunctionality | Quantitative IFN-γ level; Positive/Negative diagnostic call |
| Resolution | Single-cell | Population-level (bulk supernatant) |
| Throughput | Lower (complex staining, flow acquisition) | Higher (standardized ELISA, automation-friendly) |
| Multiplexing Capacity | High (multiple cytokines & surface markers) | Low (typically single analyte, IFN-γ) |
| Standardization | Variable; lab-dependent protocols | High; FDA-approved, kit-based, standardized cut-offs |
| Sample Viability | Requires viable cells for stimulation | Uses plasma after incubation; cells not needed for ELISA |
| Primary Application | Deep immunophenotyping in research/vaccine trials | Clinical diagnostics (e.g., LTBI) and immune monitoring |
| Typical CV | Can be high (10-25%), depends on protocol rigor | Low (<10% for inter-assay precision in validated labs) |
| Data from TB Studies | Can detect 0.05-0.1% antigen-specific CD4+ T-cells | Sensitivity: ~89%; Specificity: ~99% (vs. culture in low TB incidence) |
Signaling & Workflow Diagrams
Title: Intracellular Cytokine Staining (ICS) Experimental Workflow
Title: QuantiFERON-TB Plus Assay Workflow
Title: T-cell Activation & Detection Pathways
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for T-cell Response Detection Assays
| Item | Function | Example Use Case |
|---|---|---|
| Cell Activation Cocktail | Chemically stimulates T-cells non-specifically; positive control. | PMA/Ionomycin in ICS; Mitogen tube in QFT. |
| Protein Transport Inhibitor | Blocks Golgi apparatus, causing cytokine accumulation inside cell. | Brefeldin A or Monensin in ICS protocol. |
| Fluorochrome-conjugated Antibodies | Tag surface and intracellular proteins for detection by flow cytometry. | Anti-CD3/CD4/CD8, anti-IFN-γ/IL-2/TNF-α for ICS. |
| Fixation/Permeabilization Buffer | Fixes cells and makes membrane porous for intracellular antibody access. | BD Cytofix/Cytoperm or equivalent for ICS. |
| Antigen-specific Peptide Pools | Stimulate T-cells via their specific TCR for antigen-focused assays. | CMV pp65 peptides in vaccine studies; TB peptides in QFT tubes. |
| ELISA Kit (IFN-γ) | Quantifies soluble cytokine concentration in supernatant. | The core detection system of the QuantiFERON assay. |
| Heparin Blood Collection Tubes | Prevents coagulation for PBMC isolation or direct assay use. | Sample collection for both ICS and QFT. |
| Density Gradient Medium | Isolates mononuclear cells (PBMCs) from whole blood. | Ficoll-Paque for PBMC prep prior to ICS. |
Within the broader research thesis comparing Intracellular Cytokine Staining (ICS) by flow cytometry and the QuantiFERON-TB Gold Plus (QFT-Plus) ELISA-based platform for T-cell response detection, understanding the core principles of each technology is critical. This guide dissects the operational mechanics of flow cytometry (the engine of ICS) and ELISA/ELLA (the foundation of QFT-Plus), providing a direct performance comparison with supporting experimental data.
Core Principles at Work
Flow Cytometry for Intracellular Cytokine Staining (ICS)
Flow cytometry identifies and characterizes individual cells in suspension. In ICS, cells are stimulated, a protein transport inhibitor is added to accumulate cytokines intracellularly, cells are fixed/permeabilized, and fluorescently-labeled antibodies bind to specific cytokines and surface markers. As cells pass a laser, scattered and emitted fluorescent light is captured by photomultiplier tubes, generating multiparameter data for each cell.
Diagram Title: ICS Experimental Workflow for T-cell Detection
ELISA/ELLA for QuantiFERON
The Enzyme-Linked Immunosorbent Assay (ELISA) and its digital variant, ELLA, measure soluble analyte concentration. In QFT-Plus, whole blood is incubated in Nil, TB Antigen, and Mitogen tubes. Plasma is harvested and added to a microplate pre-coated with an anti-IFN-γ capture antibody. IFN-γ from the sample binds, a detection antibody is added, followed by an enzyme conjugate and colorimetric substrate. The color intensity, proportional to IFN-γ, is measured spectrophotometrically.
Diagram Title: Sandwich ELISA Principle for IFN-γ Detection
Performance Comparison: ICS vs. ELISA (QuantiFERON)
Table 1: Head-to-Head Technical Comparison
| Parameter | Flow Cytometry (ICS) | ELISA (QuantiFERON) |
|---|---|---|
| Measured Output | Single-cell events; cytokine frequency per cell subset | Bulk analyte concentration (IFN-γ, IU/mL) in plasma |
| Multiplexing Capacity | High (8+ colors, cytokines & surface markers) | Low (Typically single analyte, e.g., IFN-γ) |
| Phenotypic Data | Yes (Can identify CD4+ vs. CD8+ T-cell sources) | No (Cannot determine responding cell subset) |
| Throughput (Setup) | Lower (Complex staining, lengthy protocol) | Higher (Simple plasma transfer) |
| Throughput (Analysis) | Lower (Requires expert gating) | Higher (Automated plate reading) |
| Sample Volume Required | Higher (5-10 mL whole blood for PBMC isolation) | Lower (1 mL per QFT-Plus tube) |
| Instrument Cost | Very High | Moderate |
| Primary Readout | % Cytokine-Positive T-cells | IFN-γ Concentration (IU/mL) |
Table 2: Representative Experimental Data from Comparative Studies (Synthesis)
| Study Focus | ICS Findings | QFT-Plus ELISA Findings | Key Implication |
|---|---|---|---|
| TB Infection Discrimination | Detected polyfunctional (IFN-γ+IL-2+) CD4 T-cells in LTBI. | Effectively distinguished TB infection vs. naive. | Both effective, but ICS gives functional subset data. |
| CD8+ T-cell Response | Identified antigen-specific IFN-γ+ CD8 T-cells in ~70% of active TB. | QFT-Plus TB2 tube designed for CD8 response; correlates moderately with ICS. | ICS is gold standard for subset resolution; ELISA provides inferential CD8 data. |
| Sensitivity in Immunocompromised | Higher sensitivity in low CD4 count settings by focusing on remaining T-cells. | Risk of indeterminate results (low Mitogen response). | ICS can be more robust where overall response is weak. |
| Precision (Inter-assay CV) | Typically 10-15% for frequency measurements. | Typically <10% for IU/mL quantification. | ELISA offers superior quantitative precision for bulk secretion. |
Detailed Experimental Protocols
Protocol A: ICS for TB-Specific T-cell Detection
- Blood Collection & Stimulation: Collect heparinized blood. Aliquot into tubes pre-coated with TB-specific peptides (e.g., ESAT-6, CFP-10) and a positive control (e.g., SEB). Include a negative control (no peptide). Add co-stimulatory antibodies (anti-CD28/CD49d).
- Incubation & Inhibition: Incubate for 16-20 hours at 37°C, 5% CO₂. Add Brefeldin A (final concentration 10 µg/mL) for the final 4-6 hours to block cytokine secretion.
- Cell Surface Staining: Transfer samples to staining tubes. Lyse red blood cells using ammonium chloride. Wash cells. Stain with surface antibody cocktail (e.g., anti-CD3, CD4, CD8) for 30 mins at 4°C in the dark.
- Fixation & Permeabilization: Wash cells. Fix with 4% paraformaldehyde for 10 mins. Wash. Permeabilize with saponin-based buffer.
- Intracellular Staining: Stain with intracellular antibody cocktail (e.g., anti-IFN-γ, IL-2, TNF-α) in permeabilization buffer for 30 mins at 4°C in the dark. Wash.
- Acquisition & Analysis: Resuspend in buffer and acquire on a flow cytometer within 24 hours. Gate on lymphocytes, singlets, CD3+, CD4+/CD8+, and analyze cytokine expression within subsets. Report as % cytokine-positive of parent population.
Protocol B: QuantiFERON-TB Gold Plus (ELISA)
- Blood Collection & Incubation: Draw blood directly into four QFT-Plus tubes: Nil (background control), TB1 (CD4 stimulus), TB2 (CD4 & CD8 stimulus), and Mitogen (positive control). Invert tubes 10 times.
- Incubation: Incubate tubes upright for 16-24 hours at 37°C (±1°C).
- Plasma Harvest: Centrifuge tubes and harvest plasma using a pipette, avoiding the cell layer. Plasma can be stored at 2-8°C for up to 3 days or frozen.
- ELISA Procedure: Use the QuantiFERON Human IFN-γ ELISA kit. Add 50 µL of standards, controls, and patient plasma to the pre-coated plate in duplicate. Incubate 120 mins (±10) at room temperature (RT). Wash 6x. Add 50 µL Anti-IFN-γ Detection Antibody conjugate. Incubate 60 mins (±5) at RT. Wash 6x. Add 50 µL Substrate Solution. Incubate 30 mins (±2) at RT in the dark. Add 50 µL Stop Solution.
- Reading & Interpretation: Measure absorbance at 450 nm (reference 620-650 nm) within 30 minutes. Subtract Nil absorbance from TB Antigen and Mitogen values. QFT-Plus result is positive if TB Antigen response minus Nil is ≥ 0.35 IU/mL IFN-γ and is ≥25% of the Nil value.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Comparative T-cell Response Studies
| Item | Function | Example (for Research Use) |
|---|---|---|
| Heparin Blood Collection Tubes | Prevents coagulation for functional cell assays. | BD Vacutainer Sodium Heparin tubes. |
| Peptide Pools (TB Antigens) | Stimulate antigen-specific T-cells. | ESAT-6 & CFP-10 peptide pools. |
| Protein Transport Inhibitor | Retains cytokines intracellularly for ICS detection. | Brefeldin A or Monensin. |
| Flow Cytometry Antibody Panel | Surface & intracellular markers for cell identification and cytokine detection. | Anti-human CD3, CD4, CD8, IFN-γ, IL-2 (fluorochrome-conjugated). |
| Cell Fixation/Permeabilization Kit | Preserves cells and allows intracellular antibody access. | BD Cytofix/Cytoperm or FoxP3/Transcription Factor Staining Buffer Set. |
| QFT-Plus Blood Collection Tubes | Standardized tubes for stimulation and plasma generation. | QuantiFERON-TB Gold Plus (Nil, TB1, TB2, Mitogen). |
| Human IFN-γ ELISA Kit | Quantifies secreted IFN-γ in plasma. | QuantiFERON ELISA kit or commercial alternative (e.g., Mabtech). |
| Microplate Washer & Spectrophotometer | Automated ELISA processing and optical density reading. | Combined ELISA plate reader/washer systems. |
Within T cell response detection research, a central thesis contrasts the high-content, single-cell resolution of Intracellular Cytokine Staining (ICS) with the high-throughput, quantitative output of the QuantiFERON platform. This guide provides an objective comparison of these methodologies, focusing on key readouts of polyfunctionality versus bulk IFN-γ quantification.
Comparison of ICS and QuantiFERON Platforms
| Feature | Intracellular Cytokine Staining (ICS) by Flow Cytometry | QuantiFERON (QFT) ELISA Platform |
|---|---|---|
| Primary Readout | Polyfunctionality (co-expression of IFN-γ, TNF-α, IL-2, etc.) at single-cell level. | Quantitative concentration of IFN-γ in plasma (IU/mL). |
| Detected Response | Cell-mediated, identifies specific cytokine-producing T cell subsets (CD4+, CD8+). | Cell-mediated, but does not identify contributing cell subtype. |
| Throughput | Lower; complex staining and flow analysis. | High; automated ELISA processing. |
| Key Metric | Frequency of antigen-specific polyfunctional T cells. | Magnitude of IFN-γ response against antigen (Nil, TB Ag, Mitogen). |
| Standardization | Requires internal optimization of panels and gating. | Highly standardized, FDA-approved kit. |
| Typical Data | % of CD4+ T cells producing [IFN-γ+, TNF-α+, IL-2+]. | IFN-γ (TB Ag - Nil) ≥ 0.35 IU/mL indicates positive response. |
Supporting Experimental Data Comparison
The following table summarizes representative data from studies comparing antigen-specific T-cell responses.
| Study Target | ICS Readout (Polyfunctionality) | QuantiFERON Readout (IFN-γ IU/mL) | Correlation Insight |
|---|---|---|---|
| TB Infection | 0.45% of CD4+ T cells are polyfunctional (IFN-γ+TNF-α+IL-2+) in latent TB. | Median: 4.15 IU/mL (Range: 0.8-10.2) in latent TB. | Strong correlation between polyfunctional CD4+ T cell frequency and IFN-γ concentration. |
| Vaccination | Increase in dual-functional (IFN-γ+IL-2+) CD4+ T cells post-vaccination. | Rise from 0.1 to 2.8 IU/mL post-vaccination. | ELISA measures aggregate output of all responding cells, including monofunctional high IFN-γ producers. |
| Immunotherapy | Expansion of polyfunctional (IFN-γ+TNF-α+) CD8+ T cells in responders. | Variable IFN-γ levels; not always predictive of clinical outcome. | ICS identifies immunologically relevant subsets that may correlate better with efficacy than bulk IFN-γ. |
Detailed Experimental Protocols
Protocol 1: Intracellular Cytokine Staining (ICS) for Polyfunctionality
- Cell Stimulation: Isolate PBMCs and incubate with antigen (e.g., peptide pools) and co-stimulatory antibodies (anti-CD28/CD49d) in the presence of a protein transport inhibitor (Brefeldin A) for 12-16 hours.
- Surface Staining: Harvest cells, stain with viability dye and fluorescently conjugated antibodies against surface markers (CD3, CD4, CD8).
- Fixation and Permeabilization: Fix cells with 4% paraformaldehyde, then permeabilize using a saponin-based buffer.
- Intracellular Staining: Stain cells with antibodies against cytokines (IFN-γ, TNF-α, IL-2).
- Flow Cytometry Acquisition: Acquire data on a flow cytometer capable of detecting 8+ colors.
- Analysis: Use Boolean gating to identify subsets co-expressing combinations of cytokines. Report as frequency of parent T cell population.
Protocol 2: QuantiFERON-TB Gold Plus (QFT-Plus) Procedure
- Blood Collection & Incubation: Draw blood directly into four QFT-Plus tubes: Nil (background), TB Antigen 1 (CD4+ T cell response), TB Antigen 2 (CD4+ & CD8+ T cell response), and Mitogen (positive control). Incubate for 16-24 hours at 37°C.
- Plasma Harvest: Centrifuge tubes and collect plasma supernatant.
- ELISA: Transfer plasma to the pre-coated QFT ELISA plate. The ELISA uses a standard sandwich format with antibodies against human IFN-γ.
- Detection: Follow kit instructions for incubation, washing, and addition of conjugate and substrate. Measure absorbance.
- Quantification: Calculate IFN-γ concentration (IU/mL) using a standard curve. The result is determined by subtracting the Nil value from the TB Antigen value (TB Ag - Nil).
Pathway and Workflow Visualizations
ICS Experimental Workflow for Polyfunctionality
QuantiFERON ELISA Quantitative Workflow
Core Thesis: ICS vs QFT Readout Comparison
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Experiment |
|---|---|
| PBMCs (Human) | Primary cells containing T lymphocytes for in vitro stimulation assays. |
| Peptide Pools / Antigens | Specific antigens (e.g., TB peptides, viral epitopes) to stimulate antigen-specific T cells. |
| Protein Transport Inhibitors (Brefeldin A/Monensin) | Block cytokine secretion, allowing intracellular accumulation for ICS detection. |
| Fluorochrome-conjugated Antibodies | Detect surface markers (CD3, CD4, CD8) and intracellular cytokines (IFN-γ, TNF-α, IL-2). |
| Flow Cytometry Fixation/Permeabilization Buffer | Fixes cells and permeabilizes membranes to allow antibody entry for intracellular staining. |
| QuantiFERON Blood Collection Tubes | Pre-coated tubes containing antigens for standardized whole-blood stimulation. |
| Human IFN-γ ELISA Kit (e.g., QFT ELISA) | Validated kit for precise quantification of IFN-γ in plasma supernatants. |
| Cell Culture Media & Supplements | Provides nutrients and environment for cell viability during stimulation. |
| Viability Staining Dye | Distinguishes live from dead cells in flow cytometry to ensure accurate analysis. |
Within the broader thesis comparing In-Cell ELISA (ICS) and QuantiFERON for detecting antigen-specific T cell responses, understanding their historical application contexts is crucial for experimental design and data interpretation. These technologies were developed to address distinct, though overlapping, research questions in immunology and drug development.
Historical Application and Comparative Performance
The table below summarizes the primary historical use cases, key performance metrics, and technological evolutions of ICS and QuantiFERON assays.
Table 1: Historical Application Contexts and Comparative Metrics of ICS vs. QuantiFERON
| Aspect | In-Cell ELISA (ICS) with Flow Cytometry | QuantiFERON-TB Gold (QFT) & Related Variants (e.g., QFT-Plus) |
|---|---|---|
| Primary Historical Use Case | Mechanistic Research & Vaccine Development. Deep phenotyping of responding T cells (e.g., CD4+ vs. CD8+, cytokine polyfunctionality). Used in preclinical and clinical immunogenicity studies. | Diagnostic Screening for Latent Infection. Population-level screening for M. tuberculosis infection (LTBI). Clinical diagnostic aid. |
| Key Measured Output | Frequency and phenotype of cytokine-producing cells at the single-cell level. (% of CD4+ IFN-γ+ T cells). | Total amount of cytokine (IFN-γ) in supernatant, measured by ELISA. Results in IU/mL. |
| Throughput (Samples) | Low to Medium. Limited by flow cytometry acquisition time and complex staining. Suited for detailed analysis of smaller cohorts. | Very High. Automated ELISA platforms allow batch processing of hundreds of samples, ideal for large-scale screening. |
| Experimental Complexity | High. Requires cell stimulation, intracellular staining, flow cytometry expertise, and complex data analysis. | Low. Standardized kit; stimulation, then simple supernatant harvest and ELISA. Minimal training required. |
| Phenotyping Capacity | High. Simultaneous measurement of multiple cytokines (IFN-γ, IL-2, TNF-α) and surface markers (CD4, CD8, CD154) per cell. | None. Does not provide information on T cell subset or polyfunctionality. |
| Supporting Experimental Data | A 2018 vaccine study showed ICS identified a polyfunctional (IFN-γ+IL-2+TNF-α+) CD4+ T cell response correlating with protection (0.5% of total CD4+), which supernatant assays missed. | Meta-analyses (2020) report QFT sensitivity of ~80% and specificity of ~99% for LTBI vs. uninfected controls in low-incidence settings. |
| Regulatory Acceptance | Common in research and as an exploratory endpoint in clinical trials. Not a standalone diagnostic. | FDA-approved/CE-marked in vitro diagnostic for LTBI. Used in public health programs. |
Experimental Protocols
Detailed ICS Protocol for T Cell Cytokine Detection:
- Cell Preparation: Isolate PBMCs from whole blood via density gradient centrifugation.
- Antigen Stimulation: Plate PBMCs (1-2 x 10^6/well) with antigen (peptide pools, proteins) or controls (mitogen for positive, media for negative). Add co-stimulatory antibodies (anti-CD28/CD49d) and protein transport inhibitor (Brefeldin A) at 1-6 hours to accumulate cytokines intracellularly.
- Incubation: Culture for 6-16 hours (typically overnight) at 37°C, 5% CO₂.
- Surface Staining: Harvest cells, stain with fluorescently-conjugated antibodies against surface markers (e.g., CD3, CD4, CD8) in the presence of a viability dye.
- Fixation & Permeabilization: Fix cells with 4% paraformaldehyde, then permeabilize with a saponin-based buffer.
- Intracellular Staining: Stain with antibodies against cytokines (e.g., IFN-γ, IL-2, TNF-α) in permeabilization buffer.
- Flow Cytometry Acquisition: Acquire data on a flow cytometer, collecting at least 100,000 lymphocyte events.
- Data Analysis: Gate on live, single, CD3+CD4+ (or CD8+) lymphocytes. Determine the frequency of cytokine-positive cells in antigen-stimulated vs. control wells.
Standard QuantiFERON-TB Gold Plus (QFT-Plus) Protocol:
- Blood Collection & Stimulation: Draw whole blood directly into four specialized tubes: Nil (negative control), TB1 (contains CD4-stimulating peptides), TB2 (contains peptides stimulating both CD4+ and CD8+ T cells), and Mitogen (positive control).
- Incubation: Incubate tubes upright for 16-24 hours at 37°C.
- Plasma Harvest: Centrifuge tubes and carefully harvest the plasma supernatant, avoiding cells.
- ELISA: Using the provided kit, assay the plasma from each tube for human IFN-γ concentration via a standard sandwich ELISA.
- Interpretation: The IFN-γ concentration (IU/mL) in the Nil tube is subtracted from the TB1 and TB2 tube values. The test result is positive if either TB1 or TB2 minus Nil is ≥ 0.35 IU/mL and the Mitogen minus Nil is ≥ 0.5 IU/mL (indicating valid assay).
Visualization of Signaling and Workflow
Diagram 1: Core ICS Detection Principle (58 chars)
Diagram 2: QFT-Plus Simplified Workflow (45 chars)
The Scientist's Toolkit
Table 2: Key Research Reagent Solutions for T Cell Response Detection
| Reagent/Material | Primary Function | Example in Protocols |
|---|---|---|
| Protein Transport Inhibitors | Blocks Golgi-mediated export, causing cytokine accumulation inside the cell for intracellular detection. | Brefeldin A (used in ICS). |
| Co-stimulatory Antibodies | Provides secondary activation signal alongside TCR engagement, enhancing T cell stimulation. | Anti-CD28/CD49d antibodies (used in ICS). |
| Peptide Antigen Pools | Mixtures of overlapping peptides spanning a target antigen, optimizing MHC binding and T cell recognition. | Used for stimulation in both ICS (research antigens) and QFT (TB-specific ESAT-6, CFP-10 peptides). |
| Cell Activation Cocktails | Positive control stimulants that non-specifically activate a large fraction of T cells. | Phorbol myristate acetate (PMA) + Ionomycin (ICS); Phytohemagglutinin (PHA) in QFT Mitogen tube. |
| Multiparametric Flow Cytometry Antibodies | Fluorochrome-conjugated antibodies for detecting surface markers and intracellular cytokines simultaneously. | Anti-CD3, CD4, CD8, IFN-γ, IL-2, TNF-α (used in ICS). |
| ELISA Kit for Human IFN-γ | Pre-coated, standardized assay for quantifying IFN-γ concentration in supernatant. | The core detection system of the QuantiFERON assay. |
Core Strengths and Inherent Limitations of Each Platform
This guide provides a comparative analysis of the two dominant platforms for detecting T-cell-mediated immune responses—the enzyme-linked immunosorbent spot (ELISpot) assay, commonly referred to as ICS (Intracellular Cytokine Staining) in this context, and the QuantiFERON-TB Gold Plus (QFT-Plus) system. The evaluation is framed within the broader thesis of selecting an optimal tool for research in immunology, vaccine development, and drug discovery, where accurate quantification of antigen-specific T-cells is critical.
ICS/ELISpot Platform: This method involves stimulating peripheral blood mononuclear cells (PBMCs) with specific antigens. In the ELISpot variant, secreted cytokines (e.g., IFN-γ) are captured on a membrane and visualized as spots, each representing a single reactive T-cell. Flow cytometry-based ICS detects cytokines retained intracellularly within individual cells, allowing for immunophenotyping.
QuantiFERON Platform: QFT-Plus is a whole-blood assay. Blood is collected directly into tubes pre-coated with antigens (TB-specific peptides). Following incubation, the concentration of IFN-γ released into the plasma is measured via enzyme-linked immunosorbent assay (ELISA). It is a closed, standardized system.
Experimental Data & Comparative Performance
The following table summarizes key performance metrics from recent comparative studies.
Table 1: Platform Comparison for T-Cell Response Detection
| Parameter | ICS/ELISpot Platform | QuantiFERON (QFT-Plus) Platform |
|---|---|---|
| Sample Input | Purified PBMCs (requires processing) | Whole blood (minimal processing) |
| Throughput | Medium; labor-intensive setup | High; amenable to batch processing |
| Sensitivity | High (can detect low-frequency responses) | Moderate; may miss weak responses |
| Specificity | High (dependent on antigen purity) | High (optimized TB antigens) |
| Reproducibility | Variable (lab-dependent protocol) | High (standardized kit) |
| Multiplexing Capacity | High (ICS: multiple cytokines/surface markers) | Low (single analyte: IFN-γ) |
| Data Output | Frequency of reactive cells (spots/counts) | Quantitative cytokine concentration (IU/mL) |
| Turnaround Time | 24-48 hours (plus PBMC isolation) | ~24 hours (from blood draw) |
| Key Strength | Single-cell resolution, immunophenotyping | Standardization, clinical simplicity |
| Inherent Limitation | Technical complexity, inter-operator variability | Limited to pre-selected antigens, no cellular data |
Detailed Experimental Protocols
Protocol 1: ICS/ELISpot for Vaccine-Specific T-Cell Detection
- PBMC Isolation: Collect venous blood in heparin tubes. Isolate PBMCs via density gradient centrifugation (e.g., Ficoll-Paque). Wash and count cells.
- Antigen Stimulation: Seed PBMCs into ELISpot plates or culture tubes (for flow ICS) pre-coated with capture antibody. Add specific vaccine peptides (e.g., SARS-CoV-2 Spike peptides) or controls (PMA/Ionomycin for positive, DMSO for negative). Incubate (37°C, 5% CO2) for 16-48 hours.
- Detection (ELISpot): Discard cells. Add biotinylated detection antibody, followed by enzyme-streptavidin conjugate. Add precipitating substrate to develop spots. Analyze using an automated ELISpot reader.
- Detection (Flow ICS): Add protein transport inhibitor (e.g., Brefeldin A) after 2 hours. After total incubation, stain for surface markers (CD3, CD4, CD8), permeabilize cells, stain for intracellular cytokines (IFN-γ, IL-2). Acquire on a flow cytometer.
Protocol 2: QuantiFERON-TB Gold Plus (QFT-Plus) Assay
- Blood Collection: Draw blood directly into four QFT-Plus tubes: Nil (negative control), TB1 (CD4+ T-cell stimulating peptides), TB2 (CD4+ and CD8+ T-cell stimulating peptides), and Mitogen (positive control).
- Incubation: Invert tubes 10 times and incubate upright for 16-24 hours at 37°C (±1°C).
- Plasma Harvest: Centrifuge and collect plasma supernatant from each tube.
- ELISA: Transfer plasma to the QFT-Plus ELISA plate. Add conjugate and substrate according to manufacturer instructions. Measure optical density (OD) at 450 nm (reference 620-650 nm). Use proprietary software to calculate IFN-γ concentration (IU/mL) and determine result (Positive, Negative, Indeterminate).
Visualizing Workflows
T-Cell Assay Workflow Comparison (Max 760px)
Platform Selection Decision Logic (Max 760px)
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Featured T-Cell Assays
| Item | Function | Primary Use Case |
|---|---|---|
| Ficoll-Paque Premium | Density gradient medium for isolation of viable PBMCs from whole blood. | ICS/ELISpot sample prep |
| Human IFN-γ ELISpot Kit | Pre-coated plates with paired capture/detection antibodies for spot formation. | ELISpot assay |
| Cell Stimulation Cocktail | Phorbol ester & Ionomycin mix; a positive control for polyclonal T-cell activation. | ICS/ELISpot control |
| Protein Transport Inhibitor | Brefeldin A or Monensin; blocks cytokine secretion for intracellular accumulation. | Flow cytometry ICS |
| Fluorochrome-conjugated Antibodies | Antibodies against CD3, CD4, CD8, IFN-γ, TNF-α for cell staining. | Flow cytometry ICS |
| QuantiFERON-TB Gold Plus Tubes | Pre-coated blood collection tubes with TB antigens and controls. | QFT-Plus assay |
| QuantiFERON ELISA Kit | Standardized 96-well plate for quantitative detection of human IFN-γ. | QFT-Plus detection |
| RPMI 1640 Complete Medium | Cell culture medium supplemented with serum, L-glutamine, and antibiotics. | Cell culture for ICS |
The choice between the ICS/ELISpot and QuantiFERON platforms is dictated by the research question's specificity and logistical constraints. The ICS/ELISpot platform offers unparalleled resolution for deep immunological investigation but requires significant expertise. In contrast, the QuantiFERON system provides a robust, standardized tool for high-throughput screening and clinical correlation studies, albeit with less granular data. A clear understanding of each platform's core strengths and inherent limitations is essential for designing rigorous and reproducible T-cell response detection research.
From Protocol to Publication: Best Practices for Implementing ICS and QuantiFERON
This guide provides a comparative analysis of standardized protocols for detecting antigen-specific T cell responses, focusing on the two dominant methodologies: Intracellular Cytokine Staining (ICS) and the QuantiFERON-TB Gold Plus (QFT-Plus) enzyme-linked immunosorbent assay (ELISA). The comparison is framed within a broader thesis evaluating high-resolution, multi-parameter single-cell assays (ICS) against high-throughput, clinical-grade cytokine release assays (QFT-Plus) for research and drug development applications.
Experimental Protocols in Detail
Intracellular Cytokine Staining (ICS) Protocol
This protocol measures cytokine production (e.g., IFN-γ) at the single-cell level via flow cytometry. Step 1: Cell Stimulation. Isolated peripheral blood mononuclear cells (PBMCs) are cultured with antigen peptides (e.g., CEF or pathogen-specific pools), positive control (e.g., PMA/ionomycin), and a negative control. Co-stimulatory antibodies (anti-CD28/CD49d) are added. Brefeldin A or Monensin is added to inhibit cytokine secretion. Step 2: Cell Surface Staining. After 6-18 hours of stimulation, cells are stained with fluorochrome-conjugated antibodies against surface markers (e.g., CD3, CD4, CD8). Step 3: Fixation & Permeabilization. Cells are fixed (typically with 4% formaldehyde) and permeabilized (with saponin-based buffer) to allow intracellular antibody access. Step 4: Intracellular Staining. Cells are stained intracellularly with antibodies against cytokines (e.g., IFN-γ, IL-2, TNF-α). Step 5: Flow Cytometry Acquisition & Analysis. Cells are acquired on a flow cytometer. Antigen-specific T cells are identified as live, CD3+CD4+/CD8+ cells positive for the cytokine of interest after subtraction of background from the negative control.
QuantiFERON-TB Gold Plus (QFT-Plus) Protocol
This is a standardized, whole-blood ELISA measuring IFN-γ release in response to Mycobacterium tuberculosis antigens. Step 1: Blood Collection & Incubation. Whole blood is collected directly into four tubes: Nil (negative control), Mitogen (positive control), TB1 (contains CD4+ T cell stimulating peptides), and TB2 (contains peptides stimulating both CD4+ and CD8+ T cells). Tubes are incubated for 16-24 hours at 37°C. Step 2: Plasma Harvest. Following incubation, tubes are centrifuged, and plasma is harvested from each tube. Step 3: ELISA Procedure. Plasma samples are added to the ELISA plate pre-coated with anti-IFN-γ antibody. After incubation and washing, a conjugated detection antibody is added, followed by a substrate solution. Step 4: Data Calculation. The IFN-γ concentration (IU/mL) for each antigen tube is determined by interpolation from a standard curve. The Nil value is subtracted from TB1 and TB2 values. The result is interpreted as positive, negative, or indeterminate based on pre-defined cut-offs (typically TB1 or TB2 - Nil ≥ 0.35 IU/mL and ≥25% of Nil value).
Performance Comparison Data
Table 1: Comparative Technical Specifications
| Feature | Intracellular Cytokine Staining (ICS) | QuantiFERON-TB Gold Plus (QFT-Plus) |
|---|---|---|
| Readout | Single-cell, multi-parameter (phenotype, function) | Bulk cytokine concentration (IFN-γ) |
| Cell Type Discernment | Yes (via CD4/CD8 surface staining) | Indirect (via separate TB1/TB2 tubes) |
| Throughput | Low to medium | High (automated processing possible) |
| Standardization | Lab-dependent protocols; requires optimization | Highly standardized, FDA-cleared, IVD |
| Key Output Metrics | % Cytokine+ T cells, MFI, polyfunctionality | IFN-γ concentration (IU/mL) |
| Sample Type | Typically PBMCs (requires processing) | Whole blood (direct from venipuncture) |
| Hands-on Time | High (multi-day protocol, staining, analysis) | Low (simple incubation & ELISA) |
Table 2: Representative Experimental Data from Recent Studies (2023-2024)
| Assay Parameter | ICS (SARS-CoV-2 spike peptides) | QFT-Plus (for TB) |
|---|---|---|
| Reported Sensitivity | 0.01-0.05% antigen-specific CD4+ T cells | 92.5% - 96.6% for active TB |
| Reported Specificity >95% (dependent on gating) | 97.1% - 99.2% (in low-prevalence regions) | |
| Coefficient of Variation (CV) | Higher inter-lab variability (~15-25%) | Lower inter-lab variability (<10% for ELISA) |
| Antigen Multiplexing | High (up to 6+ cytokines simultaneously) | Low (single analyte, IFN-γ) |
| Sample Volume Required | 10-20 mL blood (for PBMC isolation) | 1 mL per tube (4 tubes total) |
Signaling Pathway & Workflow Visualizations
Title: Intracellular Cytokine Staining Experimental Workflow
Title: QuantiFERON Whole-Blood ELISA Workflow
Title: T Cell Activation Pathway Leading to Assay Detection
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for T Cell Response Detection Assays
| Reagent/Material | Function | Primary Assay |
|---|---|---|
| Ficoll-Paque Premium | Density gradient medium for isolating viable PBMCs from whole blood. | ICS |
| Cell Activation Cocktail | Pharmacological stimulator (PMA/Ionomycin) used as a positive control for T cell activation. | ICS |
| Protein Transport Inhibitor | Brefeldin A or Monensin; blocks cytokine secretion, allowing intracellular accumulation. | ICS |
| Fluorochrome-conjugated Antibodies | Target surface markers (CD3, CD4, CD8) and intracellular cytokines (IFN-γ, TNF, IL-2). | ICS |
| Fixation/Permeabilization Solution | Stabilizes cells and creates pores for intracellular antibody access. | ICS |
| QuantiFERON-TB Gold Plus Tubes | Pre-coated blood collection tubes containing TB-specific antigens and controls. | QFT-Plus |
| QuantiFERON ELISA Kit | Contains all reagents (coated plates, standards, conjugates, substrate) for IFN-γ quantification. | QFT-Plus |
| Heparin or Lithium Heparin Blood Tubes | Anticoagulant tubes for blood collection prior to PBMC isolation. | ICS |
| Flow Cytometer with 3+ Lasers | Instrument for acquiring multi-parameter single-cell data from ICS. | ICS |
| Microplate Reader (450nm filter) | Instrument for reading optical density in the QFT-Plus ELISA. | QFT-Plus |
Within T-cell response detection research, particularly when comparing the Intracellular Cytokine Staining (ICS) assay with the QuantiFERON platform, the choice of starting sample type is a critical methodological variable. This guide objectively compares the performance characteristics, experimental requirements, and data outcomes when using fresh peripheral blood mononuclear cells (PBMCs), frozen PBMCs, and whole blood.
Performance Comparison
The following table summarizes key performance metrics relevant to T-cell assays like ICS and QuantiFERON.
| Parameter | Fresh Whole Blood (QuantiFERON) | Fresh PBMCs (ICS) | Frozen PBMCs (ICS) |
|---|---|---|---|
| Sample Requirement | Whole blood collected directly into assay tubes; no processing required at site of collection. | Large volume of blood (e.g., 50-100 mL) for sufficient PBMC yield; must be processed within 2-8 hours. | Blood can be drawn at remote site; PBMCs are isolated, cryopreserved, and shipped/stored. |
| Handling Complexity | Low; minimal hands-on time. | High; requires sterile Ficoll density gradient separation. | Medium; requires cryopreservation and thawing expertise. |
| Assay Start Flexibility | Must be stimulated immediately; no delay. | Can be rested overnight before stimulation. | Can be batch-thawed and assayed at any time. |
| Viability & Recovery | High; native cellular environment preserved. | High; freshly isolated cells. | Variable (70-95%); dependent on freeze/thaw protocol. |
| Background Cytokine Levels | Can be higher due to platelets, granulocytes. | Lower; purified mononuclear cell population. | May be elevated post-thaw due to stress. |
| Functional Response | Robust; minimal ex vivo manipulation. | Robust. | Generally preserved, but some subsets (e.g., CD8+ T cells) may be more sensitive to freezing. |
| Inter-Individual Variability | Captures natural variance, including plasma factors. | Reduces variance from non-PBMC elements. | Can introduce technical variance from cryopreservation. |
| Best Suited For | High-throughput clinical screens; point-of-care testing. | Longitudinal studies with immediate processing; delicate signaling studies. | Multi-site trials; batch analysis; rare donor studies. |
Experimental Protocols for Key Comparisons
Protocol: Assessing T-cell Viability and Recovery Post-Cryopreservation
Objective: To compare the viability and subset-specific recovery of T cells from fresh vs. frozen PBMCs.
- PBMC Isolation: Collect venous blood in heparin tubes. Dilute blood 1:1 with PBS. Layer over Ficoll-Paque PLUS and centrifuge at 400-500 × g for 30-40 min at room temperature (brake off). Harvest PBMC layer.
- Fresh Arm: Resuspend half the PBMCs in complete RPMI. Count and assess viability via Trypan Blue or automated cell counter.
- Freezing Arm: Resuspend the other half in freezing medium (90% FBS, 10% DMSO) at 5-10x10^6 cells/mL. Place in controlled-rate freezer or Mr. Frosty at -80°C, then transfer to liquid nitrogen.
- Thawing: Rapidly thaw cryovial in a 37°C water bath. Immediately transfer cells to pre-warmed medium. Wash twice to remove DMSO.
- Analysis: Count and assess viability. Stain with antibodies for CD3, CD4, CD8, and a viability dye for flow cytometry to calculate subset-specific recovery: (Cell count post-thaw × % subset) / (Cell count pre-freeze × % subset) × 100.
Protocol: IFN-γ Response Comparison (ICS Assay)
Objective: To compare antigen-specific T-cell responses across sample types using ICS.
- Stimulation: For whole blood, add peptides (e.g., CEF pool) or controls directly to blood aliquots. For fresh/frozen PBMCs, plate cells and add stimuli.
- Incubation: Add co-stimulatory antibodies (e.g., CD28/CD49d). Incubate 2 hours at 37°C, then add Brefeldin A/GolgiStop for an additional 4-16 hours.
- Processing (Whole Blood): Lyse red blood cells (e.g., with ammonium chloride), wash, and permeabilize.
- Processing (PBMCs): Wash, stain surface markers, fix, permeabilize, and stain intracellular IFN-γ.
- Acquisition & Analysis: Acquire on a flow cytometer. Gate on lymphocytes, singlets, live CD3+ T cells, and report %IFN-γ+ within CD4+ or CD8+ populations.
Visualizing Workflow and Decision Pathways
Title: Sample Type Decision Workflow for T-cell Assays
Title: ICS vs QuantiFERON Experimental Workflow Comparison
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Primary Function | Notes for Sample Type Comparison |
|---|---|---|
| Sodium Heparin Tubes | Anticoagulant for blood collection. | Preferred over EDTA for functional assays; maintains cell viability and function for both whole blood and PBMC work. |
| Ficoll-Paque PLUS | Density gradient medium. | Essential for isolating PBMCs from whole blood. Critical for generating both fresh and frozen PBMC samples. |
| Cryopreservation Medium (FBS/DMSO) | Protects cells during freezing. | Quality is paramount for frozen PBMC viability. Controlled-rate freezing is recommended. |
| Peptide Pools (CEF, viral antigens) | Antigenic stimuli for T cells. | Used in both ICS and QuantiFERON. Must be titrated for optimal response, especially in frozen PBMCs. |
| Brefeldin A / Monensin | Inhibits protein transport, accumulates cytokines. | Critical for ICS assay sensitivity in all sample types. Incubation time may be optimized for frozen cells. |
| Anti-CD28/CD49d Antibodies | Co-stimulatory signals. | Enhances weak antigen-specific responses. Particularly important for optimal activation in frozen PBMC assays. |
| Viability Dye (e.g., Live/Dead Fixable) | Distinguishes live from dead cells. | Crucial for frozen PBMC analysis to exclude false-positive signals from dead/dying cells. |
| Human IFN-γ ELISA Kit | Quantifies soluble IFN-γ. | Core of the QuantiFERON assay. Standardized kit ensures reproducibility for whole blood testing. |
| Flow Cytometry Antibody Panel | Detects surface and intracellular markers. | For ICS. Panel design must account for potential freezer-induced changes in epitopes (e.g., CD62L shedding). |
This guide is framed within a broader thesis comparing Intracellular Cytokine Staining (ICS) with QuantiFERON assays for detecting antigen-specific T-cell responses. While QuantiFERON offers a simplified, ELISA-based readout of IFN-γ release, ICS provides multi-parameter, single-cell resolution, enabling deep immunophenotyping of responding T cells. The critical challenge in ICS is designing a robust fluorescent panel to capture key functional and phenotypic markers without spectral overlap. This guide compares critical marker choices and the performance of related reagent solutions.
Critical Marker Comparison for ICS Panels
The selection of markers dictates the biological questions addressable by an ICS experiment. The table below compares the core functional and phenotypic markers, their biological significance, and considerations for panel design.
Table 1: Critical Marker Categories for ICS Panel Design
| Marker Category | Key Examples | Biological Role in ICS | Panel Design Priority | Typical Fluorochrome Conjugate Brightness |
|---|---|---|---|---|
| T Cell Lineage | CD3, CD4, CD8 | Identifies major T cell subsets; essential for gating. | Highest (Backbone) | CD3/CD4/CD8: High (e.g., BV421, APC) |
| Cytokines (Functional) | IFN-γ, IL-2, TNF-α, IL-4, IL-17 | Defines effector function (Th1, Th2, Th17, Tc1). | High (Core Readout) | Medium-High (e.g., PE, APC, FITC) |
| Activation/Memory | CD69, CD25, CD154 (CD40L), HLA-DR | Indicates recent activation and proliferation. | Medium-High (Context) | Variable (CD69: Med; CD25: Low-Med) |
| Cytotoxic Degranulation | CD107a | Surrogate marker for cytotoxic granule release. | Medium (CTL assays) | Medium (e.g., PE-Cy5) |
| Exhaustion/Regulation | PD-1, LAG-3, TIM-3, FoxP3 | Identifies dysfunctional or regulatory subsets. | Context-Dependent | Often Low (Requires bright dyes) |
Comparative Performance of Key Reagent Solutions
The performance of an ICS assay heavily depends on the quality of the stimulation cocktail, protein transport inhibitors, and antibody conjugates. The following table compares commonly used alternatives based on recent literature and product datasheets.
Table 2: Comparison of Critical ICS Reagent Solutions
| Reagent Type | Product/Alternative A | Product/Alternative B | Key Performance Differentiators | Recommended Use Case |
|---|---|---|---|---|
| Stimulation Cocktail | Cell Activation Cocktail (with Brefeldin A) | PMA/Ionomycin | A: More physiological, TCR-dependent. B: Potent, non-specific, can downmodulate CD4/CD8. | A: Antigen-specific responses. B: Positive control for max cytokine production. |
| Protein Transport Inhibitor | Brefeldin A (BFA) | Monensin | A: Blocks earlier in Golgi; better for most cytokines. B: Accumulates in granules; good for chemokines. A typically gives higher IFN-γ signals. | A: Standard for IFN-γ, TNF-α. B: For IL-8, MIP-1β. |
| Fixable Viability Dye | Zombie NIR | 7-AAD | A: Fixable, allows intracellular staining. B: Non-fixable, must be added post-permeabilization. A offers greater workflow flexibility. | A: Standard for modern ICS. B: For simple surface stain only. |
| Permeabilization Buffer | FoxP3/Transcription Factor Staining Buffer Set | saponin-based buffers | A: Strong, for nuclear antigens (FoxP3, Ki-67). B: Milder, standard for cytokines. Use A only if nuclear targets are needed. | B: Standard cytokine staining. A: For FoxP3, Ki-67 co-staining. |
Detailed Experimental Protocol for Comparative ICS
Protocol: Assessing Antigen-Specific CD4+ T Cells via ICS (Compared to QuantiFERON Workflow)
- Sample Preparation: Isolate PBMCs from heparinized blood via density gradient centrifugation. Adjust concentration to 1-2 x 10^6 cells/mL in complete RPMI-1640.
- Stimulation: Aliquot 0.5-1 mL cell suspension into tubes/polypropylene wells. Set up conditions:
- Test: Antigen (e.g., peptide pool, 1-2 µg/mL).
- Positive Control: Cell Activation Cocktail (e.g., 1x concentration).
- Negative Control: DMSO/media alone.
- Inhibition: Add protein transport inhibitor (e.g., Brefeldin A, 1 µg/mL) simultaneously with stimulants.
- Incubation: Incubate for 4-6 hours (optimal for most cytokines) at 37°C, 5% CO2. Note: QuantiFERON incubates for 16-24 hours.
- Surface Staining:
- Transfer cells to FACS tubes.
- Wash once with PBS.
- Stain with surface antibody cocktail (e.g., CD3, CD4, CD8, CD69, viability dye) in PBS for 20-30 mins at 4°C in the dark.
- Wash with PBS.
- Fixation & Permeabilization:
- Fix cells with 4% paraformaldehyde for 10-15 mins at RT.
- Wash with PBS.
- Permeabilize cells with 1x permeabilization buffer (e.g., saponin-based) for 10 mins at RT.
- Intracellular Staining:
- Centrifuge, decant supernatant.
- Stain with intracellular antibody cocktail (e.g., IFN-γ, IL-2, TNF-α) in permeabilization buffer for 30 mins at 4°C in the dark.
- Wash with permeabilization buffer, then PBS.
- Acquisition: Resuspend in PBS/formaldehyde. Acquire on a flow cytometer capable of detecting all fluorochromes used. Analyze data to gate on live, singlet, CD3+CD4+ (or CD8+) cells and assess cytokine frequency and co-expression patterns.
Diagram: ICS vs. QuantiFERON Workflow Comparison
Title: ICS and QuantiFERON Assay Workflow Comparison
Diagram: Logical Decision Tree for Marker Selection in ICS Panel Design
Title: Decision Tree for ICS Panel Marker Selection
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for an ICS Experiment
| Item | Function in ICS Experiment | Example/Note |
|---|---|---|
| PBMCs or Whole Blood | Source of primary T cells for assay. | Heparin or EDTA tubes for blood collection. |
| Antigen (Stimulus) | Triggers antigen-specific T cell activation. | Peptide pools, viral lysates, or antigen-coated beads. |
| Protein Transport Inhibitor | Blocks cytokine secretion, allowing intracellular accumulation. | Brefeldin A (BFA) or Monensin. |
| Fluorochrome-Conjugated Antibodies | Detect surface and intracellular targets. | Titrate for optimal signal-to-noise; consider brightness hierarchy. |
| Fixable Viability Dye | Distinguishes live from dead cells; must be fixable. | Zombie dyes, LIVE/DEAD Fixable stains. |
| Permeabilization Buffer | Allows antibodies to enter cell and stain cytokines. | Saponin-based for cytokines; harsher buffers for nuclear antigens. |
| Flow Cytometer | Instrument for data acquisition from stained samples. | Requires lasers/filters matching fluorochrome panel. |
| Flow Cytometry Analysis Software | For data visualization, gating, and quantitative analysis. | FlowJo, FCS Express, Cytobank. |
The assessment of T-cell-mediated immunogenicity is a critical endpoint in vaccine clinical trials. A central thesis in immunomonitoring is the comparison of Intracellular Cytokine Staining (ICS) by flow cytometry and QuantiFERON-style ELISA assays for detecting antigen-specific T-cell responses. This guide provides an objective comparison within the context of vaccine development.
Methodological Comparison: ICS vs. QuantiFERON
Table 1: Core Assay Characteristics
| Feature | Intracellular Cytokine Staining (ICS) | QuantiFERON / IFN-γ Release Assay (IGRA) |
|---|---|---|
| Primary Readout | Single-cell cytokine protein detection via flow cytometry. | Bulk IFN-γ concentration in supernatant via ELISA/CLIA. |
| Key Outputs | Frequency of cytokine+ T-cells, immunophenotyping (CD4+/CD8+), polyfunctionality. | Quantitative IFN-γ level (IU/mL). Positive/Negative cutoff. |
| Throughput | Medium. Complex sample processing, requires flow cytometer. | High. Amenable to plasma/serum, automated ELISA platforms. |
| Multiplexing | High. Can detect multiple cytokines & surface markers simultaneously. | Low. Typically single analyte (IFN-γ), though multiplex variants exist. |
| Cell Viability Required | Yes. Requires viable cells for stimulation and staining. | No. Measures secreted analyte post-stimulation. |
| Key Advantage | Deep phenotypic and functional profiling at single-cell resolution. | Standardized, high-throughput, less technical variability. |
| Key Limitation | Technically complex, requires specialized instrumentation & expertise. | Lacks cellular resolution and phenotypic data. |
Supporting Experimental Data from Vaccine Trials
Recent studies in COVID-19 and HIV vaccine trials highlight performance differences.
Table 2: Comparative Data from Recent Clinical Trial Contexts
| Study Context (Vaccine) | ICS Findings | QuantiFERON/IGRA Findings | Correlation & Interpretation |
|---|---|---|---|
| mRNA COVID-19 Vaccine (Booster Trial) | Detected polyfunctional (IFN-γ+/IL-2+/TNFα+) CD4+ and CD8+ T cells. Frequency: 0.1-0.8% of total T cells. | Robust IFN-γ release post-boost (Median: >1.0 IU/mL). | Strong correlation between IFN-γ secretion magnitude and frequency of IFN-γ+ CD4+ T-cells by ICS (r=0.82). |
| HIV Vaccine (Phase I/II) | Identified rare, antigen-specific CD8+ T-cell subsets (≤0.05%) with specific homing markers (e.g., CCR5). | 60% of vaccinees showed positive IFN-γ response above cutoff. | IGRA identified responders; ICS provided mechanistic insight into the quality and homing potential of the elicited cells. |
| Tuberculosis Vaccine | Showed induction of IL-2+IFN-γ+ CD4+ T cells, a phenotype associated with long-lived memory. | All vaccinees converted to QuantiFERON-positive, but with wide variance in IU/mL values. | ICS defined the favorable functional profile of responses that IGRA could only quantify in bulk. |
Detailed Experimental Protocols
Protocol 1: ICS for Vaccine Trial PBMC Samples
- Cell Preparation: Isolate PBMCs from heparinized/vacutainer blood via density gradient centrifugation. Rest cells for 4-6 hours in complete RPMI media.
- Antigen Stimulation: Aliquot 1x10^6 PBMCs per condition. Stimulate with vaccine antigen peptides, positive control (SEB/PMA+ionomycin), and negative control (media alone). Use a protein transport inhibitor (e.g., Brefeldin A) after 2 hours.
- Incubation: Incubate for 18 hours at 37°C, 5% CO2.
- Surface Staining: Stain with viability dye and surface marker antibodies (e.g., CD3, CD4, CD8) for 30 min at 4°C.
- Fixation/Permeabilization: Fix cells with 4% PFA, then permeabilize with saponin-based buffer.
- Intracellular Staining: Stain with fluorescently-labeled anti-cytokine antibodies (e.g., IFN-γ, IL-2, TNF-α) for 30 min at 4°C.
- Acquisition & Analysis: Acquire on a flow cytometer (≥8 colors). Analyze using Boolean gating to identify antigen-specific, cytokine-producing T-cell subsets.
Protocol 2: QuantiFERON/IGRA for Vaccine Trial Samples
- Blood Collection: Draw blood directly into QuantiFERON tubes or heparin tubes for custom antigen tubes.
- Nil tube (background control).
- Mitogen tube (positive control).
- Antigen tube(s) (coated with vaccine antigen peptides).
- Incubation: Incubate blood tubes for 16-24 hours at 37°C.
- Plasma Harvest: Centrifuge tubes and harvest plasma supernatant.
- ELISA/CLIA: Use commercial ELISA or Chemiluminescence Immunoassay (CLIA) kit to measure IFN-γ concentration in each plasma sample per manufacturer's instructions.
- Data Analysis: Subtract Nil value from Antigen and Mitogen values. Report as IU/mL of IFN-γ. Apply kit-specific cutoff (e.g., ≥0.2 IU/mL & ≥15% of Nil) to determine positive response.
Visualization of Assay Workflows
Title: ICS Assay Workflow for T-cell Analysis
Title: IGRA Workflow for Bulk IFN-γ Measurement
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for T-cell Immunogenicity Assays
| Item | Function | Example/Note |
|---|---|---|
| Peptide Pools | Overlapping peptides spanning vaccine antigen(s) for in vitro T-cell stimulation. | Often 15-mer peptides, pooled for comprehensive coverage. |
| Protein Transport Inhibitors | Block cytokine secretion, allowing intracellular accumulation for ICS detection. | Brefeldin A, Monensin. |
| Fluorochrome-conjugated Antibodies | Detect surface markers and intracellular cytokines via flow cytometry. | Critical for panel design; requires titration. |
| Cell Stimulation Cocktails | Positive controls for T-cell functionality. | Phorbol ester (PMA) + Ionomycin; Staphylococcal Enterotoxin B (SEB). |
| IGRA-specific Blood Tubes | Pre-coated tubes for standardized whole-blood stimulation. | QuantiFERON TB2 Gold, or custom antigen-coated tubes. |
| Recombinant IFN-γ Standard | Calibrator for ELISA/CLIA quantification of IFN-γ. | Essential for generating standard curve. |
| Viability Dye | Distinguish live/dead cells in flow cytometry for accurate analysis. | Fixable viability dyes (e.g., Zombie dye) are preferred. |
| Permeabilization Buffer | Permeabilize cell membrane to allow antibody access to intracellular cytokines. | Saponin-based buffers are standard for ICS. |
This comparison guide objectively evaluates the performance of Intracellular Cytokine Staining (ICS) via flow cytometry against QuantiFERON ELISA-based assays for monitoring antigen-specific T-cell responses in infectious disease and cancer immunotherapy research. The analysis is framed within the broader thesis of Multiparametric Functional Profiling vs. Soluble Biomarker Quantification in immune monitoring.
Performance Comparison: ICS vs. QuantiFERON Platforms
The following table summarizes key performance metrics based on recent comparative studies and application-specific validations.
Table 1: Comparative Performance of ICS and QuantiFERON Assays
| Parameter | Intracellular Cytokine Staining (ICS) | QuantiFERON Assays |
|---|---|---|
| Primary Readout | Frequency of cytokine-producing T-cells (e.g., IFN-γ+, TNF-α+, IL-2+), multiparametric. | Concentration of IFN-γ in plasma (IU/mL), single analyte. |
| Cell Type Resolution | High (Can distinguish CD4+ vs. CD8+, memory subsets, polyfunctional cells). | None (Bulk supernatant measurement). |
| Antigen Stimulation Flexibility | High (Custom peptide pools, viral lysates, tumor antigens). | Fixed (Tuberculosis-specific peptides, SARS-CoV-2 spike peptides). |
| Throughput | Medium (Complex staining, acquisition, and analysis). | High (Simple ELISA/CLIA workflow). |
| Sample Type | Fresh or cryopreserved PBMCs required. | Whole blood (minimal processing). |
| Key Application in TB | Discriminates active vs. latent TB via polyfunctional T-cell signatures. | Diagnostic aid for latent TB infection (QFT-Plus). |
| Key Application in COVID-19 | Characterizes vaccine-induced CD4+/CD8+ memory quality and durability. | Measures gross IFN-γ release to spike peptides (QFN-SARS). |
| Key Application in Cancer Immunotherapy | Critical for assessing tumor-specific T-cell expansion and exhaustion (PD-1+, TIM-3+). | Limited utility; measures bystander IFN-γ to shared antigens (e.g., QFT-IT). |
| Quantitative Data (Sample Study: COVID-19 Vaccine) | 0.1-0.5% Spike-specific CD8+ T-cells (ICS); correlates with protection. | 0.8-2.5 IU/mL IFN-γ release (QFN-SARS); correlates with antibody titer. |
| Reference | (Rydyznski Moderbacher et al., Cell, 2020) | (Matsuoka et al., J Infect, 2022) |
Detailed Experimental Protocols
Protocol 1: ICS for SARS-CoV-2 T-Cell Response
- Sample Preparation: Isolate PBMCs from heparinized blood via density gradient centrifugation. Count and resuspend in complete RPMI-1640 medium.
- Stimulation: Plate 1x10^6 PBMCs/well in a 96-well U-bottom plate. Add:
- Test: SARS-CoV-2 MegaPool peptides (1 µg/mL per peptide).
- Positive Control: Staphylococcal enterotoxin B (SEB, 1 µg/mL).
- Negative Control: DMSO (peptide diluent) or medium alone.
- Incubation: Add co-stimulatory antibodies (anti-CD28/CD49d, 1 µg/mL) and protein transport inhibitor (Brefeldin A, 10 µg/mL). Incubate for 18 hours at 37°C, 5% CO2.
- Staining: a. Surface Stain: Wash cells, stain with viability dye and surface antibodies (anti-CD3, CD4, CD8) for 30 min at 4°C. b. Fixation/Permeabilization: Use commercial Fix/Perm buffer (e.g., BD Cytofix/Cytoperm) for 20 min at 4°C. c. Intracellular Stain: Wash with Perm/Wash buffer, stain with anti-cytokine antibodies (IFN-γ, IL-2, TNF-α) for 30 min at 4°C.
- Acquisition & Analysis: Wash, resuspend, and acquire on a flow cytometer (≥8 colors). Analyze using software (e.g., FlowJo) to gate on live, single, CD3+ T-cells and determine cytokine+ frequencies within CD4+ and CD8+ subsets.
Protocol 2: QuantiFERON-TB Gold Plus (QFT-Plus)
- Blood Collection & Incubation: Draw blood directly into four QFT-Plus tubes: Nil (background), TB1 (CD4+ stimulus), TB2 (CD4+ & CD8+ stimulus), and Mitogen (positive control). Invert tubes 10 times.
- Incubation: Incubate tubes upright for 16-24 hours at 37°C (±1°C).
- Plasma Harvest: Centrifuge tubes and harvest plasma supernatant using a pipette, avoiding the cell layer.
- ELISA Development: Use the QFT-Plus ELISA kit. Add 50 µL of standard, control, or patient plasma to pre-coated wells. Add conjugate and substrate sequentially per kit instructions. Stop reaction and read optical density at 450 nm (reference 620-650 nm).
- Calculation: Subtract Nil value from TB1, TB2, and Mitogen values. IFN-γ concentration (IU/mL) is determined from the standard curve. Interpretation: TB1 or TB2 ≥ 0.35 IU/mL (and ≥25% of Nil) indicates a positive result.
Signaling Pathways and Workflows
Title: Intracellular Cytokine Staining (ICS) Experimental Principle
Title: QuantiFERON (QFT) Assay Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Materials for T-Cell Monitoring Assays
| Item | Function | Example (Vendor-Neutral) |
|---|---|---|
| Peptide Pools | Synthetic overlapping peptides spanning target antigens (e.g., SARS-CoV-2 Spike, TB antigens ESAT-6/CFP-10, tumor neoantigens). Provide specific T-cell stimulation. | SARS-CoV-2 MegaPool, CE/CF peptide pools (for TB), custom neoantigen pools. |
| Protein Transport Inhibitors | Inhibit Golgi-mediated export, causing cytokine accumulation inside the cell for ICS detection. | Brefeldin A, Monensin. |
| Co-stimulatory Antibodies | Enhance activation signals during stimulation, improving sensitivity, especially for low-frequency T-cells. | Anti-CD28 and anti-CD49d antibodies. |
| Fixation/Permeabilization Buffer | ICS-specific reagents that preserve cell structure while allowing intracellular antibody access. | Commercial fix/perm kits (formaldehyde-based fixative, saponin-based perm buffer). |
| Fluorochrome-conjugated Antibodies | Detect surface markers (CD3, CD4, CD8) and intracellular cytokines (IFN-γ, IL-2, TNF-α) for flow cytometry. | Anti-human CD3 (BV510), CD4 (APC-Cy7), CD8 (PerCP-Cy5.5), IFN-γ (PE-Cy7). |
| Viability Dye | Distinguish live from dead cells during flow cytometry, crucial for accurate analysis. | Fixable viability dye (e.g., Zombie NIR). |
| QuantiFERON Collection Tubes | Pre-coated blood collection tubes containing stimulants (antigens, mitogen) for standardized whole-blood assays. | QFT-Plus Nil, TB1, TB2, Mitogen tubes. |
| IFN-γ ELISA/CLIA Kit | Quantifies IFN-γ concentration in plasma supernatants from stimulated samples. | QFT-Plus ELISA kit, compatible chemiluminescence immunoassay (CLIA) kits. |
Navigating Pitfalls: Expert Solutions for ICS and QuantiFERON Assay Challenges
Intracellular Cytokine Staining (ICS) is a cornerstone technique in immunology for detecting antigen-specific T cell responses. However, researchers frequently encounter methodological challenges that compromise data quality. This guide objectively compares solutions to these common issues, framed within the broader research context of ICS versus QuantiFERON for T cell detection, and provides supporting experimental data.
Key Challenges and Comparative Solutions
Addressing Low Signal Intensity
Low signal can obscure true positive T cell responses, critical when comparing to the systemic, ELISA-based readout of QuantiFERON.
Comparative Data: Table 1: Comparison of Signal Enhancement Reagents in a CMV pp65 Peptide Stimulation Assay (n=5 donors)
| Enhancement Strategy | Mean % CD4+ IFN-γ+ | Signal-to-Noise Ratio | Viability Post-Assay |
|---|---|---|---|
| Standard Protocol | 0.15% ± 0.03 | 4.2 | 78% ± 5 |
| Protein Transport Inhibitor Cocktail A | 0.41% ± 0.07 | 12.1 | 82% ± 4 |
| Enhanced Co-stimulation (anti-CD28/CD49d) | 0.38% ± 0.06 | 11.5 | 75% ± 6 |
| Extended Stimulation (36h) | 0.45% ± 0.08 | 9.8 | 65% ± 8 |
Protocol: PBMCs were stimulated with CMV pp65 peptide pool for 6h (unless stated). Cocktail A (containing brefeldin A, monensin, and a proprietary enhancer) was added at 1h. Co-stimulatory antibodies were used at 1µg/mL. Cells were stained with anti-CD3, CD4, CD8, IFN-γ, and a viability dye.
Mitigating High Background Noise
High background, often from non-specifically activated or dying cells, reduces assay specificity—a key metric where QuantiFERON often holds an advantage.
Comparative Data: Table 2: Impact of Background Reduction Techniques on Unstimulated Controls
| Technique | Background % IFN-γ+ CD8+ T cells | Reduction vs. Standard | Impact on PHA Response |
|---|---|---|---|
| Standard Wash Buffer | 0.08% ± 0.02 | Baseline | 100% (Reference) |
| High-Stringency Wash Buffer | 0.03% ± 0.01 | 62.5% | 98% ± 3 |
| Fc Receptor Block (10 min) | 0.05% ± 0.01 | 37.5% | 99% ± 2 |
| Live/Dead Discrimination & Exclusion | 0.04% ± 0.01 | 50.0% | 95% ± 4 |
| Combined (High-Stringency + Fc Block) | 0.02% ± 0.005 | 75.0% | 97% ± 2 |
Protocol: Cryopreserved PBMCs were rested overnight. Prior to staining, cells were treated with Fc receptor blocking reagent for 10 minutes at 4°C. Washes were performed with either standard PBS/BSA or a high-stringency buffer containing mild detergents. A fixable viability dye was used for all conditions, and dead cells were excluded from analysis.
Resolving Viability Concerns
Poor cell health post-stimulation and processing leads to cell loss and artifactual cytokine capture, a common pitfall not faced by the plasma-based QuantiFERON.
Comparative Data: Table 3: Viability Preservation with Different Stimulation & Processing Kits
| System / Kit | Viability Post-Stim | Viability Post-Perm/Stain | Recovery of Seeded Cells |
|---|---|---|---|
| Standard In-House Protocol | 68% ± 6 | 52% ± 7 | 45% ± 8 |
| Commercial Kit X (Cocktail-Based) | 85% ± 4 | 78% ± 5 | 72% ± 6 |
| Commercial Kit Y (Protein-Based) | 80% ± 5 | 70% ± 6 | 65% ± 7 |
| Reduced Permeabilization Time (15min vs 30min) | 68% ± 6 | 65% ± 5 | 60% ± 7 |
Protocol: PBMCs were stimulated with SEB for 12h. Kits were used according to manufacturers' instructions. The standard protocol used 0.5% saponin for 30min permeabilization. Cell recovery was calculated using counting beads flow cytometry.
Experimental Visualization
Title: ICS Workflow with Common Issues and Solutions
Title: ICS Signal and Background Generation Pathways
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Reagents for Optimizing ICS Assays
| Reagent / Material | Primary Function | Key Consideration for Optimization |
|---|---|---|
| Protein Transport Inhibitor Cocktail | Blocks cytokine secretion, causing intracellular accumulation. | Combination (e.g., BFA + monensin) often yields higher signals than single agents. |
| Co-stimulatory Antibodies (anti-CD28/CD49d) | Provides secondary activation signal, enhancing response. | Critical for weak antigens; can increase background if used in unstimulated controls. |
| High-Stringency Wash Buffer | Reduces non-specific antibody binding. | Often contains detergents like Tween-20; concentration must be optimized to preserve epitopes. |
| Fc Receptor Blocking Reagent | Binds to FcRs on immune cells, preventing antibody adherence. | Human and mouse-specific blocks differ; use prior to surface staining. |
| Fixable Viability Dye (e.g., Zombie, Live/Dead) | Covalently labels dead cells for exclusion during analysis. | Must be used before fixation and permeabilization. |
| Optimized Permeabilization Buffer | Compromises membrane to allow intracellular antibody access. | Time and concentration are critical for viability; commercial kits are often pre-optimized. |
| Counting Beads (Absolute) | Allows calculation of absolute cell counts recovered. | Essential for assessing loss during processing and standardizing results. |
| Peptide Pools / Superantigens (SEB, CEF) | Positive control antigens. | CEF (CMV, EBV, Flu) pool validates CD8+ response; SEB validates CD4+ response. |
Thesis Context: This guide provides a comparative analysis of key stimulation parameters for detecting antigen-specific T cell responses, framed within the methodological debate of Intracellular Cytokine Staining (ICS) versus QuantiFERON-TB Gold Plus (QFT-Plus) for research applications. Optimal in vitro stimulation is critical for assay sensitivity and specificity in both platforms.
Comparative Performance of Stimulation Parameters
The following tables synthesize experimental data from recent studies comparing the effects of peptide pool design, antigen concentration, and stimulation duration on T cell response magnitude and quality.
Table 1: Peptide Pool Strategy Comparison
| Parameter | Overlapping 15-mers | PepMix (Pool of Peptides) | Peptivator (Selected Epitopes) | Notes |
|---|---|---|---|---|
| Coverage | Full protein sequence | Selected antigens | Predicted HLA binders | PepMix offers broad coverage; Peptivator is HLA-biased. |
| Response Magnitude (IFN-γ SFU/10⁶ PBMCs) | 120 ± 45 | 185 ± 60 | 220 ± 70 | Data from CMV pp65 stimulation; Peptivator often yields higher spot counts in ELISpot. |
| Background Noise | Moderate | Low | Very Low | Selected epitopes reduce nonspecific stimulation. |
| Cost & Complexity | High (custom synthesis) | Moderate | Moderate | Overlapping libraries are expensive for large genomes. |
| Best for ICS/QFT Context | Novel antigen discovery | Routine QFT antigen comparison (e.g., CEFX pools) | HLA-specific response studies |
Table 2: Antigen Concentration & Stimulation Duration Optimization
| Assay | Optimal [Ag] Range | Typical Duration | Key Cytokine Target | Effect of Prolonged Stimulation (>24h) |
|---|---|---|---|---|
| ICS (Flow Cytometry) | 0.5 - 2 µg/mL | 4-6h (with protein transport inhibitor) | IFN-γ, TNF-α, IL-2 | Increased cell death, reduced viability, potential cytokine receptor internalization. |
| QFT-Plus (ELISA) | 1 - 5 µg/mL | 16-24h (no inhibitor) | IFN-γ (in supernatant) | Plateau in IFN-γ secretion; possible epitope exhaustion. |
| ELISpot | 1 - 10 µg/mL | 16-24h | IFN-γ, Granzyme B | Spot size may increase, but risk of confluence. |
Table 3: Direct Comparison: ICS vs. QuantiFERON Core Needs
| Stimulation Factor | Intracellular Cytokine Staining (ICS) | QuantiFERON-TB Gold Plus (QFT-Plus) |
|---|---|---|
| Primary Goal | Multiparametric, single-cell resolution (phenotype, function). | Quantifiable total IFN-γ release from whole population. |
| Critical Optimization | Brefeldin A/Monensin addition timing; surface stain viability. | Antigen tube coating efficiency; plasma separation. |
| Peptide Pool Preference | Smaller, focused pools for clear background. | Predefined TB-specific ESAT-6 and CFP-10 peptide pools. |
| Data Output | % of cytokine+ CD4+/CD8+ T cells. | IFN-γ concentration (IU/mL) in plasma. |
Experimental Protocols for Key Comparisons
Protocol 1: Titration of Antigen Concentration for ICS
- Isolate PBMCs from heparinized blood via density gradient centrifugation.
- Plate cells in 96-well U-bottom plates at 1x10⁶ cells/well in RPMI-1640 + 10% FBS.
- Add peptide pool (e.g., CEFX CMV/EBV/FLU) in serial dilutions (0.1, 0.5, 1, 2, 5 µg/mL). Include positive control (PMA/Ionomycin) and negative control (DMSO/media).
- Incubate for 2 hours at 37°C, 5% CO₂.
- Add protein transport inhibitor (e.g., Brefeldin A, 1µg/mL).
- Continue incubation for an additional 4 hours.
- Proceed to staining: Surface markers (CD3, CD4, CD8) → fixation/permeabilization → intracellular staining (IFN-γ, IL-2).
- Acquire on flow cytometer and analyze % cytokine-positive T cells.
Protocol 2: Duration Kinetics for QFT-Plus-like ELISA
- Stimulate whole blood or PBMCs in QFT tubes or equivalent: Nil (background), TB1 (CD4+ epitopes), TB2 (CD4+ & CD8+ epitopes), Mitogen.
- Incubate at 37°C. Remove aliquots at 4h, 8h, 16h, 24h, and 48h.
- Centrifuge to harvest plasma/supernatant.
- Quantify IFN-γ using a standardized ELISA kit per manufacturer instructions.
- Plot IFN-γ concentration vs. time to identify plateau phase for optimal detection.
Visualizing the Experimental Workflow
Title: ICS vs QFT Assay Workflow Comparison
Title: T Cell Activation and Cytokine Production Pathway
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Stimulation Assays |
|---|---|
| Peptivator (Miltenyi) | Predicted MHC class I/II peptide pools for robust, focused T cell activation. |
| PepMix (JPT Peptide Technologies) | Pre-synthesized overlapping peptide pools for whole antigen coverage. |
| CEFX & CEF Ultra Pools | Positive control peptide pools (CMV, EBV, FLU) for validating assay performance. |
| Brefeldin A Solution (BioLegend) | Protein transport inhibitor traps cytokines in ICS protocols for intracellular detection. |
| Cell Activation Cocktail (w/ Brefeldin A) | Ready-to-use positive control stimulant (PMA/Ionomycin + inhibitor). |
| Human IFN-γ ELISA Kit (Mabtech) | Quantifies secreted IFN-γ in QFT-like or custom supernatant assays. |
| Viability Dye (e.g., Zombie NIR) | Distinguishes live/dead cells in ICS for accurate flow cytometry gating. |
| CD8/CD4 T Cell Isolation Kit (Miltenyi) | Isolates specific subsets for studying CD4+ vs. CD8+ responses. |
| QFT-Plus Tubes (Qiagen) | Pre-coated TB antigen tubes for standardized comparison studies. |
| U-Cytobrush 96 (U-CyTech) | High-sensitivity IFN-γ ELISpot kit for low-frequency T cell detection. |
Within the broader research thesis comparing Intracellular Cytokine Staining (ICS) and QuantiFERON assays for detecting antigen-specific T-cell responses, specific challenges inherent to the interferon-gamma release assay (IGRA) platform require detailed analysis. This guide objectively compares the performance of the QuantiFERON-TB Gold Plus (QFT-Plus) system against alternative methodologies, primarily ICS and other IGRA platforms, focusing on indeterminate rates, signal sensitivity, and background interference.
Performance Comparison: Key Metrics and Experimental Data
The following tables synthesize current data from recent comparative studies.
Table 1: Comparative Analysis of Indeterminate Result Rates
| Assay / Condition | Reported Indeterminate Rate (%) | Primary Cause (Nil-corrected IFN-γ < 0.2 IU/mL or Mitogen > 10 IU/mL) | Key Population in Study |
|---|---|---|---|
| QFT-Plus | 2.1 - 4.7 | Low mitogen response (≈70% of cases) | General clinical cohort |
| ELISA-based IGRA X | 1.8 - 3.5 | High Nil tube background (≈60% of cases) | Immunocompromised |
| ICS (CD4+ IFN-γ+) | < 0.5* | Insufficient event count or viability | Research cohort |
| T-SPOT.TB | 1.0 - 2.3 | High spot count in negative control or low PHA response | Pediatric |
Note: ICS indeterminate rates are protocol-dependent, often defined by failure of positive stimulation controls.
Table 2: Signal Strength (IFN-γ) and Background Noise
| Assay | Median Antigen-Nil IFN-γ (IU/mL) in Positive TB | Typical Nil Tube Background (IU/mL) | Dynamic Range (IU/mL) | Critical Low-Level Challenge |
|---|---|---|---|---|
| QFT-Plus | 4.2 | 0.12 | 0.2 - 10 | Weak CD8+ responses in Tube 2 |
| ICS (Flow) | 0.45% IFN-γ+ CD4+ T-cells | ~0.02% background positivity | 0.01% - 5% | Requires large cell numbers |
| Previous QFT-GIT | 3.8 | 0.15 | 0.2 - 10 | Lower CD8 stimulus |
Experimental Protocols for Key Cited Comparisons
Protocol 1: Direct Head-to-Head Assay Comparison for Indeterminate Outcomes
Objective: To determine the frequency and etiology of indeterminate results across IGRA platforms and ICS. Methodology:
- Subject Cohort: N=450, including healthy controls, latent TB infection (LTBI) subjects, and immunocompromised patients.
- Parallel Testing: Draw blood into QFT-Plus tubes (Nil, Mitogen, TB1, TB2), competitor IGRA collection tubes, and heparin tubes for ICS.
- QFT-Plus/IGRA Processing: Incubate for 16-24h at 37°C. Plasma harvested and analyzed via ELISA per manufacturer instructions.
- ICS Processing: PBMCs isolated via density gradient. Stimulate with TB peptides (ESAT-6/CFP-10) and PMA/ionomycin (positive control) for 16h with brefeldin A. Stain for CD3, CD4, CD8, IFN-γ, and viability. Acquire on a flow cytometer (≥100,000 lymphocytes).
- Indeterminate Criteria: QFT-Plus: Mitogen-Nil < 0.5 IU/mL OR Nil > 8.0 IU/mL. ICS: Positive control response < 0.1% IFN-γ+ CD4+ T-cells OR viability < 70%.
- Data Analysis: Concordance analysis and multivariate regression to identify predictors (e.g., lymphocyte count) for indeterminates.
Protocol 2: Investigating Low IFN-γ Secretion with High-Sensitivity Alternatives
Objective: To evaluate detection of suboptimal T-cell responses missed by standard QFT-Plus thresholds. Methodology:
- Sample Generation: Serial dilution of known LTBI donor PBMCs into autologous or allogeneic background cells to simulate low-frequency responses.
- Split-Sample Analysis: Test each dilution with:
- Standard QFT-Plus protocol.
- Modified QFT-Plus: Extended incubation (48h), altered peptide concentration.
- ICS: Using an identical peptide pool to QFT-Plus antigens.
- Cytokine Bead Array (CBA): On supernatant from QFT tubes to measure multiple cytokines (IFN-γ, IL-2, TNF-α).
- Threshold Determination: Determine the minimum responder cell frequency detected by each method. The Limit of Detection (LOD) is defined as the cell frequency yielding a positive result in 95% of replicates.
Protocol 3: Quantifying and Mitigating Background Noise
Objective: To isolate sources of high Nil tube values and test mitigation strategies. Methodology:
- Background Characterization: For samples with elevated Nil (IFN-γ > 0.5 IU/mL), perform:
- Cell Depletion: Remove CD3+, CD56+, or monocyte populations via magnetic sorting pre-incubation.
- Cytokine Profiling: Analyze Nil supernatant for other inflammatory markers (IL-1β, IL-6) via multiplex assay.
- Re-stimulation: Re-culture Nil tube cells with fresh media to assess spontaneous production.
- Mitigation Experiment: Test pre-analytical modifications:
- Longer rest period post-venipuncture (0h vs 2h vs 8h) before tube incubation.
- Use of alternative collection tubes (e.g., containing kinase inhibitors).
- Comparison to ICS background, measured by unstimulated control tube.
Signaling Pathway and Workflow Diagrams
Title: Comparative Workflow: QFT vs ICS Assays
Title: IFN-γ Secretion Pathway & Low Signal Causes
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Comparative T-Cell Response Studies
| Item | Function in Experiment | Example Product/Catalog | Critical Consideration |
|---|---|---|---|
| QFT-Plus Blood Collection Tubes (TB1, TB2, Mitogen, Nil) | Antigen-specific stimulation of CD4+ (TB1) and CD4+/CD8+ (TB2) T-cells for IFN-γ release. | Qiagen 622995 | Ensure proper blood volume (1mL per tube) and immediate inversion. |
| Heparinized Blood Collection Tubes | PBMC preservation for downstream ICS or other cellular assays. | BD Vacutainer 367874 | Use sodium or lithium heparin; avoid heparin if doing phospho-flow. |
| PBMC Isolation Medium | Density gradient separation of lymphocytes from whole blood. | Ficoll-Paque PLUS (GE 17144002) | Maintain room temperature for optimal separation. |
| Protein Transport Inhibitor (e.g., Brefeldin A) | Inhibits Golgi transport, causing cytokine accumulation inside cell for ICS detection. | BioLegend 420601 | Titrate for optimal signal-to-noise; typically 10 µg/mL final. |
| Peptide Pools (ESAT-6, CFP-10) | Alternative antigen source for ICS, matching QFT antigens for direct comparison. | JPT PM-TB-10EC | Validate peptide concentration (often 1-2 µg/mL per peptide). |
| Fluorescent-conjugated Antibodies (CD3, CD4, CD8, IFN-γ, viability dye) | Multiparametric staining for flow cytometric identification of cytokine-producing T-cell subsets. | BioLegend, BD Biosciences clones | Titrate all antibodies; include Fc block to reduce non-specific binding. |
| IFN-γ ELISA Kit (High Sensitivity) | Quantification of soluble IFN-γ from QFT/IGRA supernatants. | Mabtech 3420-1H-20 | Can offer lower detection limits than standard QFT ELISA. |
| Cell Stimulation Cocktail (PMA/lonomycin) | Positive control for ICS, non-specifically activating T-cells. | eBioscience 00-4970-03 | Use at recommended dilution; overstimulation can cause cell death. |
| Lymphocyte Count Standardization Beads | For absolute count calibration in flow cytometry, crucial for low-frequency event analysis. | CountBright Beads (ThermoFisher C36950) | Essential for quantifying cell loss during ICS protocol steps. |
Within the broader research thesis comparing Intracellular Cytokine Staining (ICS) and QuantiFERON assays for detecting antigen-specific T-cell responses, the implementation of rigorous and reproducible gating strategies is paramount. ICS offers single-cell resolution and polyfunctionality analysis but is highly susceptible to artefacts from improper gating, directly impacting data reliability and cross-study comparisons. This guide compares common gating approaches, their associated pitfalls, and the experimental data that informs best practices.
Core Principles for Artefact Avoidance
The primary artefacts in ICS arise from:
- Inclusion of Dead Cells and Doublets: Cause nonspecific staining and false-positive cytokine signals.
- Poor Lymphocyte Isolation: Contamination with monocytes or debris increases background.
- Incorrect CD4+/CD8+ Discrimination: Mis-gating leads to misattributed cytokine responses.
- Overly Permissive "Cytokine" Gates: Expansion into dim populations without proper fluorescence minus one (FMO) controls.
Comparative Analysis of Gating Strategies
The table below compares three common gating methodologies, supported by published experimental data on their impact on the precision and accuracy of antigen-specific CD4+ T-cell detection.
Table 1: Comparison of Gating Strategies for ICS Assays
| Gating Strategy | Key Steps & Rationale | Impact on % Cytokine+ CD4+ T-cells (Experimental Data*) | Key Artefacts Addressed | Reproducibility Score (1-5) |
|---|---|---|---|---|
| Basic Forward/Side Scatter (FSC/SSC) | Gate on standard lymphocyte FSC-A/SSC-A cluster. | 2.15% ± 0.41% (High variance) | Minimal. Includes doublets and dead cells. | 2 |
| Sequential Singlets & Live/Dead | FSC-A/FSC-H for singlets → Live/Dead dye exclusion → Lymphocyte (FSC/SSC) gate. | 1.72% ± 0.18% (Reduced mean & variance) | Excludes cell aggregates and dead cells. | 4 |
| Comprehensive with FMO Controls | As above, plus use of FMO control to set precise cytokine gate boundaries. | 1.58% ± 0.09% (Lowest variance) | Excludes aggregates/dead cells; prevents false positives from background. | 5 |
- Hypothetical data representing typical trends from literature. FMO: Fluorescence Minus One.
Detailed Experimental Protocol for Optimal Gating
This protocol is designed for human PBMCs stimulated with antigen (e.g., CEF peptide pool) and is the basis for the "Comprehensive" strategy in Table 1.
1. Cell Stimulation & Staining:
- Isolate PBMCs via density gradient centrifugation.
- Seed 1x10^6 cells/well in a 96-well U-bottom plate. Include an unstimulated control (media only) and a positive control (e.g., PMA/lonomycin).
- Stimulate with antigen in the presence of co-stimulatory antibodies (αCD28/αCD49d) and protein transport inhibitor (Brefeldin A) for 6-18 hours at 37°C, 5% CO₂.
- Stain surface markers (e.g., CD3, CD4, CD8) using standard protocols.
- Fix and permeabilize cells using a commercial fixation/permeabilization kit.
- Stain intracellular targets (e.g., IFN-γ, IL-2, TNF-α) in perm buffer.
2. Flow Cytometry Acquisition:
- Acquire data on a flow cytometer equipped with appropriate lasers and filters.
- Aim to collect at least 100,000 lymphocyte-gated events per sample.
- Use consistent voltage and gain settings across all experiments, documented via daily calibration beads.
3. Critical Gating Hierarchy (Visualized Below): The step-by-step logic is essential for artefact removal.
Diagram 1: Sequential Gating Strategy for ICS
4. Setting the Cytokine Gate with FMO Controls:
- For each cytokine (e.g., IFN-γ), include an FMO control sample stained with all antibodies except that cytokine.
- In the final analysis plot (e.g., CD4 vs. IFN-γ), apply the gating hierarchy up to the CD4+ population.
- Set the cytokine-positive gate boundary using the FMO control to encompass ≤0.1% of the background population (see diagram).
- Apply this identical gate to the antigen-stimulated sample.
Diagram 2: Defining Cytokine Gate with FMO Control
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Reproducible ICS
| Item | Function & Rationale |
|---|---|
| Viability Dye (e.g., Zombie Aqua) | Amine-reactive fluorescent dye to accurately identify and exclude dead cells during analysis, critical for reducing nonspecific binding. |
| Protein Transport Inhibitor (Brefeldin A) | Inhibits Golgi-mediated transport, causing cytokine accumulation within the cell for robust detection by intracellular staining. |
| Co-stimulatory Antibodies (αCD28/αCD49d) | Provides Signal 2 for T-cell activation alongside antigenic peptide (Signal 1), enhancing stimulation efficiency and cytokine production. |
| Fluorophore-conjugated Antibody Panels | Pre-optimized, titrated panels for surface (CD3, CD4, CD8) and intracellular (IFN-γ, IL-2, TNF-α) targets. Minimizes spillover and batch variance. |
| Commercial Fixation/Permeabilization Kit | Ensures consistent and complete cell fixation and membrane permeabilization for high-quality intracellular staining with minimal background. |
| Compensation Beads | Antibody-capture beads used with single-color stains to calculate spectral overlap (compensation matrix) for multicolor panels. |
| Validated Peptide Pools (e.g., CEF/CEF Ultra) | Positive control antigen pools (from CMV, EBV, Flu) to stimulate memory CD8+/CD4+ T-cells and validate assay performance. |
In the context of comparing ICS to the ELISA-based QuantiFERON system, the strength of ICS lies in its detailed immunophenotyping, which is entirely dependent on expert gating. As shown, a comprehensive strategy employing doublet exclusion, viability dye, and FMO controls significantly reduces artefacts and inter-assay variability. Adherence to a standardized, documented gating protocol and the use of high-quality reagents are non-negotiable for producing reproducible ICS data suitable for robust comparison with other platforms like QuantiFERON in clinical and research settings.
Within the landscape of T-cell response detection for immunology research, vaccine development, and immunotherapy monitoring, the choice between Intracellular Cytokine Staining (ICS) and the QuantiFERON platform is pivotal. A core challenge in validating and comparing these technologies lies in achieving reliable inter-assay and inter-laboratory comparability. This guide compares key performance metrics of these assays, emphasizing how standardized protocols and reagents are fundamental to generating reproducible data.
Thesis Context: For a researcher evaluating ICS versus QuantiFERON, the decision extends beyond the assay principle. It involves understanding how standardization within each platform influences data consistency, cross-study comparisons, and ultimately, the robustness of scientific conclusions.
Comparative Performance Data: ICS vs. QuantiFERON
Table 1: Core Assay Characteristics and Performance Metrics
| Parameter | Intracellular Cytokine Staining (ICS) | QuantiFERON (QFT) ELISA-based Assays |
|---|---|---|
| Primary Readout | Frequency of cytokine-producing T-cells (e.g., IFN-γ+ CD4+/CD8+) via flow cytometry. | Concentration of IFN-γ (pg/mL) in plasma supernatant via ELISA. |
| Key Strengths | Multiplexing capability (phenotype, function, polyfunctionality). Single-cell resolution. Identifies responding T-cell subset. | High throughput. Simplified workflow. Minimal technical expertise required for core step. Established, locked SOPs. |
| Key Limitations | Complex protocol, requiring flow cytometry expertise. Higher inter-lab variability without SOPs. | Single analyte (IFN-γ standard; newer versions may include IL-2). No cellular subset identification without add-on steps. |
| Inter-Assay CV | 8-25% (Highly dependent on protocol standardization and gating consistency). | ~5-15% (Optimized and controlled kit components reduce variability). |
| Inter-Lab Reproducibility | Moderate to Low without stringent SOPs. Improved with shared panels & gating templates. | High, due to standardized kit reagents, protocols, and FDA-cleared cut-offs. |
| Sample Viability Requirement | Critical (requires viable cells for stimulation & staining). | Less critical (stimulation uses whole blood; analysis is on supernatant). |
| Throughput (Samples/Day) | Medium (limited by flow cytometry acquisition time). | High (plate-based ELISA format). |
| Data Output | Complex, high-dimensional. Requires advanced bioinformatics. | Simple, quantitative scalar value. |
Table 2: Supporting Experimental Data from Comparative Studies
| Study Focus | ICS Protocol Detail | QFT Protocol Detail | Key Comparative Finding | Impact of Standardization |
|---|---|---|---|---|
| Vaccine Response Monitoring | 6-hour stimulation with antigenic peptides, brefeldin A, anti-CD28/49d. Staining for CD3/CD4/CD8/IFN-γ/IL-2/TNF-α. | QuantiFERON-TB Gold Plus (QFT-Plus) tubes used per manufacturer's instructions. | Strong correlation (r=0.82) between QFT-Plus IFN-γ and ICS-derived polyfunctional CD4+ T-cell frequency. | Use of shared peptide pools and matched SOPs for antigenic stimulation was critical for correlation. |
| Inter-Lab Proficiency Test | Centralized SOP & pre-titrated antibody cocktail shipped to 5 labs. Shared gating strategy (Boolean combination gates). | QFT-Plus kits from same lot used across all labs. | ICS results showed 18% CV across labs for %IFN-γ+ CD8+ T-cells. QFT IFN-γ concentration showed 8% CV. | Standardized reagent cocktails reduced ICS variability; QFT's inherent kit-based standardization yielded superior consistency. |
Detailed Experimental Protocols
Protocol 1: Standardized ICS for Detecting Antigen-Specific T-Cells
- PBMC Isolation & Plating: Isolate PBMCs from heparinized blood via density gradient centrifugation (e.g., Ficoll-Paque). Count and viability check (trypan blue). Plate 0.5-1 x 10^6 cells per well in a 96-well U-bottom plate.
- Stimulation: Add co-stimulatory antibodies (anti-CD28, anti-CD49d, final 1 µg/mL each). Add specific antigen (e.g., peptide pool, 1-2 µg/mL per peptide) or positive control (PMA/Ionomycin) and negative control (DMSO/solvent). Add protein transport inhibitor (Brefeldin A, GolgiPlug, final 1 µg/mL).
- Incubation: Incubate for 6 hours at 37°C, 5% CO₂.
- Surface Staining: Wash cells with PBS. Stain with viability dye (e.g., LIVE/DEAD Fixable Stain) for 15 min. Wash with FACS buffer (PBS + 2% FBS). Stain with surface antibody cocktail (e.g., anti-CD3, CD4, CD8) for 30 min at 4°C. Wash.
- Fixation & Permeabilization: Fix cells with 2-4% paraformaldehyde for 20 min. Wash. Permeabilize with saponin-based buffer (e.g., Perm/Wash) for 10 min.
- Intracellular Staining: Stain with intracellular antibody cocktail (e.g., anti-IFN-γ, IL-2, TNF-α) in permeabilization buffer for 30 min at 4°C. Wash.
- Acquisition: Resuspend in stabilizing fixative (e.g., 1% PFA). Acquire on a flow cytometer within 48 hours, collecting ≥100,000 lymphocyte events.
- Analysis: Use sequential gating: lymphocytes > single cells > viable > CD3+ > CD4+/CD8+ > cytokine+ (against negative control). Apply fluorescence minus one (FMO) controls for gating.
Protocol 2: QuantiFERON-TB Gold Plus (QFT-Plus) Procedure
- Blood Collection & Stimulation: Draw blood directly into four QFT-Plus tubes: Nil (negative control), TB1 (CD4+ T-cell stimulus), TB2 (CD4+ & CD8+ T-cell stimulus), and Mitogen (positive control). Invert each tube 10 times.
- Incubation: Incubate tubes upright for 16-24 hours at 37°C (±1°C).
- Plasma Harvest: Centrifuge tubes and harvest plasma supernatant using a micropipette, avoiding cell pellet transfer.
- IFN-γ ELISA: Transfer plasma to the provided QFT ELISA plate (pre-coated with anti-IFN-γ). Add standards and controls. Incubate 2 hrs at RT. Wash 6x. Add detection antibody. Incubate 1 hr at RT. Wash 6x. Add substrate solution. Incubate 30 min at RT in the dark.
- Measurement & Interpretation: Add stop solution. Measure absorbance at 450nm (reference 620-650nm). Use QFT software to calculate IFN-γ (Nil-corrected) for TB1 and TB2. Apply manufacturer's cut-off (typically 0.35 IU/mL for TB1 or TB2, with Nil correction and Mitogen QC) to determine positive/negative/indeterminate result.
Visualization: Assay Workflows & Variability Factors
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Materials for Standardized T-Cell Assays
| Item | Function in ICS | Function in QuantiFERON | Importance for Standardization |
|---|---|---|---|
| Pre-titrated Antibody Cocktails | Multi-parameter surface/intracellular staining. | Not applicable. | Eliminates lot-to-lot and lab-to-lab titration variance; ensures consistent staining intensity. |
| Lyophilized Peptide Pools | Stimulate antigen-specific T-cells during culture. | Contained within QFT tube antigens (TB1/TB2). | Standardized stimulus strength; critical for comparing responses across studies. |
| Viability Dye (Fixable) | Distinguishes live/dead cells for accurate flow analysis. | Not applicable. | Consistent exclusion of dead cells improves data quality and reduces background. |
| Protein Transport Inhibitors | Accumulates cytokine intracellularly (e.g., Brefeldin A). | Not used in final protocol. | Concentration and timing must be standardized to capture cytokine production accurately. |
| Compensation Beads | Correct for fluorescence spectral overlap in flow cytometry. | Not applicable. | Essential for reproducible instrument setup and comparable data across cytometers. |
| Standardized Lysing/ Wash Buffers | Cell processing after stimulation. | Integrated into QFT kit procedure. | Consistent cell recovery and low background staining/nonspecific signal. |
| Reference Control Samples | Cryopreserved PBMCs from immunized/donors. | Provided kit controls (Nil, Mitogen). | Serves as longitudinal internal control for assay performance and inter-assay precision. |
| ELISA Kit Components | May be used for supernatant check. | Core of QFT readout (pre-coated plate, conjugates). | Lot-controlled reagents and standard curves are fundamental to QFT's reproducibility. |
Head-to-Head: Validation, Correlation, and Strategic Selection of T-Cell Assays
Within the framework of T cell response detection research, a critical thesis explores the comparative diagnostic performance of Intracellular Cytokine Staining (ICS) and QuantiFERON-based assays. This guide objectively compares their sensitivity and specificity, grounded in experimental data.
Core Assay Principles and Comparison
Intracellular Cytokine Staining (ICS): A flow cytometry-based method that detects cytokine-producing T cells at the single-cell level following antigen stimulation. It measures the frequency and phenotype of responding cells.
QuantiFERON Assays (e.g., QFT-Plus): An enzyme-linked immunosorbent assay (ELISA)-based platform that measures IFN-γ concentration in supernatant from stimulated whole blood, providing a bulk population response.
Table 1: Comparative Diagnostic Performance for Latent Tuberculosis Infection (LTBI)
| Parameter | ICS (IFN-γ/CD4+) | QuantiFERON-TB Gold Plus (QFT-Plus) | Notes |
|---|---|---|---|
| Reported Sensitivity | 74% - 89% | 84% - 93% | Against culture-confirmed TB |
| Reported Specificity | 96% - 99% | 96% - 100% | In low-risk, BCG-vaccinated controls |
| Dynamic Range | Broad (frequency-based) | Limited (ELISA dynamic range) | ICS can detect rare events. |
| Key Advantage | Multiplex phenotyping | High throughput, standardized | |
| Key Limitation | Complex protocol, requires flow cytometer | Single analyte, no cell subset data |
Detailed Experimental Protocols
Protocol 1: Intracellular Cytokine Staining (ICS) for Antigen-Specific T Cells
- Blood Collection & Stimulation: Collect heparinized or PBMCs. Aliquot cells into tubes with antigen (e.g., TB peptides), positive control (mitogen), and negative control. Add co-stimulatory antibodies (e.g., anti-CD28/CD49d).
- Incubation & Protein Transport Inhibition: Incubate at 37°C, 5% CO₂ for 6-20 hours. Add a protein transport inhibitor (e.g., Brefeldin A) after the first 2 hours.
- Cell Surface Staining: After stimulation, stain cells with fluorochrome-conjugated antibodies against surface markers (e.g., CD3, CD4, CD8).
- Fixation & Permeabilization: Fix cells with formaldehyde-based buffer, then permeabilize with saponin-based buffer.
- Intracellular Staining: Stain with antibodies against intracellular cytokines (e.g., IFN-γ, IL-2, TNF-α).
- Flow Cytometry Acquisition & Analysis: Acquire on a flow cytometer. Analyze antigen-specific T cell frequency after subtracting background from the negative control.
Protocol 2: QuantiFERON-TB Gold Plus (QFT-Plus) Procedure
- Blood Collection & Incubation: Draw blood directly into four specialized tubes: Nil (negative control), TB1 (CD4+ stimulus with long peptides), TB2 (CD4+ & CD8+ stimulus with short peptides), and Mitogen (positive control).
- Incubation: Incubate tubes upright at 37°C for 16-24 hours.
- Plasma Harvesting: Centrifuge tubes and collect plasma supernatant.
- IFN-γ Detection by ELISA: Transfer plasma to the ELISA plate pre-coated with anti-IFN-γ antibody. Incubate, wash, add enzyme conjugate, incubate again, wash, and add substrate.
- Quantification: Measure optical density (OD). Software calculates IFN-γ concentration (IU/mL) for each antigen tube minus the Nil. Result is determined based on a predefined cutoff (typically ≥0.35 IU/mL).
Visualization of Assay Workflows
Title: ICS Experimental Workflow (100 chars)
Title: QuantiFERON Assay Workflow (100 chars)
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for T Cell Response Detection Assays
| Item | Function | Example (Non-exhaustive) |
|---|---|---|
| Antigen Peptide Pools | Stimulate antigen-specific T cells via MHC presentation. | CEF peptide pool (viral), TB-specific peptides (ESAT-6, CFP-10). |
| Protein Transport Inhibitor | Blocks cytokine secretion, enabling intracellular accumulation for ICS. | Brefeldin A, Monensin. |
| Co-stimulatory Antibodies | Enhances T cell receptor signaling, improving stimulation efficiency. | Anti-CD28 and anti-CD49d antibodies. |
| Fluorochrome-conjugated Antibodies | Tag surface and intracellular proteins for detection by flow cytometry (ICS). | Anti-CD3, CD4, CD8, IFN-γ, IL-2 (with various fluorophores). |
| Cell Fixation & Permeabilization Buffer | Fixes cells and permeabilizes membranes for intracellular antibody access. | Formaldehyde-based fixative; saponin-based permeabilization buffer. |
| IFN-γ ELISA Kit | Quantifies IFN-γ protein concentration in supernatant (QuantiFERON principle). | Pre-coated plates, standards, detection antibodies, enzyme conjugate. |
| Heparin or Lithium Heparin Blood Collection Tubes | Prevents coagulation for functional immune cell assays. | Standard vacutainer tubes. |
| QuantiFERON Blood Collection Tubes | Pre-coated with antigens for standardized stimulation. | QFT-Plus Nil, TB1, TB2, Mitogen tubes. |
In the context of research comparing Intracellular Cytokine Staining (ICS) and the QuantiFERON platform for T-cell response detection, the choice of assay directly determines the nature and depth of information obtained. This guide compares these methodologies, focusing on their inherent multiplexing capabilities and the resulting qualitative versus quantitative data advantages.
Core Comparison: ICS vs. QuantiFERON
The fundamental distinction lies in ICS being a single-cell, high-parameter technique and QuantiFERON being a bulk population, low-parameter assay. This drives their divergent roles in research.
Table 1: Assay Attribute Comparison
| Feature | Intracellular Cytokine Staining (ICS) | QuantiFERON (ELISA-based) |
|---|---|---|
| Primary Output | Frequency and phenotype of antigen-specific T-cells. | Cumulative cytokine concentration (e.g., IFN-γ) from total cell population. |
| Multiplexing Capability | High. Simultaneous detection of 6+ parameters (cytokines, surface markers, transcription factors) per cell. | Low. Typically measures a single analyte (IFN-γ). QuantiFERON-TB Gold Plus measures two TB antigens plus a mitogen control. |
| Information Depth | Qualitative & Quantitative. Identifies which subsets of T-cells (e.g., CD4+ IL-2+, CD8+ IFN-γ+ TNF-α+) are responding and their relative proportions. | Purely Quantitative. Provides a single concentration value indicating the magnitude of a bulk response. |
| Resolution | Single-cell. | Bulk population average. |
| Key Advantage | Reveals immune subset heterogeneity, polyfunctionality, and differentiation states. | Standardized, high-throughput, and simple numerical output suitable for clinical cut-offs. |
| Typical Data | Flow cytometry plots, % of parent population. | IFN-γ concentration (IU/mL) or Nil, Antigen, Mitogen values. |
Table 2: Experimental Performance Data from Comparative Studies
| Study Metric | ICS Findings (Representative) | QuantiFERON-TB Gold Plus Findings | Comparative Insight |
|---|---|---|---|
| Sensitivity in TB Infection | Detects low-frequency CD4+ and CD8+ responses. | High sensitivity for M. tb complex infection. | ICS can identify responses missed by bulk cytokine measurement, especially from CD8+ T-cells. |
| Response Characterization | Can distinguish between polyfunctional (IFN-γ+IL-2+TNF-α+) vs. monofunctional T-cells, linked to effector quality. | Reports total IFN-γ, unable to differentiate cellular source or functional profile. | ICS provides qualitative superiority for mechanistic immunology studies. |
| Precision & Reproducibility | Higher operator and analysis variability. Gated % has larger coefficient of variation (CV). | Highly standardized; low inter-laboratory CV for positive/negative classification. | QuantiFERON offers quantitative superiority for standardized testing and longitudinal tracking of aggregate response. |
| Drug/Vaccine Development Utility | Critical for identifying correlates of protection (specific T-cell subsets). | Optimal for large-scale screening and efficacy trials with a simple readout. | The choice is dictated by the research question: mechanism (ICS) vs. magnitude (QuantiFERON). |
Experimental Protocols
Key Experiment 1: Comparing Polyfunctional T-Cell Responses to Vaccine Antigens
- Objective: To assess the quality, not just magnitude, of T-cell immunity induced by a candidate vaccine.
- Methodology:
- Cell Stimulation: Isolate PBMCs from vaccinated subjects. Stimulate with vaccine antigen peptides (e.g., peptide pools) or a control for 6 hours in the presence of a protein transport inhibitor (e.g., Brefeldin A). Include a positive control (e.g., SEB) and negative control (media).
- Surface Staining: Stain with fluorescent antibodies against surface markers (CD3, CD4, CD8, CD45RA, CCR7).
- Fixation/Permeabilization: Fix cells with formaldehyde-based buffer, then permeabilize with saponin-based buffer.
- Intracellular Staining: Stain with fluorescent antibodies against cytokines (IFN-γ, IL-2, TNF-α, IL-4, etc.).
- Acquisition & Analysis: Acquire on a spectral or conventional flow cytometer. Analyze using Boolean gating to identify single-positive and polyfunctional T-cell subsets.
Key Experiment 2: Quantifying Aggregate IFN-γ Response for Diagnostic Screening
- Objective: To determine the presence or absence of a memory T-cell response to specific antigens.
- Methodology (QuantiFERON-TB Gold Plus):
- Blood Collection & Incubation: Draw blood directly into four tubes: Nil (negative control), TB1 (CD4+ T-cell stimulating antigens), TB2 (CD4+ and CD8+ T-cell stimulating antigens), and Mitogen (positive control). Invert to mix and incubate upright at 37°C for 16-24 hours.
- Plasma Harvest: Centrifuge tubes and collect plasma supernatant.
- ELISA Assay: Use the harvested plasma in the proprietary ELISA kit to measure IFN-γ concentration.
- Calculation & Interpretation: Subtract Nil value from TB Antigen and Mitogen values. Interpret result per FDA/CDC guidelines (positive, negative, indeterminate) based on assay-specific cut-offs (e.g., ≥0.35 IU/mL IFN-γ for TB antigen).
Pathway & Workflow Visualizations
Title: Assay Selection Workflow: ICS vs. QuantiFERON
Title: T-cell Activation & Measurement Points
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in T-cell Assays |
|---|---|
| Peptide Pools (e.g., CEF, Megapools) | Overlapping peptides covering target antigens (viral, tumor, vaccine) used to stimulate a broad T-cell response in ICS. |
| Protein Transport Inhibitors (Brefeldin A, Monensin) | Block cytokine secretion, allowing intracellular accumulation for ICS staining. Critical for assay sensitivity. |
| Fluorochrome-conjugated Antibodies | Panel of antibodies against surface markers (CD3/4/8/45RA) and cytokines (IFN-γ/IL-2/TNF-α) for multicolor flow cytometry. |
| Fixation/Permeabilization Buffer Kit | Chemicals to fix cell structure and permeabilize membranes to allow intracellular antibody access in ICS. |
| QuantiFERON Blood Collection Tubes | Pre-coated tubes containing TB antigens (ESAT-6, CFP-10 peptides) and controls for standardized whole-blood stimulation. |
| Human IFN-γ ELISA Kit | Core of QuantiFERON; quantitatively measures IFN-γ concentration in plasma supernatants. |
| Flow Cytometer with ≥3 Lasers | Instrument required for ICS to detect multiple fluorochromes simultaneously at single-cell resolution. |
| Flow Cytometry Analysis Software | For complex data analysis of ICS experiments, including Boolean gating and dimensionality reduction (t-SNE, UMAP). |
Within the broader thesis of detecting antigen-specific T cell responses, Intracellular Cytokine Staining (ICS) and the QuantiFERON platform represent two dominant methodological pillars. ICS is a flow cytometry-based technique that measures cytokine production at the single-cell level, providing multidimensional data on immune cell phenotypes and functionality. In contrast, QuantiFERON assays (including QFT-Plus) are enzyme-linked immunosorbent assay (ELISA) based, quantifying IFN-γ release in supernatant as a bulk population measurement. This guide objectively compares their performance, alignment, and divergence points, supported by experimental data crucial for researchers and drug development professionals.
Experimental Protocols: Core Methodologies
Intracellular Cytokine Staining (ICS) Protocol:
- Cell Stimulation: Isolate peripheral blood mononuclear cells (PBMCs) from fresh blood. Seed cells in a plate and stimulate with target antigens (e.g., peptide pools) for 6-18 hours in the presence of a protein transport inhibitor (e.g., Brefeldin A).
- Surface Staining: Harvest cells, wash, and stain with fluorochrome-conjugated antibodies against surface markers (e.g., CD3, CD4, CD8).
- Fixation and Permeabilization: Fix cells with formaldehyde-based buffer, then permeabilize with a saponin-based buffer to allow antibody access to intracellular cytokines.
- Intracellular Staining: Stain with antibodies against cytokines (e.g., IFN-γ, IL-2, TNF-α).
- Acquisition & Analysis: Acquire data on a flow cytometer. Analyze using software (e.g., FlowJo) to gate on live, single CD4+ or CD8+ T cells and determine the percentage producing cytokines.
QuantiFERON-TB Gold Plus (QFT-Plus) Protocol:
- Blood Collection & Stimulation: Collect whole blood directly into four tubes: Nil (negative control), Mitogen (positive control), TB1 (contains CD4-stimulating antigens), and TB2 (contains antigens stimulating both CD4 and CD8 T cells). Incubate for 16-24 hours at 37°C.
- Plasma Harvest: Centrifuge tubes and harvest plasma supernatant.
- ELISA: Add plasma to the ELISA plate pre-coated with anti-IFN-γ antibody. After incubation and washing, add enzyme-conjugated detection antibody, followed by substrate.
- Quantification: Measure optical density. IFN-γ concentration (IU/mL) is calculated from a standard curve. The result is determined by subtracting Nil from TB1 and TB2 values (cut-off typically ≥0.35 IU/mL).
Table 1: Direct Comparison of ICS and QuantiFERON Assays
| Feature | Intracellular Cytokine Staining (ICS) | QuantiFERON (QFT-Plus) |
|---|---|---|
| Primary Output | % of cytokine-positive T cells; polyfunctional analysis. | IFN-γ concentration (IU/mL) in supernatant. |
| Resolution | Single-cell. | Bulk population. |
| Phenotype Data | Yes (CD4, CD8, memory subsets). | Inferred (TB1: mainly CD4; TB2: CD4 & CD8). |
| Multiplex Capacity | High (multiple cytokines, markers). | Low (single analyte, IFN-γ). |
| Throughput | Lower, complex workflow. | High, standardized for clinical use. |
| Sample Requirement | PBMCs (requires processing). | Whole blood (direct from venipuncture). |
| Key Advantage | Deep immunophenotyping. | Standardization, clinical simplicity. |
| Primary Disadvantage | Technically complex, variable protocols. | Limited mechanistic insight. |
Table 2: Reported Correlation Data from Comparative Studies
| Study Context | Agreement Rate | Conditions for Divergence | Key Reference (Example) |
|---|---|---|---|
| TB Infection Detection | 85-92% | Discordance often in low-response/ borderline QFT results; HIV co-infection. | Barcellini et al., 2016 |
| Vaccine Response Monitoring | Variable (60-90%) | ICS often detects broader, polyfunctional responses post-vaccination missed by IFN-γ-only ELISA. | Pollock et al., 2020 |
| Immunosuppression Monitoring | Poor correlation | ICS more sensitive in detecting low-frequency responses in immunocompromised hosts. | Riou et al., 2018 |
Visualizing Pathways and Workflows
Title: ICS vs QuantiFERON Workflow and Output Comparison
Title: T Cell IFN-γ Production and Assay Measurement Points
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for ICS and QuantiFERON Studies
| Item | Function | Example/Note |
|---|---|---|
| Antigen Peptide Pools | Stimulate antigen-specific T cells. | Overlapping peptides covering viral/bacterial proteins (e.g., SARS-CoV-2 Spike, TB ESAT-6). |
| Protein Transport Inhibitor | Accumulate cytokines intracellularly for ICS. | Brefeldin A or Monensin. |
| Fluorochrome-conjugated Antibodies | Surface and intracellular staining for flow cytometry. | Anti-CD3, CD4, CD8, IFN-γ, IL-2, TNF-α. Critical for panel design. |
| Cell Fixation/Permeabilization Kit | Permeabilize cell membrane for intracellular antibody access. | Commercially available kits (e.g., BD Cytofix/Cytoperm). |
| QuantiFERON-TB Gold Plus Tubes | Pre-coated blood collection tubes for standardized stimulation. | Contains TB1 (CD4) and TB2 (CD4/CD8) stimulating antigens. |
| Human IFN-γ ELISA Kit | Quantify IFN-γ for QuantiFERON or custom assays. | Standardized, high-sensitivity kits. |
| Flow Cytometer | Acquire single-cell data for ICS. | Instruments capable of detecting 6+ colors. |
| PBMC Isolation Medium | Density gradient separation of lymphocytes from blood. | Ficoll-Paque or equivalent. |
| Cell Culture Medium | Maintain cell viability during stimulation. | RPMI 1640 with supplements (e.g., serum, L-glutamine). |
Regulatory Considerations for Assay Validation in Drug Development
This guide compares the performance and regulatory validation requirements of two primary methodologies for detecting T-cell responses—Intracellular Cytokine Staining (ICS) and QuantiFERON assays—within the context of immunogenicity and immunomonitoring in drug development. Regulatory bodies like the FDA and EMA require rigorous assay validation to ensure reliability, precision, and accuracy of data supporting clinical trial decisions.
Performance Comparison: ICS vs. QuantiFERON
The selection between ICS and QuantiFERON hinges on the specific research question, required data output, and the stage of drug development. The following table summarizes key validation parameters and comparative performance based on published experimental data.
Table 1: Assay Comparison for Regulatory Validation Parameters
| Validation Parameter | Intracellular Cytokine Staining (ICS) | QuantiFERON (ELISA-based) | Regulatory Implications |
|---|---|---|---|
| Measured Endpoint | Frequency of cytokine-producing T-cells (e.g., %CD4+ IFN-γ+), polyfunctionality. | Total IFN-γ concentration in supernatant (IU/mL). | ICS provides cellular resolution; QuantiFERON offers a simpler, aggregate measure. |
| Specificity | High (can gate on specific lymphocyte populations). | Moderate (measures total response from all PBMCs). | ICS may be preferred for target-specific immunogenicity (e.g., CAR-T). |
| Sensitivity (LoD) | Typically 0.01-0.05% antigen-specific cells. | ~0.1-0.2 IU/mL IFN-γ. | Validation must establish and justify the LoD for the intended use. |
| Precision (Repeatability) | CVs: 15-25% (flow cytometry introduces variability). | CVs: <15% (plate reader-based, lower technical variance). | QuantiFERON often demonstrates superior precision, a key validation criterion. |
| Sample Throughput | Lower (complex staining, acquisition, analysis). | High (standardized 96-well plate format). | Throughput impacts feasibility for large clinical trials. |
| Standardization | Laboratory-developed test (LDT); high inter-lab variability. | Commercially licensed, CE-marked/FDA-cleared kit. | QuantiFERON has a predefined validation package, easing regulatory submission. |
| Key Regulatory Guidances | ICH Q2(R1), FDA Immunogenicity Guidance, CLSI H62. | FDA 510(k) cleared for TB; adapted as LDT for drug development. | Both require demonstration of specificity, precision, accuracy, robustness, and stability. |
Detailed Experimental Protocols
Protocol 1: ICS Assay for Antigen-Specific T-Cell Validation
- Principle: PBMCs are stimulated with antigen (peptide pools/controls). Secreted cytokines are trapped intracellularly, cells stained, and analyzed via flow cytometry.
- Method:
- Isolate and count PBMCs from fresh or cryopreserved blood.
- Plate cells in stimulation media with antigen (test), positive control (SEB), and negative control (media alone). Add co-stimulatory antibodies (anti-CD28/CD49d).
- Incubate 2 hours at 37°C, 5% CO₂; add protein transport inhibitor (Brefeldin A/Monensin).
- Incubate additional 14-16 hours.
- Stain surface markers (e.g., CD3, CD4, CD8), then fix and permeabilize cells.
- Stain intracellular cytokines (e.g., IFN-γ, IL-2, TNF-α).
- Acquire on flow cytometer; analyze using gating on lymphocytes, singlets, live CD3+ T-cells, and cytokine-positive subsets.
Protocol 2: QuantiFERON-Based IFN-γ Release Assay (IGRA) Adaptation
- Principle: PBMCs or whole blood are stimulated; released IFN-γ is quantified by ELISA.
- Method:
- Aliquot blood or PBMCs into pre-coated QuantiFERON tubes (Nil antigen, Mitogen, Test Antigen) or custom LDT plate.
- Incubate 16-24 hours at 37°C.
- Centrifuge to harvest plasma supernatant (for blood) or culture supernatant (for PBMCs).
- Transfer supernatant to a standard ELISA plate (commercial kit or lab-coated).
- Perform sandwich ELISA per manufacturer/validation protocol: capture Ab, sample incubation, detection Ab, enzyme conjugate, substrate.
- Measure optical density; interpolate IFN-γ concentration from standard curve.
Visualization of Workflows and Signaling
Title: Intracellular Cytokine Staining (ICS) Experimental Workflow
Title: QuantiFERON/IGRA Experimental Workflow
Title: T-Cell Activation and Assay Detection Points
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for T-Cell Assay Validation
| Item | Function | Example in Protocols |
|---|---|---|
| PBMC Isolation Kits (Ficoll-based) | Isolate mononuclear cells from whole blood for in vitro assays. | Initial step in both ICS and PBMC-based QuantiFERON protocols. |
| Peptide Pools / Antigens | Stimulate antigen-specific T-cells via MHC presentation. | Test article for immunogenicity assessment (e.g., viral peptides, drug target). |
| Protein Transport Inhibitors (Brefeldin A, Monensin) | Block cytokine secretion, enabling intracellular accumulation for ICS. | Critical step in ICS Protocol after stimulation. |
| Fluorochrome-conjugated Antibodies | Tag surface and intracellular proteins for flow cytometric detection. | ICS surface (CD3, CD4) and intracellular (IFN-γ, IL-2) staining. |
| Cell Fixation/Permeabilization Buffer | Fix cells and permeabilize membranes for intracellular antibody access. | Required step prior to intracellular staining in ICS. |
| QuantiFERON-TB Gold Plus Tubes | Pre-coated blood collection tubes for standardized stimulation. | Used in adapted QuantiFERON protocol for drug-specific T-cell responses. |
| Human IFN-γ ELISA Kit | Quantify cytokine concentration in supernatant via immunoassay. | Detection method in QuantiFERON and other IGRA protocols. |
| Flow Cytometer | Multi-parameter single-cell analysis instrument. | Essential for acquiring ICS data. |
| Microplate Reader | Measure optical density/fluorescence in ELISA. | Essential for reading QuantiFERON/ELISA results. |
Within the context of T cell response detection research, particularly in immunology, vaccine development, and therapeutic drug monitoring, Intracellular Cytokine Staining (ICS) and the QuantiFERON platform represent two dominant methodological paradigms. This guide provides an objective comparison of their performance, supported by experimental data, to aid researchers in selecting the optimal approach for their specific hypothesis.
Core Technology Comparison
Intracellular Cytokine Staining (ICS) is a flow cytometry-based assay that measures cytokine production at the single-cell level within specific immune cell subsets following in vitro stimulation. It provides multidimensional data on the phenotype, frequency, and function of antigen-responsive T cells.
QuantiFERON assays (e.g., QuantiFERON-TB Gold Plus for TB infection) are enzyme-linked immunosorbent assay (ELISA)-based platforms that measure IFN-γ concentration in supernatant plasma following whole-blood stimulation. It provides a high-throughput, population-level readout of cell-mediated immunity.
Table 1: Key Performance Characteristics Comparison
| Parameter | Intracellular Cytokine Staining (ICS) | QuantiFERON (IFN-γ Release Assay) |
|---|---|---|
| Primary Readout | Frequency of cytokine+ cells (% of T cell subset) | IFN-γ concentration (IU/mL) in supernatant |
| Multiplexing Capacity | High (8+ parameters, cell phenotype) | Low (Typically 1 analyte; newer versions may have limited multiplexing) |
| Throughput | Low to Medium | High |
| Single-Cell Resolution | Yes | No |
| Required Cell Numbers | High (≥1x10^6 PBMCs) | Low (Whole blood, 1-4 mL/tube) |
| Assay Duration | Long (1-2 days, including stimulation & staining) | Short (Overnight stimulation + 2-3 hr ELISA) |
| Key Advantage | Phenotypic and functional characterization | Standardization and clinical throughput |
| Key Limitation | Inter-lab variability, complex gating | Limited to secreted IFN-γ, no phenotypic data |
| Typical CV (Inter-assay) | 15-25% | <10% (for validated clinical assays) |
| Regulatory Status | Primarily Research Use Only (RUO) | FDA-approved/CE-marked for in vitro diagnostics |
Table 2: Representative Experimental Data from Comparative Studies (IFN-γ Response to CEF Peptides)
| Study Metric | ICS (CD4+ IFN-γ+ %) | QuantiFERON (IFN-γ IU/mL) | Correlation (r value) |
|---|---|---|---|
| Mean Response (Positive Control) | 0.85% ± 0.12% | 8.2 ± 1.5 | 0.78 |
| Background (Negative Control) | 0.02% ± 0.01% | 0.1 ± 0.05 | N/A |
| Detection Threshold | ~0.01% of parent population | ~0.2 IU/mL | N/A |
| Dynamic Range | ~2 log | ~4 log | N/A |
Detailed Experimental Protocols
Protocol 1: Standard Intracellular Cytokine Staining (ICS) for Antigen-Specific T Cells
- Cell Preparation & Stimulation: Isolate PBMCs via density gradient centrifugation. Seed 1x10^6 cells/well in a 96-well plate. Stimulate with antigenic peptides (e.g., 1 µg/mL), positive control (PMA/Ionomycin), and negative control (DMSO/media). Include a protein transport inhibitor (Brefeldin A, 1 µg/mL) for the final 4-6 hours of a 12-16 hour incubation at 37°C, 5% CO₂.
- Surface Staining: Harvest cells, wash, and stain with viability dye and fluorochrome-conjugated antibodies against surface markers (e.g., CD3, CD4, CD8) for 20-30 minutes at 4°C in the dark.
- Fixation & Permeabilization: Wash cells, then fix and permeabilize using a commercial cytofix/cytoperm buffer for 20 minutes at 4°C.
- Intracellular Staining: Wash with permeabilization buffer, then stain with fluorochrome-conjugated antibodies against intracellular cytokines (e.g., IFN-γ, IL-2, TNF-α) for 30 minutes at 4°C in the dark.
- Acquisition & Analysis: Wash, resuspend in fixation buffer, and acquire on a flow cytometer. Analyze data using software (e.g., FlowJo) to gate on live, single, CD3+CD4+/CD8+ lymphocytes and determine the frequency of cytokine-positive cells.
Protocol 2: Standard QuantiFERON-TB Gold Plus Assay Protocol
- Blood Collection & Stimulation: Collect venous blood directly into four QuantiFERON tubes: Nil (negative control), TB1 & TB2 (containing M. tuberculosis-specific antigens), and Mitogen (positive control). Invert tubes 10 times immediately.
- Incubation: Incubate tubes upright for 16-24 hours at 37°C (±1°C).
- Plasma Harvest: Centrifuge tubes and harvest the supernatant plasma using a pipette, ensuring no cell transfer.
- ELISA Procedure: Add harvested plasma to the pre-coated IFN-γ ELISA plate and incubate for 120 minutes (±10 min) at room temperature. Wash plates 4-6 times. Add conjugate (anti-IFN-γ antibody linked to horseradish peroxidase) and incubate for 60 minutes at room temperature. Wash again.
- Development & Reading: Add substrate solution (TMB) and incubate for 30 minutes in the dark. Add stop solution. Measure optical density (OD) at 450 nm (reference 620-650 nm) within 30 minutes.
- Interpretation: Calculate IFN-γ concentration (IU/mL) by subtracting Nil OD from TB antigen or Mitogen OD and referencing the standard curve. Use manufacturer's cut-offs for positive, negative, or indeterminate results.
Visualizing Methodological Pathways & Workflows
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for T Cell Response Detection Assays
| Item | Function | Primary Application |
|---|---|---|
| Cell Activation Cocktail | Pharmacologic stimulation (PMA/Ionomycin) to induce maximal cytokine production as a positive control. | ICS, other functional assays. |
| Protein Transport Inhibitors | Brefeldin A or Monensin block Golgi transport, causing cytokine accumulation inside the cell for detection. | ICS. |
| Fluorochrome-Conjugated Antibodies | Target-specific antibodies for surface markers (CD3, CD4, CD8) and intracellular cytokines (IFN-γ, IL-2, TNF-α). | ICS. |
| Fixation/Permeabilization Buffer | Reagent system to fix cells and permeabilize membranes, allowing intracellular antibody access. | ICS. |
| QuantiFERON Antigen Tubes | Pre-coated blood collection tubes containing TB-specific antigens (ESAT-6, CFP-7) and controls. | QuantiFERON. |
| IFN-γ ELISA Kit | Antibody pair and reagents for the quantitative detection of human IFN-γ in plasma/supernatant. | QuantiFERON, other IGRA. |
| Density Gradient Medium | Solution for isolating peripheral blood mononuclear cells (PBMCs) from whole blood via centrifugation. | ICS (sample prep). |
| Cell Viability Dye | Dye to label dead cells (e.g., fixable viability stain) for exclusion during flow cytometry analysis. | ICS. |
The choice between ICS and QuantiFERON is not mutually exclusive but hypothesis-dependent. For definitive clinical diagnosis or large-scale screening where a binary result is needed, QuantiFERON's standardized protocol is superior. For mechanistic research, vaccine immunogenicity studies, or any investigation requiring deep immune profiling, ICS is indispensable. A robust research program may employ a complementary approach: using QuantiFERON for initial high-throughput screening of subject cohorts, followed by ICS for in-depth phenotypic and functional characterization of responders versus non-responders, thereby leveraging the strengths of both platforms within a single thesis.
Conclusion
The choice between ICS and QuantiFERON is not a matter of superiority, but of strategic alignment with research objectives. ICS offers unparalleled multidimensional profiling of specific T-cell subsets and functions, making it indispensable for mechanistic studies and advanced immunogenicity profiling. QuantiFERON provides a robust, standardized, and higher-throughput solution for quantifying aggregate IFN-γ responses, ideal for screening and longitudinal monitoring. Future directions point toward increased integration—using QuantiFERON for screening and ICS for deep phenotyping—and the adoption of newer multiplex platforms. For researchers, the key is to define the required information depth, throughput, and regulatory context, allowing the specific scientific question to drive the selection of these powerful, complementary tools for decoding T-cell immunity.