High-Resolution OCT Imaging of the Eustachian Tube Nasopharyngeal Region: A Comprehensive Guide for Researchers and Drug Developers
This article provides a detailed examination of Optical Coherence Tomography (OCT) for visualizing the Eustachian tube (ET) nasopharyngeal orifice.
High-Resolution OCT Imaging of the Eustachian Tube Nasopharyngeal Region: A Comprehensive Guide for Researchers and Drug Developers
Abstract
This article provides a detailed examination of Optical Coherence Tomography (OCT) for visualizing the Eustachian tube (ET) nasopharyngeal orifice. Tailored for researchers, scientists, and drug development professionals, it explores the fundamental principles of OCT in this challenging anatomical niche, outlines a step-by-step methodological protocol, addresses common troubleshooting and optimization challenges, and validates its utility through comparative analysis with other imaging modalities. The goal is to equip the biomedical community with the knowledge to leverage this non-invasive, high-resolution imaging technique for advancing studies in ET dysfunction, mucosal pathology, and therapeutic interventions.
Unveiling the Anatomy: OCT Fundamentals for Eustachian Tube Nasopharyngeal Imaging
Why Image the Eustachian Tube Orifice? Clinical and Research Motivations.
Imaging the Eustachian tube orifice (ETO), or nasopharyngeal orifice, is a critical frontier in otology and rhinology. The ETO serves as the primary dynamic gateway to the middle ear, regulating pressure, clearing secretions, and protecting from nasopharyngeal reflux. Direct, high-resolution imaging of this region provides unparalleled insights into its pathophysiology, which is implicated in conditions like obstructive Eustachian tube dysfunction (ETD), otitis media, and patulous ET. Within the broader thesis on Optical Coherence Tomography (OCT) imaging of the nasopharyngeal region, focusing on the ETO is justified by its role as the functional unit of ET physiology. This document outlines the application notes and protocols for its study.
Key Motivations: Clinical and Research
| Motivation Category | Specific Objectives | Quantitative Impact / Relevance |
|---|---|---|
| Clinical Diagnostics | Differentiate obstructive vs. patulous ETD in vivo. | Up to 80% of chronic otitis media cases are linked to ETD. Accurate subtype diagnosis is currently subjective. |
| Assess ETO mucosal pathology (edema, hyperplasia). | Mucosal thickness >1.5mm at the ETO is correlated with obstruction (OCT-derived metric). | |
| Guide balloon dilation catheter placement and sizing. | Balloon dilation procedures have a ~70-80% success rate; precise ETO measurements could optimize patient selection. | |
| Drug Development & Therapy | Evaluate topical drug delivery efficacy to the ETO. | <5% of topical nasal sprays typically reach the ETO region. Imaging quantifies coating and retention. |
| Monitor pharmacodynamic response (e.g., anti-inflammatory reduction of edema). | Enables quantification of mucosal thickness change (%) post-therapy in clinical trials. | |
| Develop and test novel ET stents or sustained-release implants. | Provides micron-scale measurements of implant apposition and tissue reaction over time. | |
| Basic Science Research | Characterize in vivo 3D microanatomy of the ETO and valve. | The cartilaginous valve is only 1-2mm in thickness; requires micron-scale resolution for study. |
| Study real-time tubal opening dynamics (pharyngeal phase of swallowing). | Normal ET opening lasts ~400ms; high-speed OCT can capture this dynamic event. | |
| Correlate ETO phenotype with genetic markers of inflammation. | Enables precise phenotyping for genotype-phenotype association studies in ETD. |
Experimental Protocol: OCT Imaging of the ETO in Human Subjects
Aim: To acquire high-resolution, cross-sectional, and volumetric OCT data of the Eustachian tube orifice in vivo.
I. Pre-Imaging Preparation
- Subject Selection & Consent: Recruit subjects (patients with ETD and healthy controls) under an IRB-approved protocol. Document otologic history and perform baseline nasoendoscopy.
- Equipment Setup:
- OCT System: A spectral-domain or swept-source OCT system with a lateral resolution of <15 µm and axial resolution of <10 µm.
- Imaging Probe: Utilize a flexible, catheter-based OCT probe (diameter ~1-2mm) compatible with the working channel of a standard nasal endoscope, or an integrated OCT-endoscopy system.
- Positioning: Secure the OCT console. Calibrate the system according to manufacturer specifications.
- Subject Preparation: Administer topical decongestant (e.g., oxymetazoline 0.05%) and anesthetic (e.g., lidocaine 2% spray) to the nasal cavity. Allow 5-10 minutes for effect.
II. Image Acquisition Procedure
- Navigated Insertion: Under endoscopic guidance, advance the nasal endoscope to the nasopharynx. Identify the ipsilateral ETO, located posterior to the inferior turbinate tail.
- Probe Placement: Pass the sterile OCT probe through the endoscope's working channel. Position the probe tip 1-2mm from, and perpendicular to, the mucosal surface of the ETO.
- Data Capture:
- Static 3D Scan: Hold position stable. Acquire a volumetric scan (e.g., 5mm x 5mm x 3mm depth) of the ETO region.
- Dynamic 2D Scan (M-mode): Position the beam at the midpoint of the ETO. Instruct the subject to perform a standardized maneuver (dry swallow, Toynbee maneuver). Acquire repeated A-scans at a single line for 2-3 seconds to capture opening/closing dynamics.
- Multi-Point Mapping: Systematically image the superior, inferior, anterior, and posterior rims of the orifice to construct a full topographic map.
- Data Storage: Save raw interferometric data and processed images with de-identified subject codes.
III. Post-Processing & Analysis
- Image Reconstruction: Use proprietary and custom software (e.g., in MATLAB) to generate 2D B-scans and 3D renderings.
- Quantitative Metrics:
- Lumen Area (A): Manually or semi-automatically segment the ETO lumen in cross-section. Calculate area (mm²).
- Mucosal Thickness (T): Measure from epithelial surface to cartilage/bone interface at four quadrants. Average.
- Dynamic Parameters: From M-mode, calculate opening duration (ms) and maximum luminal diameter (mm).
- Statistical Analysis: Compare metrics between cohorts using appropriate tests (t-test, ANOVA).
Visualization of Research Context & Workflow
Diagram 1: Thesis Research Workflow Integrating ETO Imaging.
Diagram 2: Pathophysiological Pathway from ETO Pathology to Disease.
The Scientist's Toolkit: Key Research Reagent Solutions
| Item / Reagent | Function in ETO Imaging Research | Example / Specification |
|---|---|---|
| Swept-Source OCT Engine | Provides the light source and detector for high-speed, high-resolution imaging. | Axsun Technologies laser; central wavelength ~1300nm for optimal tissue penetration. |
| Flexible OCT Imaging Catheter | Enables transnasal access to the nasopharynx for in vivo imaging. | 1.5-2.0mm OD, rotational scanning probe, compatible with endoscope working channels. |
| Integrated OCT-Endoscope System | Allows simultaneous white-light navigation and OCT imaging. | Custom systems from research collaborations or prototypes from companies like NinePoint Medical. |
| Topical Decongestant | Reduces nasal mucosa vascular congestion for clearer passage and imaging. | Oxymetazoline HCl 0.05% solution. |
| Topical Mucosal Anaesthetic | Ensures patient comfort during probe manipulation. | Lidocaine hydrochloride spray (2-4%). |
| Image Processing Software | For reconstruction, analysis, and quantification of OCT data. | MATLAB with custom scripts, IntelliSlice (OCT-specific), or Amira. |
| Synthetic Phantoms | Calibrate OCT system resolution and validate measurements before human studies. | Agarose or silicone phantoms with embedded microstructures of known dimensions. |
| Validated Patient-Reported Outcome Measures | Correlate imaging metrics with subjective symptoms. | ETDQ-7 (Eustachian Tube Dysfunction Questionnaire-7). |
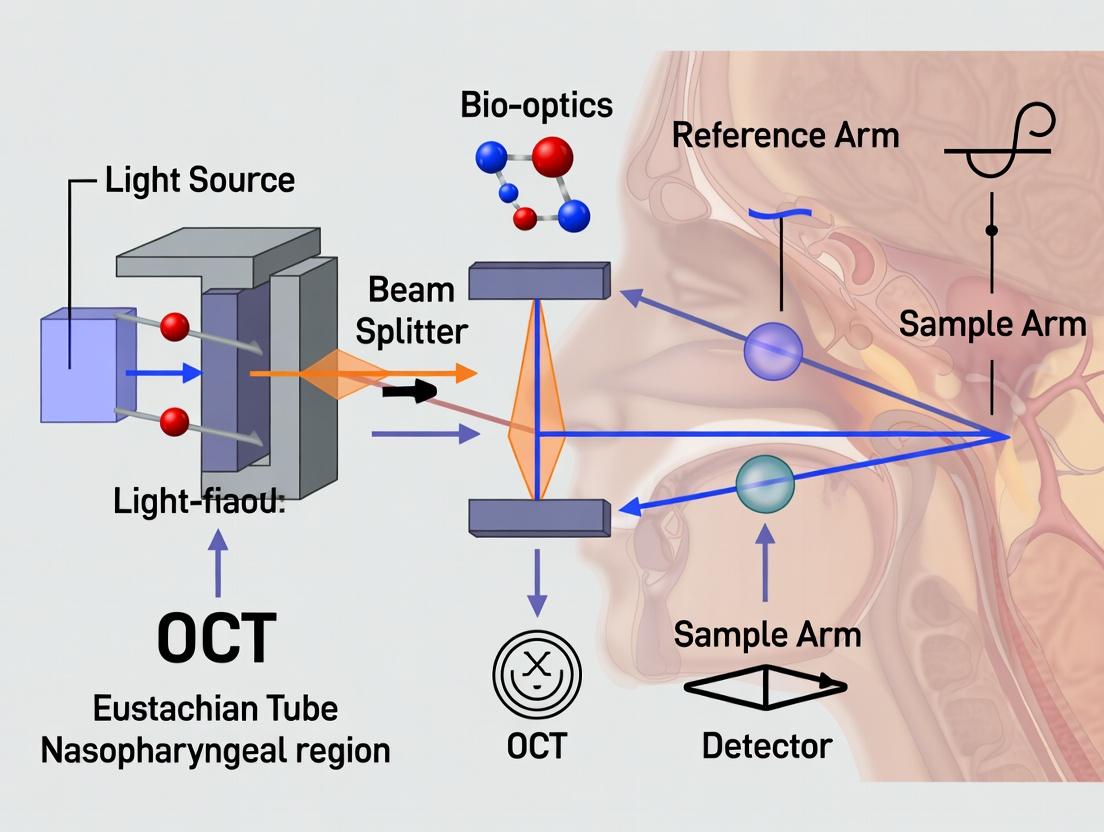
This document provides detailed application notes and experimental protocols on the core principles of Optical Coherence Tomography (OCT), framed within ongoing thesis research on imaging the Eustachian tube (ET) and nasopharyngeal region. The evolution from Time-Domain (TD-OCT) to Spectral-Domain (SD-OCT) systems is critical for advancing in vivo, non-invasive, high-resolution morphological and functional imaging of this complex anatomical area. This supports research in pathophysiology, therapeutic intervention, and drug delivery assessment.
Core Principles and System Evolution
OCT is an interferometric technique that measures backscattered light to generate cross-sectional, micron-scale images of biological tissue.
Time-Domain OCT (TD-OCT)
The original implementation. A broadband light source is split into sample and reference arms. Interference occurs only when the optical path lengths of both arms match within the coherence length of the source. Depth information (A-scan) is acquired by mechanically scanning the reference mirror, varying the time delay.
Key Limitation: Imaging speed and signal-to-noise ratio (SNR) are fundamentally limited by the need for mechanical scanning.
Spectral-Domain OCT (SD-OCT)
Also known as Fourier-Domain OCT. Comprises Spectral-Domain (SD) and Swept-Source (SS) variants. The core principle: the depth-dependent information is encoded in the frequency spectrum of the interferometric signal. The reference mirror is fixed. The combined spectrum is captured by a spectrometer (SD) or a rapidly tuned laser (SS) and Fourier-transformed to derive depth information.
Key Advantages: Superior imaging speed (50-100x faster than TD-OCT) and enhanced SNR, enabling real-time, volumetric imaging.
Quantitative Comparison of TD-OCT vs. SD-OCT
Table 1: Performance Comparison of TD-OCT and SD-OCT Systems for Biological Imaging
| Parameter | Time-Domain OCT (TD-OCT) | Spectral-Domain OCT (SD-OCT) |
|---|---|---|
| Axial Resolution | 5-15 µm | 1-5 µm |
| Imaging Speed (A-scans/sec) | 400 - 2,000 | 20,000 - 300,000+ |
| Typical Sensitivity (dB) | 90 - 105 | 95 - 110+ |
| Key Limitation | Mechanical scanning limits speed/SNR | Spectral fall-off, depth range limit |
| Suitability for in vivo ET Imaging | Limited for dynamic studies | Excellent for 3D volumes & motility |
Application Notes for ET/ Nasopharyngeal Imaging
Imaging the ET presents challenges: tortuous anatomy, mucosal surface, dynamic function (opening/closing), and proximity to the middle ear.
SD-OCT Advantages in this Context:
- High-Speed Volumetric Imaging: Captures 3D morphology of the ET lumen and nasopharyngeal orifice in a single acquisition, minimizing motion artifact.
- Functional Assessment: Rapid line scans (M-mode) can track tubal dilation over time in response to stimuli (e.g., swallowing, drug application).
- Enhanced Detail: Improved resolution reveals epithelial layers, submucosal glands, and vascular patterns relevant to inflammation and treatment response.
Experimental Protocols
Protocol 1:Ex VivoET Tissue Morphometry
Aim: To quantitatively compare layer thickness and scattering properties of healthy vs. inflamed ET mucosa. Materials: See Scientist's Toolkit. Method:
- Excise ET tissue samples (animal model or human cadaveric) and mount in oxygenated Krebs solution.
- Using a 1300 nm SD-OCT system, acquire 3D volumetric scans (Field of View: 5x5x3 mm).
- Generate cross-sectional B-scans. Use built-in or custom software (e.g., ImageJ) to measure epithelial and subepithelial layer thickness at 10 predefined points per sample.
- Calculate spatially resolved scattering coefficient (µs) from the depth-dependent signal decay using a fitting algorithm.
- Perform histology (H&E) on the same samples for correlation and validation.
Data Analysis: Compare mean layer thickness and µs between experimental groups using a two-tailed t-test. Present as mean ± SD.
Protocol 2:In VivoET Patency/Dynamics Monitoring
Aim: To dynamically assess ET opening in a live animal model pre- and post-topical drug application. Method:
- Anesthetize and position animal (e.g., guinea pig) per IACUC protocol.
- Introduce a miniature endoscopic SD-OCT probe (e.g., 2.7 mm diameter) transnasally to visualize the nasopharyngeal ET orifice.
- Acquire a baseline 3D volume. Then, perform high-speed M-mode imaging (5,000 A-scans/sec) at the orifice during a swallow stimulus (induced by tactile pharyngeal stimulation). Repeat 5x.
- Topically apply 100 µL of a mucolytic or decongestant test formulation to the orifice.
- Wait 10 minutes, then repeat step 3 for post-treatment imaging.
- Process M-mode images: measure the maximum lateral distension of the orifice and the duration of opening for each swallow event.
Data Analysis: Report pre- and post-treatment mean opening diameter and duration. Statistical significance assessed via ANOVA.
Visualization of OCT System Principles & Workflow
Diagram 1: OCT System Evolution from TD to SD
Diagram 2: ET OCT Imaging Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for OCT Imaging of the Eustachian Tube
| Item / Reagent | Function / Purpose | Example/Notes |
|---|---|---|
| SD-OCT System | Core imaging hardware. | Central wavelength: 1300 nm (deeper penetration) preferred for ET. |
| Endoscopic OCT Probe | Miniaturized optics for in vivo access. | Rotary-pullback catheter or side-firing probe (≤2.7mm diameter). |
| Animal Model | In vivo physiology & drug testing. | Guinea pig or chinchilla (established ET anatomy/physiology). |
| Krebs-Ringer Solution | Maintain tissue viability ex vivo. | Physiological buffer for oxygenated tissue bath. |
| Topical Drug Formulations | Test articles for functional studies. | Decongestants (e.g., Phenylephrine), corticosteroids, mucolytics. |
| Immersion Fluid | Index matching for mucosal imaging. | Saline or viscous gel to reduce surface refraction artifacts. |
| Histology Fixative | Gold-standard correlation. | 10% Neutral Buffered Formalin for post-OCT tissue fixation. |
| Image Analysis Software | Quantitative morphometry & dynamics. | Custom MATLAB/Python scripts, ImageJ, commercial OCT software. |
This application note details the anatomical and cellular framework of the nasopharyngeal Eustachian Tube (ET) region, a critical focus for optical coherence tomography (OCT) imaging technique research. Precise anatomical mapping is foundational for developing in vivo OCT protocols to assess ET dysfunction, monitor therapeutic interventions, and evaluate drug delivery efficacy in preclinical and clinical models.
Quantitative Anatomical & Histological Data
Key dimensional and cellular data for the human ET nasopharyngeal region are summarized below.
Table 1: Key Anatomical Dimensions of the Human Eustachian Tube (Mean ± SD)
| Structure | Measurement | Value (mm) | Notes/Source |
|---|---|---|---|
| Total ET Length | From tympanic to pharyngeal orifice | 31 – 38 mm | Adult average; cartilaginous portion ~26 mm. |
| Lumen Diameter | At nasopharyngeal orifice (resting) | 3 – 5 mm | Highly dynamic; can open to 8-10 mm. |
| Torus Tubarius | Vertical height | 8 – 10 mm | Primary landmark for OCT probe positioning. |
| Cartilage Lamin a | Thickness | 1.5 – 2.5 mm | Critical for OCT signal penetration analysis. |
| Mucosa Thickness | Lining epithelium & lamina propria | 0.5 – 1.2 mm | Primary layer for inflammatory assessment via OCT. |
Table 2: Cellular Composition of ET Mucosa (Key Metrics)
| Cell Type | Approx. Density / Proportion | Primary Function | Relevance to OCT/Research |
|---|---|---|---|
| Pseudostratified Ciliated Columnar | ~60-70% of epithelial layer | Mucociliary clearance | OCT can assess cilia density/health via scattering. |
| Goblet Cells | ~15-20% of epithelial layer | Mucin secretion | OCT contrast agents may target mucin. |
| Basal Cells | ~10-15% of epithelial layer | Epithelial regeneration | Biomarker for injury/repair in longitudinal OCT. |
| Immune Cells (e.g., Mast Cells) | 50-100 cells/mm² in lamina propria | Inflammatory response | OCT can detect edema from immune activity. |
| Submucosal Glands | Density varies regionally | Seromucous secretion | Glandular ducts may be visualized via high-res OCT. |
Experimental Protocols for Anatomical Validation
Protocol 1: Ex Vivo Tissue Preparation for OCT-Histology Correlation
Objective: To prepare fresh human or porcine nasopharyngeal ET specimens for correlative OCT imaging and histology, enabling validation of OCT image features.
Materials: See "Research Reagent Solutions" below. Procedure:
- Tissue Acquisition & Orientation: Obtain fresh cadaveric or surgical specimen. Identify the torus tubarius and pharyngeal ostium using a dissection microscope. Mark the anterior margin with surgical ink.
- Sample Mounting for OCT: Embed the specimen in optimal cutting temperature (OCT) compound within a custom cylindrical holder designed to fit the OCT sample arm. Orient the mucosal surface perpendicular to the imaging beam.
- OCT Imaging: Acquire 3D OCT volumes (e.g., 1300 nm central wavelength, 5 µm axial resolution) across the region of interest. Use a scanning protocol of 1000 A-scans per B-scan, 500 B-scans per volume. Save data in raw format.
- Fiducial Marker Placement: Post-OCT, insert two or more thin (≤100 µm) nylon sutures perpendicularly through the tissue block as fiducial markers for spatial registration.
- Fixation & Processing: Immerse the block in 10% neutral buffered formalin for 24-48 hours. Process through graded ethanol series and xylene, then embed in paraffin.
- Sectioning & Staining: Serially section the block at 5 µm thickness in the plane corresponding to key OCT B-scans. Perform Hematoxylin & Eosin (H&E) and Alcian Blue/PAS staining on alternating slides.
- Digital Correlation: Digitize histology slides. Use fiducial markers and distinctive anatomical landmarks (e.g., gland openings) to co-register H&E images with the corresponding OCT B-scan using rigid/affine transformation software (e.g., 3D Slicer).
Protocol 2: In Vivo Murine OCT Imaging of the Nasopharyngeal ET
Objective: To perform in vivo, non-invasive OCT imaging of the murine ET nasopharyngeal region for longitudinal studies of disease models or treatment.
Materials: Custom murine OCT endoscope, isoflurane vaporizer, stereotaxic holder, heating pad, topical decongestant (e.g., oxymetazoline, 0.05%). Procedure:
- Animal Preparation: Anesthetize mouse with 2-3% isoflurane in oxygen. Secure in a supine position in a stereotaxic head holder with a heating pad. Maintain anesthesia at 1-2% isoflurane.
- Nasopharyngeal Access: Apply 5 µL of diluted topical decongestant to each nare. Using a modified otoscope, gently advance a blunt-tipped guide sheath through the nasal cavity until resistance at the nasopharynx is felt.
- OCT Probe Positioning: Insert a miniaturized rotational OCT probe (e.g., 0.9 mm outer diameter, 1300 nm) through the guide sheath. Under real-time OCT visualization, position the probe tip at the nasopharyngeal orifice, identified by the characteristic semi-lunar shape of the torus tubarius.
- Image Acquisition: Acquire radial cross-sectional scans (1000 A-scans per frame) while slowly pulling back the probe at 0.5 mm/s over a 3-5 mm trajectory. Record multiple pullbacks.
- Recovery & Monitoring: Gently withdraw the probe and sheath. Monitor animal until fully recovered from anesthesia. Post-procedure analgesic (e.g., buprenorphine) is administered as per IACUC protocol.
- Image Analysis: Reconstruct 3D volumes. Quantify metrics such as lumen patency (area in mm²), mucosal thickness (µm), and scattering intensity in the lamina propria as a proxy for cellular infiltration.
Visualization: Pathways and Workflows
Title: OCT-Histology Correlation Workflow for ET Research
Title: Inflammatory Pathway & OCT-Detectable Features in ET
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for ET Region OCT Research
| Item | Function/Application | Example Product/Catalog # | Notes |
|---|---|---|---|
| Custom OCT Endoscope | In vivo access to nasopharynx. | Prototype, 0.9-1.2 mm OD, 1300 nm. | Requires rotational junction; key for small animal imaging. |
| Tissue Optical Clearing Agent | Reduces scattering for deeper OCT penetration in ex vivo studies. | FocusClear (CellExplorer); 80% Glycerol. | Crucial for visualizing deep cartilage-bone interface. |
| Mucolytic Agent (for ex vivo) | Clears surface mucus to improve imaging clarity. | Dithiothreitol (DTT), 10 mM solution. | Apply topically for 2 mins before imaging, then rinse. |
| Decalcification Solution | For processing bony ET specimens for histology. | EDTA, 10% pH 7.4 (slow); Formical-2000 (fast). | Required for full cross-section histology; monitor with radiography. |
| Alcian Blue / PAS Stain Kit | Differentiates neutral (PAS+) and acidic (Alcian Blue+) mucins. | Sigma-Aldrich AB-1 & 395B kits. | Critical for assessing goblet cell hyperplasia and metaplasia. |
| Stereotaxic Head Holder (Murine) | Stabilizes head for reproducible OCT probe alignment. | David Kopf Instruments, Model 923-B. | Must be modified with a supine platform and nose clamp. |
| Fiducial Markers | For OCT-Histology coregistration. | Ethilon Nylon Sutures, 10-0. | Inert, thin, creates clear hypo-reflective shadow in OCT. |
| Digital Histology Coregistration Software | Aligns OCT and histology images. | 3D Slicer; custom MATLAB/Python scripts. | Essential for validating OCT image interpretation. |
Within the context of advancing research on imaging techniques for the Eustachian tube (ET) and nasopharyngeal (NP) region, Optical Coherence Tomography (OCT) offers a transformative toolset. This application note details the core advantages of OCT—non-invasiveness, high-resolution, and real-time cross-sectional imaging—and provides specific protocols for its application in preclinical and clinical research of ET function and drug delivery assessment. These protocols are designed for researchers and drug development professionals aiming to quantify morphological and functional parameters in this anatomically complex and clinically critical area.
Core Advantages & Quantitative Metrics
The utility of OCT in ET/NP research is grounded in its technical specifications, which enable studies previously constrained by the limitations of histology or lower-resolution imaging modalities.
Table 1: Key OCT Performance Metrics Relevant to ET/NP Imaging
| Advantage | Quantitative Metric | Typical Range (System-Dependent) | Significance for ET/NP Research |
|---|---|---|---|
| Non-Invasive | Penetration Depth | 1-3 mm in mucosal tissue | Allows repeated, in vivo assessment of the same subject/animal over time, enabling longitudinal studies of disease progression or treatment efficacy without terminal endpoints. |
| High-Resolution | Axial Resolution | 1-15 µm | Capable of delineating epithelial layers, identifying submucosal glands, measuring mucosal thickness, and detecting early metaplastic or dysplastic changes. |
| High-Resolution | Lateral Resolution | 5-30 µm | Provides clear delineation of tissue microstructures, such as the ET lumen, mucosal folds, and lymphoid tissue in the nasopharynx. |
| Real-Time | A-scan Rate | 50,000 - 500,000 scans/second | Enables dynamic imaging of ET opening/closing (tubal function), ciliary motility, and real-time guidance for precise probe placement or biopsy. |
| Cross-Sectional | Scan Dimension (B-scan) | Up to 16 mm (width) x 3 mm (depth) | Generates tomographic "virtual histology" views of the ET lumen and surrounding tissues, correlating directly with pathological sections. |
Experimental Protocols
Protocol 1:In VivoMurine ET Patency and Mucosal Morphometry
Objective: To longitudinally assess ET lumen patency and mucosal thickness in a mouse model of allergic rhinitis/otitis media.
Materials:
- Anesthetized murine model.
- Spectral-Domain or Swept-Source OCT system with a flexible micro-probe (e.g., 1.0 mm outer diameter).
- Stereotactic mount for stable head positioning.
- Automated syringe pump for allergen challenge.
- Image analysis software (e.g., ImageJ, Amira).
Methodology:
- Anesthesia & Positioning: Induce and maintain anesthesia. Secure the mouse in a supine position in a stereotactic device.
- Probe Insertion: Under microscopic guidance, gently advance the sterile OCT probe through the narcs into the nasopharynx, orienting it toward the pharyngeal orifice of the ET.
- Baseline Imaging: Acquire a series of 500 consecutive cross-sectional (B-scan) images at the target site. Record 3D volumetric scans by performing raster scans over a 2mm x 2mm region.
- Challenge: Introduce a controlled volume of allergen (e.g., ovalbumin) or inflammatory mediator via the narcs using the syringe pump.
- Time-Series Imaging: Repeat imaging at the identical location at T=5, 15, 30, and 60 minutes post-challenge.
- Data Analysis:
- Lumen Area: Manually or automatically segment the ET lumen in each B-scan. Calculate mean lumen cross-sectional area (µm²) per time point.
- Mucosal Thickness: Measure the distance from the epithelial surface to the cartilage interface at 4 standardized points per image.
- Statistical Analysis: Use repeated-measures ANOVA to compare pre- and post-challenge metrics.
Protocol 2: Real-Time Monitoring of Topical Drug Delivery & Coating
Objective: To visualize and quantify the distribution and clearance of a topical formulation on human ET mucosa ex vivo.
Materials:
- Human NP/ET tissue specimen (from surgery).
- OCT system with a benchtop scanner.
- Test formulation (e.g., mucoadhesive gel containing microspheres or a contrast agent like Intralipid).
- Precision applicator.
- Environmental chamber to maintain tissue humidity and temperature.
Methodology:
- Tissue Preparation: Mount the fresh tissue specimen in the chamber with the mucosal surface exposed.
- Baseline Scan: Acquire high-resolution 3D OCT volumes of the target region.
- Formulation Application: Apply a precise volume (e.g., 10 µL) of the test formulation to the tissue surface using the applicator.
- Real-Time Imaging: Immediately initiate continuous, rapid B-scan imaging at a fixed position for 10 minutes. Subsequently, perform volumetric scans every 5 minutes for up to 1 hour.
- Clearance Simulation: Apply a simulated mucociliary clearance flow (using a peristaltic pump delivering saline at 5 µL/min) and continue imaging.
- Data Analysis:
- Coating Thickness: Measure the thickness of the high-scattering formulation layer over time.
- Coverage Persistence: Calculate the percentage of the image area where the formulation signal remains above a set threshold.
- Penetration Depth: Analyze signal attenuation to estimate formulation ingress into mucosal crypts or glands.
Visualization of Research Workflow
Title: OCT Imaging Workflow for ET Research
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for OCT Imaging of the ET/ Nasopharyngeal Region
| Item | Function & Relevance |
|---|---|
| Flexible OCT Micro-Probe (e.g., 0.9-1.2 mm OD) | Enables access to the narrow, curved anatomy of the nasopharynx and ET orifice without traumatic injury, crucial for in vivo studies. |
| Intralipid 20% Intravenous Fat Emulsion | A biocompatible scattering agent. Used as a contrast agent to simulate or enhance the signal from topical drug formulations for clearance studies. |
| Mucoadhesive Gel Base (e.g., Carbopol) | Serves as a vehicle for test compounds. Its retention properties can be directly visualized and measured by OCT, informing formulation design. |
| Animal Model (e.g., OVA-sensitized mouse) | Provides a controlled system for studying inflammatory-mediated ET dysfunction and testing therapeutic interventions longitudinally. |
| Stereotactic Positioning System | Ensures precise, repeatable positioning of the OCT probe relative to the target tissue across multiple imaging sessions, critical for longitudinal data integrity. |
| Spectral-Domain/Swept-Source OCT Engine (Central λ: ~1300 nm) | Provides the optimal combination of resolution (~5-10 µm) and penetration depth (2-3 mm) for imaging beneath the mucosal surface in the NP/ET region. |
Recent advancements in Optical Coherence Tomography (OCT) imaging of the Eustachian Tube (ET) and nasopharyngeal (NP) region have focused on enhancing resolution, depth penetration, and functional assessment to overcome the limitations of standard endoscopic techniques. The current research frontier integrates anatomical imaging with physiological metrics, aiming to establish OCT as a quantitative tool for diagnosing ET dysfunction (ETD) and evaluating therapeutic interventions.
Key Research Themes (2022-2024):
- High-Definition Anatomical Mapping: Use of swept-source OCT (SS-OCT) and microscope-integrated OCT (MI-OCT) to delineate the mucosal microstructure, lymphoid tissue distribution in the torus tubarius, and the cartilaginous-vascular architecture of the protympanum.
- Functional & Dynamic Imaging: Development of Doppler-OCT and phase-sensitive OCT to assess mucociliary activity, blood flow dynamics in the tubal mucosa, and real-time tubal dilation during swallowing or Valsalva.
- In Vivo Translational Studies: Pioneering pilot studies in human subjects using transnasal OCT catheters and probe-based systems to correlate OCT biomarkers (e.g., epithelial thickness, submucosal gland density) with ETD severity scores.
- Therapeutic Monitoring: Application of OCT to longitudinally track mucosal healing, drug depot retention, and biomaterial integration post-surgical or pharmacological intervention.
Table 1: Performance Metrics of Recent OCT Systems for ET/NP Imaging
| Study (Year) | OCT Modality | Central Wavelength (nm) | Axial Resolution (µm) | Imaging Depth (mm) | Key Measured Parameter | Mean Value (±SD) | Application Context |
|---|---|---|---|---|---|---|---|
| Chen et al. (2023) | Spectral-Domain (SD-OCT) | 1310 | 7.5 | 2.8 | ET Lumen Patency Index | 0.62 ± 0.18 | In vivo, during Valsalva |
| Park & Lee (2022) | Swept-Source (SS-OCT) | 1060 | 5.2 | 4.2 | Mucosal Thickness (Torus Tubarius) | 415 ± 89 µm | Ex vivo tissue validation |
| Alvarez et al. (2024) | Doppler-OCT | 1300 | 9.0 | 3.0 | Mucociliary Transport Velocity | 1.4 ± 0.3 mm/min | In vivo, rabbit ET model |
| Iyer et al. (2023) | Probe-based (μOCT) | 800 | 2.0 | 1.5 | Submucosal Gland Density (#/mm²) | 12.5 ± 3.2 | Biopsy correlation study |
Table 2: Correlation of OCT Biomarkers with Clinical ETD Scores (Recent Clinical Pilot)
| OCT-Derived Biomarker | Pearson's r with ETDQ-7 Score | p-value | Cohort (n) | Diagnostic Threshold (Proposed) |
|---|---|---|---|---|
| Minimum Lumen Diameter (Rest) | -0.78 | <0.001 | 45 | < 0.8 mm |
| Mucosal Roughness Index | +0.71 | <0.001 | 45 | > 25.0 AU |
| Dilation Responsiveness (% Change) | -0.82 | <0.001 | 30 | < 15% increase |
| Lamina Propria Signal Intensity | +0.65 | <0.005 | 45 | > 110% reference |
Experimental Protocols
Protocol 1: In Vivo Functional Imaging of Human ET with Catheter-Based SS-OCT
- Objective: To quantitatively assess ET lumen dynamics and mucosal morphology in awake patients.
- Materials: 2.7Fr rotational SS-OCT imaging catheter, commercial SS-OCT engine (λ=1310nm), nasal endoscope for guidance, topical anesthetic/decongestant.
- Procedure:
- Patient preparation: Administer topical oxymetazoline and lidocaine to the nasal cavity.
- Catheter positioning: Under endoscopic visualization, advance the sterile OCT catheter through the nasal cavity to the ET orifice.
- Baseline scan: Acquire a pullback scan (20 mm length, 100 fps) of the pharyngeal ET segment at rest.
- Functional maneuver: Instruct patient to perform a standardized Valsalva maneuver. Trigger a second synchronized pullback scan during the maneuver.
- Data acquisition: Repeat 3x for statistical robustness. Record patient-reported ETD questionnaire (ETDQ-7) scores.
- Processing: Use custom software to compute lumen cross-sectional area, diameter, and mucosal texture metrics from 3D reconstructed data. Calculate percentage change between rest and Valsalva states.
Protocol 2: Ex Vivo Murine ET Tissue Imaging for Drug Penetration Analysis
- Objective: To visualize and quantify the penetration depth and distribution of a fluorescently tagged topical drug surrogate using OCT.
- Materials: Fresh murine ET/temporal bone specimens, SD-OCT system (λ=850nm), fluorescent dye (e.g., ICG), confocal microscope for validation.
- Procedure:
- Tissue preparation: Dissect ET complex and mount in artificial CSF-moistened chamber.
- Baseline OCT: Acquire high-resolution 3D scan of the mucosal surface and underlying cartilage.
- Drug application: Apply 10 µL of ICG solution (1 mg/mL) topically to the ET lumen.
- Time-lapse imaging: Acquire OCT B-scans at the same location every 30 seconds for 20 minutes.
- Signal analysis: Measure the depth-resolved OCT signal increase (relative to baseline) correlated with fluorescence. Use Doppler algorithms to track fluid flow.
- Validation: Fix tissue and perform confocal microscopy on cryosections to correlate OCT signal with fluorescence distribution.
Visualizations
OCT Biomarker Validation Workflow for ETD (760px)
From Light to 3D Image: OCT Signal Pathway (760px)
The Scientist's Toolkit: Research Reagent & Material Solutions
Table 3: Essential Materials for Advanced OCT Imaging of the ET/NP Region
| Item / Reagent Solution | Primary Function in Research | Example Application / Note |
|---|---|---|
| Swept-Source Laser Module | High-speed, long-depth-range imaging light source. | Enables full-length ET scanning without depth ambiguity. Central λ ~1060-1310nm. |
| Rotational OCT Imaging Catheter (≤3Fr) | Miniaturized probe for in vivo intraluminal imaging. | Critical for translational human studies; provides 360° circumferential views. |
| Microscope-Integrated OCT (MI-OCT) System | Intraoperative, real-time cross-sectional imaging during surgery. | For guiding myringotomy or ET balloon dilation with subsurface visualization. |
| Phase-Stable Acquisition Software | Enables Doppler and quantitative flow imaging. | Measures mucociliary transport and blood flow dynamics in the ET mucosa. |
| Topical Tissue Optical Clearing Agent | Temporarily reduces scattering to increase imaging depth. | Glycerol-based solutions used ex vivo to enhance cartilage boundary visibility. |
| Synthetic Mucus Surrogate (e.g., Polyacrylamide) | Mimics rheological properties of ET mucus for functional studies. | Used in bench-top models to calibrate Doppler-OCT flow measurements. |
| Fluorescently-Labeled Dextran Conjugates | Drug/delivery vehicle surrogate for co-localization studies. | Validates OCT's ability to track topical drug penetration in animal models. |
| 3D Segmentation & Analysis Software (e.g., Amira, Custom MATLAB) | Extracts quantitative metrics from complex 3D OCT datasets. | Essential for calculating lumen volume, surface area, and texture parameters. |
Step-by-Step Protocol: Executing OCT Imaging of the Eustachian Tube Orifice
Application Notes
Optical Coherence Tomography (OCT) is a non-invasive, high-resolution imaging modality enabling real-time, cross-sectional visualization of tissue microstructure. In the context of a thesis focused on imaging the Eustachian Tube (ET) nasopharyngeal region, proper equipment selection is paramount due to the anatomical complexity, limited access, and specific research questions (e.g., assessing ET lumen patency, mucosal dynamics, or drug delivery effects). These notes detail the critical considerations for system and probe selection.
1. Core OCT System Specifications: The choice between Spectral-Domain (SD-OCT) and Swept-Source (SS-OCT) technologies is fundamental. For deep, narrow anatomical regions like the nasopharyngeal ET orifice, SS-OCT is generally superior due to its longer imaging range and higher imaging speed, which mitigates motion artifacts. Key quantitative parameters are summarized below.
Table 1: Comparative Analysis of OCT System Specifications for ET Imaging
| Parameter | Spectral-Domain (SD-OCT) | Swept-Source (SS-OCT) | Rationale for ET Research |
|---|---|---|---|
| Central Wavelength | ~840 nm, ~1310 nm | ~1310 nm, >1300 nm | Longer wavelengths (1310 nm) offer better tissue penetration, crucial for the mucosal-covered ET region. |
| Axial Resolution | 1-5 µm (in tissue) | 3-10 µm (in tissue) | High axial resolution (<7 µm) is needed to delineate epithelial layers and mucus gland structures. |
| A-Scan Rate | 50 - 200 kHz | 100 - 2,000+ kHz | High speed (>100 kHz) is critical for in vivo 3D imaging to freeze physiological motion. |
| Imaging Range (Depth) | 1-3 mm (in tissue) | 3-10+ mm (in tissue) | Extended range (≥5 mm) is required to capture the entire ET lumen profile and surrounding tissue. |
| Key Advantage | High resolution, cost-effective | Deeper penetration, faster speed, reduced sensitivity roll-off | SS-OCT's range and speed are often decisive for robust nasopharyngeal ET imaging. |
2. Probe Specifications and Design: The probe is the critical interface with the anatomy. For transnasal insertion, form factor and steering capability are essential.
Table 2: Probe Design Specifications for Nasopharyngeal ET Access
| Feature | Specification | Research Application |
|---|---|---|
| Probe Diameter | ≤2.5 mm (outer diameter) | Enables comfortable transnasal passage to the nasopharynx. |
| Working Length | 200 - 300 mm | Sufficient to reach the ET orifice from the naris. |
| Steering Mechanism | Motorized distal articulation or manual pull-wire. | Allows precise aiming at the dynamic ET opening. |
| Sheath Compatibility | Integrated or separate biocompatible, clear sheath. | Protects the probe, ensures hygiene, and can provide a stable imaging window. |
| Scanning Method | Distal side-viewing rotary or linear pullback. | Rotary scans provide circumferential views of the lumen; linear pullback generates longitudinal views. |
| Focusing | Fixed focus (e.g., 2-5 mm from probe sheath) or adjustable. | Must be optimized for the typical distance to the ET tissue surface. |
Experimental Protocols
Protocol 1: System Characterization and Calibration for ET Imaging Objective: To verify the performance metrics of the selected OCT system and probe prior to biological imaging. Materials: OCT system, imaging probe, calibrated resolution test target (USAF 1951), mirror, attenuation filters.
- Lateral Resolution: Image the USAF 1951 target. Place the smallest resolvable group element in the center of the field of view. Calculate lateral resolution from the known line pair spacing.
- Axial Resolution & System Roll-off: Place a mirror at the probe focus. Acquire an A-scan. Measure the Full-Width at Half-Maximum (FWHM) of the interference peak to determine axial resolution in air, then convert to tissue (divide by refractive index, ~1.33). Measure signal drop over depth using a translation stage.
- Sensitivity: Using neutral density filters, measure the minimum reflectivity detectable above the noise floor. Record as dB.
- Geometric Calibration: For rotary probes, perform a rotational correction using a target with known angular markings. For pullback probes, calibrate pullback distance using a stage with known translation.
Protocol 2: Ex Vivo Human Nasopharyngeal Tissue Imaging Objective: To validate imaging quality and assess tissue microstructural contrast. Materials: SS-OCT system with side-viewing rotary probe (≤2.5 mm), fresh cadaveric nasopharyngeal specimen (ET region), specimen mounting stage, phosphate-buffered saline (PBS), 4% paraformaldehyde.
- Obtain IRB/IACUC approval for cadaveric tissue use.
- Isolate the nasopharyngeal specimen containing the ET orifice. Keep moist with PBS.
- Mount the tissue on a stabilized stage with the ET lumen accessible.
- Insert the sterile-sheathed OCT probe into the nasal cavity, guiding it to the nasopharynx under endoscopic visualization (simulated).
- Position the probe tip perpendicular to and lightly touching the ET orifice.
- Acquire 3D volumetric data: 1000 A-scans per B-scan, 500 B-scan positions over a 5 mm pullback.
- Fix the imaged tissue in paraformaldehyde for 24h for potential histology correlation (H&E staining).
- Process OCT data (logarithmic scaling, filtering). Correlate OCT features (epithelium, lamina propria, cartilage) with histology.
Protocol 3: In Vivo Assessment of ET Lumen Dynamics Objective: To quantitatively measure ET lumen cross-sectional area changes during swallowing or Valsalva. Materials: SS-OCT system with articulated, side-viewing probe, sterile single-use sheath, nasal endoscope for guidance, vital signs monitor, data acquisition software.
- Recruit subjects under an approved human subjects protocol. Obtain informed consent.
- Perform nasal decongestion and topical anesthesia per clinical protocol.
- Under endoscopic guidance, advance the sheathed OCT probe to the nasopharyngeal ET orifice.
- Position the probe using articulation to align the imaging plane for a cross-sectional view of the ET lumen.
- Instruct the subject to perform baseline breathing, then swallow. Simultaneously, acquire a high-speed M-mode dataset (5 kHz A-scan rate) at a fixed position.
- Perform a 3D rotary pullback scan (5 mm length) during baseline and during sustained Valsalva maneuver.
- Segment the ET lumen border in the cross-sectional images using semi-automated software. Calculate lumen area over time from M-mode and compare 3D volumetric renders between baseline and Valsalva states.
Diagrams
Title: OCT System & Probe Selection Workflow for ET Research
Title: In Vivo ET Lumen Dynamics Imaging Protocol
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions & Materials for OCT ET Studies
| Item | Function / Purpose |
|---|---|
| SS-OCT System (e.g., Thorlabs OCS1300SS, Axsun Technologies) | Provides long imaging range (>5 mm in tissue) and high A-scan rate (>100 kHz) essential for capturing the ET lumen in 3D. |
| Side-Viewing Rotary/Pullback Probe (≤2.5mm OD) | The critical form factor for transnasal access; enables circumferential scanning of the ET lumen. |
| Disposable, Clear Probe Sheath | Maintains sterility, protects the probe from biofluids, and provides a consistent optical window. |
| Topical Vasoconstrictor/Anesthetic (e.g., Lidocaine/Phenylephrine) | Standard clinical preparation for nasal procedures, minimizes discomfort and mucosal swelling for clear imaging. |
| Phosphate-Buffered Saline (PBS) | Keeps ex vivo tissue specimens hydrated to maintain near-physiological optical scattering properties. |
| 4% Paraformaldehyde (PFA) Solution | Fixes tissue specimens post-OCT imaging for precise histological correlation (e.g., H&E staining). |
| Lumen Segmentation Software (e.g., Amira, ITK-SNAP, custom MATLAB/Python) | Enables quantitative analysis of ET lumen patency (cross-sectional area, volume) from 3D OCT data. |
| Calibration Targets (USAF 1951, Mirror) | Validates system resolution and sensitivity, ensuring data integrity and comparability across experiments. |
Patient/Subject Preparation and Positioning for Optimal Access
Within the broader thesis on developing standardized, high-resolution Optical Coherence Tomography (OCT) imaging protocols for the Eustachian Tube (ET) nasopharyngeal region, patient/subject preparation and positioning are critical, non-imaging variables. These factors directly influence the consistency of probe access, the quality of acquired data, and the validity of longitudinal studies assessing ET function or therapeutic intervention efficacy in drug development.
Key Preparation and Positioning Parameters
Optimal access to the nasopharyngeal ET orifice requires control of anatomical and physiological variables. Key parameters and their target states are summarized below.
Table 1: Quantitative Targets for Patient Preparation
| Parameter | Target State / Value | Rationale for OCT Imaging |
|---|---|---|
| Nasal Mucosa Tone | Minimal to no edema, reduced congestion | Reduces anatomical obstruction, improves probe clearance. |
| Mucus Secretion | Minimized | Prevents OCT signal attenuation and lens contamination. |
| Patient Anxiety | Low (e.g., STAI-S score < 37) | Minimizes movement, sympathetic-driven mucosal congestion. |
| ET Dynamicity | Controlled baseline (pre-Valsalva/Toynbee) | Essential for assessing functional response in protocols. |
Table 2: Standardized Positioning Metrics
| Position Component | Protocol Specification | Purpose |
|---|---|---|
| Head Orientation | Head neutral, slight neck extension (~30°), Frankfort plane horizontal. | Aligns nasal floor with nasopharynx for straight probe passage. |
| Body Position | Supine, torso elevated 15-20°. | Compromises between patient comfort and gravitational reduction of mucosal blood flow. |
| Imaging Side | Contralateral nostril to target ET (e.g., image right ET via left nostril). | Utilizes natural nasopharyngeal anatomy for a more favorable probe approach angle. |
| Operator Position | At patient's head, facing cephalad. | Optimal for axial control of the OCT probe during advancement. |
Detailed Experimental Protocols
Protocol 2.1: Pre-Imaging Subject Preparation Objective: To standardize the nasopharyngeal environment and subject state prior to OCT probe insertion.
- Screening & Consent: Exclude subjects with active URI, significant septal deviation, or bleeding disorders. Obtain informed consent per IRB protocol.
- Pharmacological Preparation (60-90 mins prior):
- Administer a topical nasal decongestant (e.g., 0.05% Oxymetazoline spray, 1 spray per nostril, 2 applications 5 mins apart).
- Apply a topical anesthetic (e.g., 2% Lidocaine gel on cotton-tipped applicator or 4% Lidocaine spray) to the proposed navigation nostril.
- Subject Acclimatization (15 mins prior): Subject rests in the procedure room in the standardized body position. Brief explanation of the procedure is given to reduce anxiety.
- Mucus Clearance: Immediately before imaging, instruct subject to gently clear nasal secretions. The operator may use a suction device under direct vision if necessary.
Protocol 2.2: Positioning and Probe Navigation for ET Orifice Access Objective: To reproducibly position the subject and navigate the OCT probe to the nasopharyngeal ET orifice.
- Positioning: Position the subject as per Table 2. Secure head position with a cushioned headrest.
- Nasal Introduction: Under endoscopic guidance (using a 2.7mm or smaller diameter endoscope), introduce the OCT probe (e.g., a rotational or linear pullback catheter) through the prepared contralateral nostril.
- Navigation Path: Advance the probe along the floor of the nasal cavity, past the inferior turbinate, until the tip reaches the posterior wall of the nasopharynx.
- ET Localization: Slowly withdraw the probe while angling the tip laterally. Identify the ET orifice (torus tubarius) using real-time endoscopic visualization. The OCT probe tip is positioned within 2-3mm of the orifice mucosa.
- Stabilization & Imaging: Stabilize the probe sheath manually or using a clamp. Initiate OCT image acquisition (e.g., 3D volumetric scan or dynamic M-mode sequence) per the broader thesis imaging protocol. Record the exact head angle and probe insertion depth for follow-up sessions.
Visualization: Preparation & Positioning Workflow
Title: Workflow for Patient Prep and ET Access
The Scientist's Toolkit: Research Reagent & Material Solutions
Table 3: Essential Materials for Preparation and Positioning Protocols
| Item / Reagent | Function in Protocol | Example Product / Specification |
|---|---|---|
| Topical Nasal Decongestant | Induces vasoconstriction to reduce mucosal edema and improve nasal patency. | Oxymetazoline HCl (0.05%) nasal spray. |
| Topical Mucosal Anesthetic | Provides patient comfort and reduces gag reflex during nasopharyngeal manipulation. | Lidocaine hydrochloride (2% gel or 4% spray). |
| Rigid Nasal Endoscope | Provides real-time visual guidance for safe, accurate OCT probe navigation. | 2.7mm diameter, 0° or 30° angle, high-resolution. |
| OCT Imaging Catheter/Probe | Delivers and collects near-infrared light for cross-sectional imaging of the ET. | Rotational or linear pullback catheter, outer diameter ≤2.0mm. |
| Adjustable Examination Chair/Table | Enables precise and reproducible subject positioning per Table 2 metrics. | Chair with adjustable backrest and headrest. |
| Angulation Measurement Tool | Quantifies and standardizes head extension angle. | Simple goniometer or digital inclinometer app. |
| Subject Anxiety Assessment | Quantifies pre-procedural anxiety as a confounding variable. | State-Trait Anxiety Inventory (STAI) Form S-1. |
1. Introduction & Context
This document provides detailed application notes and protocols for the precise navigation of optical coherence tomography (OCT) probes within the nasopharynx, specifically targeting the Eustachian Tube (ET) orifice and surrounding tissues. The methodology is framed within a broader thesis on developing a standardized, high-resolution imaging technique for in vivo assessment of ET function, mucosal dynamics, and therapeutic intervention outcomes. Mastery of these probe insertion and guidance techniques is critical for generating reproducible, high-fidelity OCT data in a challenging anatomical region.
2. Quantitative Data Summary: Anatomical & Imaging Parameters
Table 1: Key Anatomical Dimensions of the Adult Nasopharynx & Eustachian Tube Orifice Relevant for Probe Navigation
| Parameter | Mean Value (±SD) | Range | Measurement Method | Relevance to Probe Design/Navigation |
|---|---|---|---|---|
| Distance from Nostril to ET orifice | 78.2 mm (±4.8) | 70-90 mm | Endoscopic measurement | Determines minimum working length of probe/sheath. |
| Height of Rosenmüller's Fossa | 11.5 mm (±2.1) | 8-16 mm | CT/MRI reconstruction | Primary landmark for ET orifice localization; probe must avoid deep insertion into fossa. |
| Antero-Posterior Diameter of Nasopharynx | 31.0 mm (±3.5) | 25-38 mm | CT measurement | Defines the general workspace for probe manipulation. |
| Typical ET Orifice Diameter (at rest) | 4.2 mm (±1.3) | 2-8 mm | Endoscopic visualization | Target for imaging; probe diameter must be <2mm for non-occlusive imaging. |
Table 2: Performance Specifications of Representative OCT Probes for Nasopharyngeal Imaging
| Probe Type | Outer Diameter | Lateral Resolution | Axial Resolution | Scan Rate (A-line/sec) | Key Advantage for Navigation | Primary Limitation |
|---|---|---|---|---|---|---|
| Rotational SS-OCT Catheter | 1.8 - 2.2 mm | ~20-30 µm | ~10-15 µm | 50,000 - 200,000 | 360° cross-sectional view aids spatial orientation. | Stiffness can make negotiation of posterior choanae challenging. |
| MEMS Scanner-based Forward-Viewing Probe | 1.5 - 2.0 mm | ~15-25 µm | ~10-15 µm | 100,000 - 500,000 | Forward-viewing simplifies direct targeting of orifice. | Limited field of view per scan; requires more user guidance. |
| Balloon-centered Imaging Catheter | 3.0 mm (deflated) | ~30-40 µm | ~15-20 µm | 50,000 - 100,000 | Centers probe in lumen, stabilizing image. | Larger size; may cause discomfort or obstruct the view. |
3. Experimental Protocols
Protocol 3.1: Standardized Patient Preparation and Positioning for In Vivo OCT Imaging
- Objective: To ensure consistent, safe, and optimal anatomical exposure for nasopharyngeal probe insertion.
- Materials: Topical anesthetic/decongestant (e.g., Lidocaine 2% + Oxymetazoline 0.05%), cotton-tipped applicators, suction apparatus, examination chair, headrest.
- Procedure:
- Obtain informed consent and position the subject in a standard examination chair, reclined at ~70 degrees, with head supported.
- Under direct vision (using a headlight or nasal speculum), apply topical anesthetic/decongestant to the middle meatus and posterior nasopharynx via cotton applicators. Wait 5-10 minutes for full effect.
- Instruct the subject to breathe slowly through the mouth and avoid swallowing during probe insertion unless instructed.
- Confirm adequate mucosal contraction and anesthesia before proceeding.
Protocol 3.2: Endoscope-Guided OCT Probe Insertion and ET Orifice Localization
- Objective: To safely navigate the OCT probe to the nasopharyngeal ET orifice using real-time endoscopic visualization.
- Materials: Flexible digital nasopharyngoscope (outer diameter ≤3mm), sterile OCT imaging probe/sheath, video monitor, saline for lens cleaning.
- Procedure:
- Insert the flexible endoscope along the floor of the nasal cavity to the nasopharynx.
- Identify key landmarks: posterior septum, contralateral ET orifice, and Rosenmüller's fossa.
- Under continuous endoscopic view, advance the OCT probe through the same or contralateral nostril, keeping its tip within the endoscopic field of view at all times.
- Use the endoscope to visually "guide" the OCT probe tip toward the target ET orifice. Minor adjustments are made by rotating or slightly retracting/advancing the probe.
- Once the OCT probe tip is positioned directly facing the ET orifice, initiate imaging. The endoscope can be gently retracted slightly to avoid interference.
Protocol 3.3: Protocol for Acquiring 4D (3D + Time) OCT Data of ET Dynamical Movement
- Objective: To capture time-resolved, volumetric data of ET opening/closing dynamics during swallowing or other maneuvers.
- Materials: Synchronized OCT imaging system, swallow trigger (e.g., EMG sensor, video recording), data acquisition software with gating capability.
- Procedure:
- Position the OCT probe per Protocol 3.2, ensuring stable placement.
- Set the OCT system to a high-speed, volumetric scan mode (e.g., repeated 3D raster scans).
- Synchronize the start of a continuous OCT acquisition with a trigger signal (e.g., from a submental EMG sensor placed on the subject).
- Instruct the subject to perform a dry swallow or a standardized Valsalva maneuver upon an audible cue, which is also recorded.
- Acquire data for 10-15 seconds, capturing multiple swallow events.
- Post-process using the trigger signals to temporally align and segment the 4D dataset for analysis of lumen area vs. time.
4. Visualization: Experimental Workflow & Pathway
Diagram Title: Workflow for Endoscope-Guided OCT Imaging of ET
5. The Scientist's Toolkit: Research Reagent & Material Solutions
Table 3: Essential Materials for Nasopharyngeal OCT Probe Navigation Studies
| Item/Category | Example Product/Specification | Function in Research |
|---|---|---|
| Topical Anesthetic/Decongestant | Lidocaine (2-4%) + Phenylephrine or Oxymetazoline spray/gel. | Reduces patient discomfort and reflexive movement; shrinks nasal mucosa for clearer passage and wider working channel. |
| Flexible Nasopharyngoscope | Digital, outer diameter ≤3mm, with working channel. | Provides real-time visual guidance for safe probe navigation and definitive anatomical landmark identification. |
| OCT Imaging Probe Sheath | Disposable, transparent, sterile polymer sheath (e.g., PE, FEP). | Protects the reusable OCT probe from contamination; allows for easy cleaning between subjects while maintaining optical clarity. |
| Medical-Grade Optical Gel | Sterile, aqueous, ultrasound-compatible gel. | Acts as an optical coupling medium between the probe sheath tip and the tissue, eliminating air gaps that cause signal artifacts. |
| Probe Positioning Fixation Device | Adjustable stereotactic arm or custom-designed nasal guide. | Stabilizes the OCT probe during prolonged acquisitions, minimizing motion artifacts for high-quality 4D functional imaging. |
| Swallowing Event Trigger | Surface EMG electrodes or accelerometer. | Provides an electronic signal synchronized to OCT acquisition to gate and analyze dynamic ET opening events (Protocol 3.3). |
| Anatomical Phantom | 3D-printed nasal/ nasopharyngeal model based on CT data. | Allows for practice and optimization of probe navigation techniques without patient involvement, improving procedural skill. |
Within the context of research for developing a standardized Optical Coherence Tomography (OCT) technique for imaging the Eustachian tube (ET) and nasopharyngeal region, a rigorously defined image acquisition protocol is paramount. This protocol ensures reproducibility, enables quantitative comparison across subjects and studies, and is critical for assessing therapeutic interventions in drug development. This document outlines the core scanning patterns and system parameters required for consistent, high-fidelity volumetric imaging of this complex anatomical region.
OCT System Configuration & Calibration Protocol
Prior to any in vivo scanning, the OCT system must be configured and calibrated using the following standardized protocol.
System Pre-Scan Calibration
Objective: To verify system performance and ensure alignment accuracy. Materials: Pre-characterized test phantom with known reflectivity and geometrical features (e.g., a layered polymer or a microsphere-embedded agarose block). Procedure:
- Mount the phantom in a fixed position relative to the scanner.
- Acquire a 3D volume scan using the system's default settings.
- Analyze the acquired data:
- Axial Resolution: Measure the full-width at half-maximum (FWHM) of the point spread function from a highly reflective interface.
- Lateral Resolution: Measure the FWHM of the beam profile from a sub-resolution scatterer.
- Signal-to-Noise Ratio (SNR): Calculate as 20*log10(Mean Signal in a Specified Layer / Mean Noise in a Signal-Void Region).
- Geometric Fidelity: Measure known distances (e.g., layer thickness) in the phantom against their ground truth.
- Compare results against the manufacturer's specifications and internal laboratory benchmarks. Document all values. Proceed only if all parameters are within acceptable tolerances (±10%).
Key Research Reagent Solutions:
| Item | Function |
|---|---|
| Structured Test Phantom | Provides known reflectivity profiles and spatial features for systematic resolution, SNR, and geometric distortion testing. |
| Index Matching Fluid | Applied between the OCT distal optics and phantom/ tissue to minimize refractive index mismatch, preserving accurate depth measurements. |
| Alignment Fixture | A mechanical stage to rigidly and reproducibly position phantoms or ex vivo specimens for calibration scans. |
Standardized Scanning Patterns for the ET/ Nasopharynx
Two primary scanning patterns are mandated for comprehensive assessment: a large-field survey and a targeted high-resolution scan.
Pattern A: Wide-Field Volumetric Survey
Purpose: To capture the global anatomical context of the nasopharynx, torus tubarius, and ET orifice.
- Pattern Type: Raster scan (serpentine).
- Field of View (FOV): 10 mm (X) x 10 mm (Y).
- Lateral Sampling Density: 512 A-scans per B-scan, 512 B-scans per volume.
- Depth (Z) Range: 3.0 mm in tissue (adjust based on system and index).
- Scan Rate: Maximize available rate (e.g., 100-200 kHz) to minimize motion artifact.
- Averaging: 2-4 frame averaging per B-scan location to improve SNR if patient motion is minimal.
Pattern B: Targeted High-Resolution Scan of ET Lumen
Purpose: To resolve detailed mucosal morphology and luminal patency of the proximal (pharyngeal) Eustachian tube.
- Pattern Type: Concentric circular scans OR dense radial B-scans.
- FOV Diameter: 3-5 mm, centered on the ET orifice.
- Lateral Sampling: 1024 A-scans per B-scan for circular pattern; 256-512 radial B-scans over 360° for radial pattern.
- Depth (Z) Range: 2.5 mm in tissue.
- Averaging: 8-16 frame averaging to maximize detail, using a short dwell time.
| Parameter | Pattern A (Survey) | Pattern B (Target) | Unit | Rationale |
|---|---|---|---|---|
| Scan Pattern | Rectangular Raster | Radial/Circular | - | Context vs. Symmetric Detail |
| X-Y FOV | 10 x 10 | 3-5 (diameter) | mm | Anatomical Coverage |
| A-scans/B-scan | 512 | 1024 (Circular) | count | Lateral Sampling Density |
| B-scans/Volume | 512 | 256 (Radial) | count | Volumetric Sampling |
| Theoretical Lateral Resolution | ~15-20 | ~10-15 | µm | Detail Resolution |
| Depth Range (in air) | ~4.2* | ~3.5* | mm | Accounts for Tissue (n~1.4) |
| Frame Averaging | 2-4 | 8-16 | count | SNR vs. Motion Trade-off |
| Total Acquisition Time | < 5 | < 8 | seconds | Minimizes Motion |
*Depth in tissue = Depth in air / Refractive Index (assume n~1.4 for soft tissue).
In VivoImaging Protocol for Human Subjects
Objective: To acquire standardized OCT volumes of the nasopharynx and ET region from consented research participants.
Pre-Imaging Preparation:
- Subject Preparation: Topical decongestant (e.g., oxymetazoline 0.05%) and anesthetic (e.g., lidocaine 2%) applied via nasal spray to the target nostril 5-10 minutes prior.
- Positioning: Subject seated in an examination chair with head stabilized in a headrest. Instructions are given to breathe gently and avoid swallowing during scan acquisition.
- Scope Insertion: A sterile, single-use OCT imaging sheath is fitted over a sterilized OCT distal imaging probe. The probe is gently introduced through the nasal cavity under endoscopic guidance until the ET orifice/torus tubarius is in view.
Image Acquisition Workflow:
- Endoscopic Localization: Use the concurrent video endoscope to identify the torus tubarius and ET orifice.
- Protocol Execution: Initiate automated scanning sequences in order: a. Acquire Pattern A (Wide-Field Survey). b. Reposition probe to center ET orifice. c. Acquire Pattern B (Targeted High-Resolution).
- Repeat: If imaging bilaterally, repeat full protocol for the contralateral side with a new sterile sheath.
- Data Labeling: Immediately label data files with unique subject ID, date, time, scan pattern, and anatomical side (Left/Right).
Diagram Title: In Vivo OCT Imaging Protocol Workflow
Post-Processing & Quality Control Protocol
Mandatory Processing Steps:
- Fixed-Path Delay Correction: Apply system-specific compensation for optical path length in the imaging sheath/air gaps.
- Spectral Resampling/DC Subtraction: Use calibrated algorithms to correct non-linear k-space sampling and remove fixed-pattern noise.
- Dispersion Compensation: Apply numerical compensation to match the calibration arm's dispersion.
- Image Registration (Optional but Recommended): Use a rigid or affine algorithm to correct for minor intrascan motion (especially critical for Pattern B with high averaging).
Quality Control Metrics (Per Volume Scan):
| Metric | Calculation Method | Acceptance Criterion |
|---|---|---|
| Signal Roll-Off | Measure dB drop from surface to maximum depth. | Shall be > 90% of phantom-based benchmark. |
| Contrast-to-Noise (CNR) | (µs - µb) / √(σs² + σb²) for a mucosal layer vs. underlying tissue. | Must be ≥ 5 dB for analysis-ready data. |
| Presence of Motion Artifacts | Visual inspection for B-scan discontinuities or striations. | Major artifacts disqualify scan; minor artifacts noted. |
Data Archiving & Metadata Standards
All acquired data must be archived with the following minimum metadata in a machine-readable format (e.g., JSON sidecar file):
- Subject Study ID
- Acquisition Date/Time
- Scan Pattern (A/B)
- Anatomical Location/Side
- OCT System Model & Serial Number
- Core Scan Parameters (as per tables above)
- Calibration Date & Performance Metrics
- Operator ID
- Post-Processing Steps Applied
Conclusion: Adherence to this detailed protocol for scanning patterns and parameters will standardize image acquisition in research investigating the ET and nasopharynx using OCT. This forms the essential foundation for reliable quantitative analysis, longitudinal monitoring, and robust assessment of drug-induced morphological changes, directly supporting the broader thesis aim of establishing a validated OCT technique for this region.
Application Notes for OCT Imaging of the Eustachian Tube Nasopharyngeal Region
Within a thesis focused on advancing Optical Coherence Tomography (OCT) techniques for imaging the Eustachian Tube (ET) nasopharyngeal region, a robust post-processing workflow is critical for transforming volumetric data into quantifiable, three-dimensional models. This enables precise morphological analysis, essential for researching ET dysfunction, assessing therapeutic interventions, and supporting targeted drug delivery development. The following notes and protocols detail the sequential steps of segmentation, measurement, and 3D reconstruction.
Image Segmentation Protocol
Objective: To accurately delineate the ET lumen and surrounding mucosal tissues from 3D-OCT volumetric datasets.
Methodology:
- Data Preprocessing: Load acquired OCT volumes (e.g., .vol, .img files). Apply median filtering (3x3 kernel) to reduce speckle noise while preserving edges. Correct for intensity decay with depth using a depth-dependent attenuation compensation algorithm.
- Semi-Automatic Segmentation: Utilize a hybrid approach.
- Initialization: Manually seed points within the lumen and mucosal region on key B-scans (e.g., every 10th slice).
- Algorithm Execution: Apply a graph-cut or region-growing algorithm (e.g., Random Walers) initialized by the manual seeds. The algorithm propagates segmentation based on pixel intensity and gradient information.
- Quality Control & Correction: Manually review the segmented contours in all orthogonal planes (axial, coronal, sagittal). Use manual brush and eraser tools in software (e.g., ITK-SNAP, 3D Slicer) to correct any obvious errors, particularly at regions of high curvature or low signal-to-noise ratio.
- Output: Generate a binary label map for the primary structure of interest (e.g., ET Lumen = Label 1, Mucosa = Label 2).
Key Quantitative Segmentation Metrics: Table 1: Common Segmentation Performance Metrics (Evaluated against manually segmented ground truth)
| Metric | Formula | Interpretation in ET OCT Context | ||||||
|---|---|---|---|---|---|---|---|---|
| Dice Similarity Coefficient (DSC) | ( DSC = \frac{2 | X \cap Y | }{ | X | + | Y | } ) | Measures volumetric overlap accuracy. Target: >0.90 for reliable analysis. |
| Hausdorff Distance (HD) | ( HD = \max{ \sup{x \in X} \inf{y \in Y} d(x,y), \sup{y \in Y} \inf{x \in X} d(x,y) } ) | Measures the maximum boundary deviation. Critical for assessing lumen patency accuracy. | ||||||
| Precision | ( Precision = \frac{TP}{TP + FP} ) | Proportion of segmented voxels that are true positives. High precision minimizes false lumen tissue. | ||||||
| Recall (Sensitivity) | ( Recall = \frac{TP}{TP + FN} ) | Proportion of true tissue voxels that are detected. High recall ensures complete lumen capture. |
Diagram: Segmentation & Analysis Workflow
Morphometric Measurement Protocol
Objective: To extract quantitative parameters from segmented ET structures relevant to physiological function and drug delivery.
Methodology:
- Lumen Dimensional Analysis:
- Using the binary label map, extract the centerline of the ET lumen.
- Compute the cross-sectional area perpendicular to this centerline at user-defined intervals (e.g., every 50 µm).
- Derive the minimum, maximum, and mean lumen area for each sample.
- Calculate the hydraulic diameter for each cross-section: ( D_h = \frac{4 \times Area}{Perimeter} ).
- Mucosal Thickness Mapping:
- For each point on the lumen boundary, compute the Euclidean distance to the outer mucosal boundary (segmented label).
- Generate a 2D thickness map unwrapped along the lumen centerline.
- Report mean and standard deviation of mucosal thickness for defined sub-regions (pharyngeal, cartilaginous, tympanic).
- Volume Calculation: Compute the total volume in µL or mm³ for the segmented lumen and mucosal tissue using voxel counting and known voxel dimensions.
Standardized Measurement Output Table: Table 2: Morphometric Parameters from ET OCT Segmentation
| Parameter | Unit | Measurement Technique | Clinical/Biological Relevance |
|---|---|---|---|
| Min. Lumen Area | mm² | Minimal cross-sectional area from centerline analysis. | Key indicator of functional obstruction or collapse. |
| Mean Hydraulic Diameter | mm | Mean of ( D_h ) across the length. | Governs fluid and air flow dynamics; critical for insufflation drug delivery. |
| Mean Mucosal Thickness | mm | Average distance from lumen to tissue boundary. | Indicator of inflammation, edema, or therapeutic response. |
| Lumen Volume | µL | Voxel summation of lumen label. | Quantifies total air space volume. |
| Mucosal Surface Area | mm² | Isosurface generation from lumen label. | Relevant for quantifying epithelial exposure in topical therapy. |
3D Surface Reconstruction & Visualization Protocol
Objective: To generate interactive 3D models from segmented labels for qualitative assessment and presentation.
Methodology:
- Surface Mesh Generation: Apply a marching cubes algorithm to the binary label map to create a polygonal mesh (e.g., .stl, .ply format). Apply smoothing (e.g., Laplacian smoothing) conservatively to reduce stair-step artifacts without compromising anatomical accuracy.
- Model Texturing: Map the original OCT intensity values or a derived parameter (e.g., local thickness) onto the mesh as a vertex color or texture to create a physiologically informative model.
- Visualization & Export: Render the 3D model in software (e.g., ParaView, MeshLab). Enable clipping planes and cross-sectional views. Export for use in presentations, publications, or for computational fluid dynamics (CFD) simulation preprocessing.
Diagram: From Segmentation to 3D Model
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials & Software for OCT ET Post-Processing
| Item Name | Category | Function & Application Notes |
|---|---|---|
| ITK-SNAP | Open-Source Software | Primary tool for manual/semi-automatic segmentation of OCT volumes. Supports 3D rendering and label map export. |
| 3D Slicer | Open-Source Platform | Comprehensive platform for advanced segmentation, registration, and quantitative analysis. Ideal for complex workflow scripting. |
| ImageJ/Fiji | Open-Source Software | For basic preprocessing (filtering), batch processing of 2D slices, and macro-based analysis. |
| ParaView | Open-Source Software | High-performance 3D visualization and rendering of exported surface meshes. Essential for creating publication-quality figures. |
| MATLAB (with Image Processing Toolbox) | Commercial Software | Custom algorithm development for novel segmentation, centerline extraction, and batch metric calculation. |
| PyTorch/TensorFlow | Open-Source Libraries | For developing and deploying deep learning-based segmentation models (e.g., U-Net) to automate processing of large OCT datasets. |
| In-house MATLAB/Python Scripts | Custom Code | For calculating DSC, HD, lumen area, thickness, and generating standardized output tables (as in Table 1 & 2). |
| High-Performance Workstation | Hardware | Equipped with dedicated GPU (e.g., NVIDIA RTX series) to accelerate volume rendering, segmentation, and deep learning inference. |
Overcoming Challenges: Optimizing OCT Image Quality and Data Interpretation
Within the thesis on Optical Coherence Tomography (OCT) imaging of the Eustachian tube (ET) and nasopharyngeal region, the optimization of image quality is paramount for accurate anatomical and functional assessment. This region presents unique challenges, including inherent patient motion, the presence of mucus, and limited signal penetration through complex tissue layers. These common artifacts can severely degrade image resolution, contrast, and diagnostic utility. These Application Notes detail the characterization of these artifacts and provide validated protocols for their mitigation to enhance the reliability of OCT data in otolaryngology research and drug development.
Artifact Characterization & Quantitative Impact
Table 1: Characterization and Impact of Common OCT Artifacts in ET Imaging
| Artifact Type | Primary Cause | Effect on OCT Image (A-scan/B-scan) | Quantitative Metric Impact |
|---|---|---|---|
| Motion | Patient breathing, swallowing, pulse. Probe instability. | Blurring, discontinuities, distortion. Replication of structures. | SNR Reduction: 5-15 dB. Axial Resolution Loss: Up to 2x theoretical. Lateral Misregistration: 10-50 µm. |
| Mucus | Secretions coating the mucosal surface. | Strong, irregular surface reflection. Signal attenuation. Sub-surface obscuration. | Surface Signal Spike: +20-30 dB above tissue. Penetration Depth Reduction: 30-60%. |
| Signal Penetration Limit | Light scattering/absorption in thick, glandular tissue & cartilage. | Exponential intensity decay. Loss of deep structural detail (e.g., ET lumen, cartilage). | Effective Imaging Depth: 1.0-1.5 mm in mucosa; <2.0 mm near cartilage. Roll-off: >25 dB/mm. |
Experimental Protocols for Artifact Mitigation
Protocol 3.1: Dual-Angle Gated Acquisition for Motion Mitigation
Objective: To synchronize OCT A-scan acquisition with physiological cycles to minimize motion artifacts. Materials: Spectral-Domain OCT system, specialized nasopharyngeal probe, physiological monitor (ECG/pressure sensor), data acquisition card, processing software (e.g., MATLAB). Procedure:
- Instrument Setup: Modify OCT system trigger input to accept an external TTL signal.
- Gating Signal Acquisition: Place a pressure sensor at the external auditory canal or use ECG leads to detect cardiac cycle. Route the analog signal to the data acquisition card.
- Synchronization: Using custom software, generate a TTL trigger pulse at a consistent phase of the physiological cycle (e.g., end-diastole for minimal carotid pulse motion).
- Data Acquisition: Program the OCT system to acquire a single A-scan per trigger. For B-scans, acquire multiple A-scans at the same spatial location over multiple cycles, then average.
- Post-Processing: Apply software-based image registration algorithms (e.g., normalized cross-correlation) to residual motion in averaged frames.
Protocol 3.2: Mucosclear-Assisted Imaging Protocol
Objective: To temporarily clear or index-match surface mucus for improved visualization of the epithelial surface and sub-mucosal structures. Materials: 1-3% acetylcysteine solution or sterile saline, controlled-pressure spray device, suction microcatheter, OCT-compatible topical anesthetic (e.g., lidocaine spray), sterile swabs. Procedure:
- Pre-Imaging Preparation: Under endoscopic guidance, administer topical anesthetic to the nasopharyngeal ostium of the ET.
- Mucus Clearance: Gently apply 0.5-1 mL of acetylcysteine solution via spray. Wait 60 seconds for mucolytic action.
- Controlled Removal: Use low-power suction to clear dissolved secretions without causing tissue trauma or inducing edema.
- Immediate OCT Imaging: Acquire OCT datasets within 2-3 minutes post-clearance. A saline control arm should be included in study design.
- Safety & Monitoring: Document any mucosal changes post-procedure. Limit repeated applications.
Protocol 3.3: Depth-Encoded Compound Imaging for Enhanced Penetration
Objective: To improve effective signal penetration and contrast by combining scans from multiple entry angles. Materials: OCT system, side-viewing rotary probe with programmable pullback, custom probe sheath with calibrated refractive elements, 3D registration software. Procedure:
- Multi-Angle Scan Planning: Define two distinct imaging paths (angles) to the same region of interest (ROI), e.g., perpendicular to and tangential along the ET lumen.
- Data Acquisition: Perform sequential 3D OCT scans from each predefined angle using a calibrated rotary pullback probe.
- Data Co-Registration: Use surface landmarks and fiduciary markers to spatially align the two volumetric datasets using rigid or affine transformation algorithms.
- Pixel-Level Fusion: For each voxel in the combined volume, assign the maximum intensity value from the co-registered datasets:
I_fused(x,y,z) = max[I_angle1(x,y,z), I_angle2(x,y,z)]. - Validation: Compare the contrast-to-noise ratio (CNR) and visualized depth of key structures (e.g., submucosal glands) between fused and single-angle images.
Visualization of Workflows and Relationships
Title: OCT Artifact Mitigation Decision Workflow
Title: Gated Acquisition Signal Pathway
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for OCT Imaging of the Nasopharyngeal/ET Region
| Item | Function & Relevance to Artifact Mitigation | Example/Specification |
|---|---|---|
| Spectral-Domain OCT Engine | Core imaging system. High A-scan rate (>50kHz) reduces motion sensitivity. Central wavelength ~1300nm for deeper penetration in tissue. | Thorlabs TELESTO-II, Santec IVS-3000 |
| Side-Viewing Rotary Probe | Enables circumferential scanning inside the ET nasopharyngeal orifice. Crucial for compound imaging (Protocol 3.3). | Custom-built, diameter ≤2.0mm, working distance 2-5mm |
| External Trigger Module | Accepts physiological signals to gate image acquisition, implementing Protocol 3.1. | National Instruments DAQ card, LabVIEW interface |
| Acetylcysteine (NAC) Solution | Mucolytic agent. Breaks disulfide bonds in mucus glycoproteins, clearing surface artifact (Protocol 3.2). | 1-3% solution in sterile saline, prepared under pharmacy protocol |
| Controlled Micro-Spray Device | Delivers mucolytic or saline solution precisely to the ET orifice without causing trauma or overwhelming the site. | LMA MAD Nasal or similar with calibrated pressure |
| Suction Microcatheter | Removes liquefied mucus post-application without inducing mucosal injury or edema. | Diameter <1mm, flexible, sterile single-use |
| Index-Matching Gel | Optional. Reduces surface specular reflection. Can be used sparingly if mucus clearance is incomplete. | Glycerol-based, sterile, biocompatible |
| 3D Co-Registration Software | Essential for fusing multi-angle datasets in Protocol 3.3 to overcome penetration limits. | Amira, 3D Slicer with custom plugins, MATLAB Image Processing Toolbox |
Application Notes
Optical Coherence Tomography (OCT) imaging of the Eustachian tube (ET) and nasopharyngeal region presents unique anatomical and technical challenges. This complex, narrow, and tortuous pathway requires meticulous probe selection to achieve diagnostic-quality images while ensuring patient safety and procedural feasibility. The core parameters—flexibility, diameter, and resolution—are intrinsically linked and must be balanced against the specific clinical or research question.
Key Considerations:
- Flexibility: Essential for navigating the ~35-45 mm path from the nasal vestibule to the tympanic orifice of the ET without causing trauma. Excessive flexibility, however, can compromise pushability and rotational control.
- Outer Diameter: Dictated by the anatomy. The narrowest point is the isthmus of the ET (1-2 mm in diameter). Probes must typically be <2.0 mm, with sub-1.0 mm ideal for unobstructed passage. Smaller diameters often come at the cost of reduced numerical aperture and signal-to-noise ratio.
- Axial & Lateral Resolution: Critical for visualizing layered microstructures (e.g., epithelium, lamina propria, mucous glands) and goblet cell density. High resolution (>5 µm axial) is desirable but is constrained by light source bandwidth, probe optics, and diameter limitations.
Recent advancements in OCT catheter technology, such as forward-viewing, side-viewing, and micro-optic components, have expanded the possibilities for intraluminal ET imaging. The choice between time-domain (TD-OCT), spectral-domain (SD-OCT), and swept-source (SS-OCT) systems further influences this balance, with SS-OCT offering superior imaging depth and speed for dynamic functional assessment.
Quantitative Parameter Comparison
Table 1: OCT System & Probe Parameter Trade-offs for ET Imaging
| Parameter | Typical Range for ET Imaging | Impact on Flexibility | Impact on Resolution | Key Trade-off |
|---|---|---|---|---|
| Probe Outer Diameter | 0.5 mm – 2.0 mm | Smaller diameter increases flexibility. | Smaller diameter limits optics, often reducing lateral resolution. | Diameter vs. Optical Performance |
| Central Wavelength | 1300 nm – 1400 nm (common) | Minimal direct impact. | Longer wavelength increases penetration depth but may reduce axial resolution. | Penetration vs. Axial Resolution |
| Axial Resolution | 3 µm – 15 µm in tissue | No direct impact. | Defines layer differentiation capability. Higher resolution requires broader bandwidth sources. | Resolution vs. System Cost/Complexity |
| Lateral Resolution | 10 µm – 30 µm in tissue | Influenced by GRIN lens/optic size. Smaller optics aid flexibility. | Directly linked to spot size. Improved by higher NA, but depth of focus reduces. | Resolution vs. Depth of Focus |
| A-Scan Rate | 50 kHz – 500 kHz+ | No direct impact. | Enables faster B-scans, reducing motion artifact for functional studies. | Speed vs. Sensitivity (for SD/SS-OCT) |
| Bending Radius | < 10 mm (critical) | Essential for navigation. Achieved via thin fiber core and flexible sheath. | Tight bending can induce signal loss or distortion. | Navigability vs. Signal Fidelity |
Table 2: Probe Type Suitability for ET Imaging Tasks
| Probe Type | Typical Diameter | Max Bending Flexibility | Best Suited For | Resolution Limitation |
|---|---|---|---|---|
| Rotational Side-Viewing | 0.9 – 2.0 mm | Moderate (stiff drive shaft) | Cross-sectional lumen morphology, static 3D pullbacks. | Lateral resolution degrades from center. |
| Forward-Viewing (Distal Scan) | 1.5 – 2.5 mm | Lower (distal scanner) | Visualizing ET orifice, guiding insertion. | Often lower resolution due to scanner size. |
| Forward-Viewing (Proximal Scan) | 0.5 – 1.0 mm | Very High (bare fiber/GRIN) | Navigating tortuous segments, ultra-narrow lumens. | Limited depth of field, fixed focus. |
| Micro-optic (Lensed Fiber) | < 0.8 mm | Extreme | Pediatric applications, distal ET assessment. | Significant trade-offs in FOV and resolution. |
Experimental Protocols
Protocol 1:Ex VivoET Lumen Morphometry and Layer Thickness Measurement
Objective: To quantitatively assess epithelial and subepithelial layer thickness in excised human or animal ET tissue using high-resolution OCT.
Materials:
- OCT System: Spectral-Domain or Swept-Source OCT engine with axial resolution ≤ 7 µm.
- Probe: Rotational side-viewing catheter (OD ~1.2 mm) for lumen profiling OR forward-viewing micro-optic probe (OD ~0.8 mm) for targeted imaging.
- Sample: Freshly excised porcine or human cadaveric ET tissue segment, stabilized in phosphate-buffered saline (PBS).
- Stages: Motorized linear translation stage and rotational stage for probe positioning.
- Software: Custom or commercial software for image analysis (e.g., ImageJ, Amira, OsiriX).
Procedure:
- System Calibration: Calibrate the OCT system using a mirror in air and water to determine axial and lateral scaling (µm/pixel).
- Probe Insertion & Alignment: Mount the ET tissue sample in a custom holder submerged in PBS. Under microscopic guidance, insert the OCT probe into the pharyngeal orifice and advance to a pre-marked region of interest (ROI).
- Image Acquisition:
- For 2D cross-sections: Acquire radial scans (500 A-scans/B-scan) at fixed rotational positions.
- For 3D data: Perform a pullback over 5-10 mm length, acquiring 1000 B-scans with 10 µm intervals.
- Image Processing: Apply standard filters (median, Gaussian) to reduce speckle noise. Use N-Scan averaging (e.g., 5 frames) if system supports live averaging.
- Morphometric Analysis:
- Manually or semi-automatically segment the luminal boundary.
- Measure epithelial thickness by delineating the hyper-reflective epithelial layer from the underlying, less reflective lamina propria at 8-12 equidistant points around the lumen circumference per B-scan.
- Calculate mean, minimum, and maximum thickness for each cross-section.
- Validation: Correlate OCT measurements with matched histological sections (H&E stain) using landmark registration.
Protocol 2:In VivoDynamic ET Function Assessment
Objective: To visualize and measure ET lumen opening dynamics (dilation, mucociliary transport) in an anesthetized animal model.
Materials:
- OCT System: High-speed Swept-Source OCT (A-scan rate > 200 kHz) for capturing dynamics.
- Probe: Ultra-flexible, forward-viewing micro-optic probe (OD ≤ 1.0 mm) integrated with a guiding sheath/channel.
- Animal Model: Anesthetized rabbit or murine model with stabilized head position.
- Stimulation Setup: Air pressure delivery system (for Toynbee or Valsalva simulation) with calibrated manometer.
- Synchronization Hardware: Data acquisition (DAQ) card to synchronize OCT frame triggers with pressure stimulus events.
Procedure:
- Probe Placement: Under endoscopic guidance, advance the probe within a guiding sheath to the nasopharyngeal ET orifice. Position the probe tip 1-2 mm from the ET mucosal surface.
- Synchronization Setup: Program the DAQ system to send a TTL pulse to the OCT system and the pressure controller to initiate simultaneous acquisition and stimulus.
- Dynamic Image Acquisition:
- Acquire M-mode scans (repeated A-scans at one spatial location) at 1 kHz for 10 seconds during a controlled air pulse stimulus (e.g., 500 ms pulse of 20 mm H₂O).
- Acquire repeated B-scans (10 frames/second) to capture 2D cross-sectional opening.
- Data Analysis:
- In M-mode images, track the leading edge of the mucosal surface to plot lumen diameter change over time. Calculate opening duration, latency, and maximum diameter.
- Use particle image velocimetry (PIV) algorithms on B-scan sequences to track and quantify mucus flow velocity (µm/s).
- Pharmacological Intervention (Optional): Administer a topical drug (e.g., nasal decongestant like oxymetazoline) or irritant. Repeat acquisition after 15 minutes to compare functional parameters.
Diagrams
Diagram 1: Probe Parameter Balance Logic
Diagram 2: Experimental Workflow for ET OCT
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions for ET-OCT Studies
| Item | Function & Rationale |
|---|---|
| High-Speed SS-OCT Engine (e.g., 1300 nm, >200 kHz) | Enables capture of rapid ET opening dynamics and reduces motion artifacts in vivo. Swept-source technology offers better depth penetration. |
| Flexible, Side-Viewing Catheter Probe (OD: 1.0-1.5 mm) | Workhorse for 3D luminal morphology. Rotational design provides 360° cross-sectional views essential for measuring lumen area and layer uniformity. |
| Ultra-Thin Forward-Viewing Probe (OD: ≤0.8 mm) | Critical for navigating the narrowest ET segments and for pediatric or murine model studies where minimal invasiveness is paramount. |
| Integrated Guidance Sheath/Channel | Provides stability for the fragile OCT probe fiber, allows for controlled pullback, and may integrate suction or fluid delivery for clearing mucus. |
| Motorized Pullback & Rotation Stage | Ensures precise, standardized, and repeatable probe translation and rotation during 3D data acquisition for volumetric analysis. |
| TTL-Controlled Pressure Stimulus System | Delivers calibrated air pressure pulses to the nasal cavity to provoke ET opening, synchronized with OCT acquisition for functional studies. |
| Mucociliary Transport Analysis Software | Custom or commercial PIV/optical flow algorithms applied to B-scan sequences to quantify mucus flow speed as a biomarker of ET health. |
| Histology Registration Software (e.g., 3D Slicer) | Software capable of co-registering OCT image volumes with serially sectioned histology slides using fiduciary markers for validation. |
Optimizing Scan Settings for Mucosal Layers vs. Cartilaginous Structures
1. Introduction and Thesis Context This application note details specialized Optical Coherence Tomography (OCT) protocols for imaging the complex morphology of the eustachian tube (ET) nasopharyngeal region. It is framed within a broader thesis aimed at establishing a standardized, high-resolution OCT imaging technique for this anatomically challenging site. The core challenge lies in simultaneously optimizing image contrast and penetration depth for two distinct tissue types: the superficial, layered mucosal epithelium and the deeper, scattering cartilaginous structures of the torus tubarius. This document provides comparative quantitative data, validated experimental protocols, and visualization tools tailored for researchers, scientists, and drug development professionals investigating ET physiology, pathophysiology, and therapeutic delivery.
2. Quantitative Comparison of Optimized Scan Settings
Table 1: Core OCT Parameter Optimization for Mucosal vs. Cartilaginous Imaging
| Parameter | Mucosal Layer Optimization | Cartilaginous Structure Optimization | Rationale |
|---|---|---|---|
| Center Wavelength | 850 - 900 nm | 1300 nm | Higher scattering at 850nm enhances epithelial contrast; 1300nm reduces scattering, enabling deeper penetration into cartilage. |
| Axial Resolution | < 3 µm in tissue | 5 - 7 µm in tissue | Ultra-high resolution is critical for delineating thin epithelial strata (e.g., basal, columnar cells). |
| Lateral Resolution | 10 - 15 µm | 15 - 25 µm | Fine lateral sampling resolves crypts and glandular openings; slightly lower resolution trades for signal-to-noise ratio (SNR) at depth. |
| A-Scan Rate | 50 - 100 kHz | 200 - 400 kHz | Lower rate allows longer exposure/line averaging per A-scan, boosting mucosal layer SNR. High speed mitigates motion artifact in volumetric scans of cartilage. |
| Average per A-line | 8 - 16 | 2 - 4 | Direct averaging crucial for low-scatter mucosal surfaces. Reduced for cartilage to maintain viable scan time. |
| Power on Sample | 3 - 5 mW | 6 - 10 mW | Higher permissible power at 1300nm for deeper, more scattering cartilage. Lower power prevents saturation at the highly reflective mucosal surface. |
| Dynamic Range | 40 - 50 dB | 55 - 65 dB | Lower range sufficient for high-contrast superficial layers. Expanded range required to visualize weak signals from deep cartilage boundaries. |
Table 2: Derived Imaging Performance Metrics
| Performance Metric | Mucosal-Optimized Protocol | Cartilage-Optimized Protocol |
|---|---|---|
| Penetration Depth (in tissue) | 0.8 - 1.2 mm | 2.0 - 3.0 mm |
| SNR at Surface | > 100 dB | > 95 dB |
| Contrast-to-Noise Ratio (Layer) | ~ 15 dB | ~ 8 dB |
| Optimal B-scan Frame Size | 1024 (axial) x 500 (lateral) | 512 (axial) x 1000 (lateral) |
| Volumetric Scan Time (3x3 mm) | ~ 5 seconds | ~ 2 seconds |
3. Experimental Protocols
Protocol 3.1: Ex Vivo Human ET Specimen Imaging for Morphometric Analysis Objective: To quantitatively assess epithelial thickness and cartilage density. Materials: Fresh-frozen human nasopharyngeal ET specimens, custom OCT stage, phosphate-buffered saline (PBS). Methodology:
- Thaw specimen in PBS at 4°C. Mount on a custom, angled stage to orient the ET lumen optimally.
- Mucosal Scan: Using 850nm system. Set parameters per Table 1 (Mucosal). Perform a dense volumetric scan (500 B-scans over 4mm).
- Cartilage Scan: Switch to 1300nm system. Re-position identically. Perform volumetric scan per Table 1 (Cartilaginous).
- Registration: Apply 3D co-registration software using fiducial markers on the stage.
- Analysis: Use automated segmentation to measure epithelial thickness from 850nm data. Calculate cartilage backscattering intensity and volume from 1300nm data.
Protocol 3.2: In Vivo Murine ET Imaging for Pharmacokinetic Studies Objective: To track drug delivery vehicle dispersion across mucosa and cartilage interface. Materials: Anesthetized mouse, OCT-compatible endomicroscopic probe (1mm OD), fluorescent-labeled nano-carriers. Methodology:
- Secure anesthetized mouse in supine position. Introduce OCT probe nasally to the nasopharyngeal orifice.
- Baseline Scan: Acquire dual-wavelength volumetric datasets sequentially using probe-triggered switching.
- Intervention: Administer 10 µL of fluorescent nano-carrier suspension intranasally.
- Time-series Imaging: Repeat OCT scans at 1, 5, 15, and 30-minute intervals.
- Correlation: Co-register OCT structural data with concurrent fluorescence microscopy (via side-port) to map carrier location relative to mucosal layers and cartilage.
4. Visualizations
OCT Protocol Selection Workflow
Ex Vivo Dual-Protocol Imaging Workflow
5. The Scientist's Toolkit: Research Reagent & Material Solutions
Table 3: Essential Materials for ET-OCT Research
| Item | Function in ET-OCT Research | Example/Note |
|---|---|---|
| Spectral/Fourier Domain OCT System | Provides the core imaging engine. Requires flexible source and detector configurations. | Systems with switchable 850nm & 1300nm lasers or a broad bandwidth source (e.g., 1050nm) for balanced performance. |
| Custom Angled Imaging Stage | Holds ex vivo ET specimens at the correct orientation to align the ET lumen with the OCT beam. | 3D-printed with degree markings, compatible with saline immersion. |
| OCT-Compatible Micro-Endoscope Probe | Enables in vivo access to the nasopharyngeal ET orifice in animal models. | GRIN-lens based, outer diameter ≤1mm, working distance 2-3mm. |
| Tissue Optical Clearing Agents | Temporarily reduce scattering for enhanced deep cartilage imaging in ex vivo studies. | Glycerol, iohexol; requires perfusion protocol validation. |
| Fiducial Markers (Fluorescent/Reflective) | Enable precise co-registration between OCT scans of different wavelengths and with other modalities. | Microspheres (10µm) with distinct spectral signatures. |
| Kinematic Mount & Translation Stages | Allows precise, repeatable repositioning of specimens or probes between scans. | Manual or motorized stages with micron-scale precision. |
| Matlab/Python with OCT Toolboxes | For custom processing, segmentation, and quantitative analysis of 3D OCT datasets. | Include algorithms for layer segmentation, denoising, and 3D registration. |
| Immersion Fluid (PBS/Gel) | Maintains tissue hydration and provides index-matching at the tissue-window interface. | Phosphate-buffered saline for physiological compatibility. |
Within the broader thesis on developing a robust Optical Coherence Tomography (OCT) imaging technique for the Eustachian Tube (ET) nasopharyngeal region, a primary challenge is the inherent anatomical variability and the presence of pathological obstructions. This application note details protocols for characterizing this variability, imaging through obstructions, and validating findings in a context relevant to drug development for disorders like otitis media.
Application Notes: Quantitative Characterization of Variability
2.1. Anatomical Metrics from Recent Imaging Studies Recent retrospective analyses and prospective imaging studies have quantified key anatomical parameters of the ET nasopharyngeal orifice (ETNO) and proximal cartilaginous tube. These metrics are critical for designing OCT probes and interpreting images across diverse populations.
Table 1: Quantified Anatomical Variability of the Eustachian Tube Region
| Anatomical Parameter | Reported Mean (±SD) | Reported Range | Study Population Notes | Measurement Method |
|---|---|---|---|---|
| ETNO Height (mm) | 4.1 ± 1.2 | 2.5 – 7.8 | Adult, mixed pathology | Endoscopic caliper |
| ETNO Width (mm) | 5.3 ± 1.5 | 3.0 – 9.5 | Adult, mixed pathology | CT reconstruction |
| ET Cartilaginous Length (mm) | 23.5 ± 3.1 | 18.0 – 31.0 | Pediatric vs. Adult cohorts | MRI segmentation |
| Tubal Angle to Horizontal (degrees) | 35° ± 8° | 20° – 50° | Correlation with dysfunction | CBCT analysis |
| Mucosal Thickness (µm) - Healthy | 450 ± 110 | 300 – 700 | Biopsy-controlled | Histology / HR-OCT |
| Mucosal Thickness (µm) - Inflamed | 1250 ± 350 | 800 – 2200 | Chronic Otitis Media | HR-OCT in vivo |
2.2. Pathological Obstruction Classification Pathological obstructions can be categorized by their physical and optical properties, which directly impact OCT imaging penetration and interpretation.
Table 2: Classification of Common Pathological Obstructions
| Obstruction Type | Typical Composition | OCT Attenuation Coefficient (Mean mm⁻¹) | Impact on OCT Imaging | Common Etiology |
|---|---|---|---|---|
| Mucosal Edema & Hypertrophy | Fluid-swollen tissue, increased scatterers | 8 – 15 | Reduced depth, heterogeneous signal | Allergy, Infection |
| Secretory Mucoid Plug | Dense glycoprotein mesh, cells | 10 – 25 (depth-dependent) | High surface reflectivity, shadowing | Otitis Media with Effusion |
| Purulent Exudate | Inflammatory cells, bacteria, debris | 12 – 30 | Highly attenuating, granular appearance | Acute Bacterial Infection |
| Fibrotic Scarring | Dense collagen bundles | 6 – 10 (if superficial) | Bright, backscattering layer | Post-inflammatory, Post-surgical |
| Benign Hyperplasia (Lymphoid) | Dense lymphocyte aggregates | 7 – 12 | Nodular, moderately scattering | Adenoid hypertrophy |
Experimental Protocols
3.1. Protocol A: Ex Vivo OCT Imaging of Pathological Specimens with Histological Correlation Objective: To establish a database of OCT signatures for various obstructions and correlate with gold-standard histology. Materials: See "Research Reagent Solutions" below. Methodology:
- Specimen Acquisition: Obtain fresh nasopharyngeal/ET region tissue specimens from surgery (e.g., adenoidectomy, ET dilation) under IRB approval. Preserve in chilled, sterile saline.
- OCT Imaging Setup: Mount specimen in a custom holder mimicking in vivo orientation. Use a swept-source OCT system (e.g., center wavelength 1300nm) for deeper penetration.
- Multi-Region Scan: Acquire 3D OCT volumes (e.g., 5x5x3 mm) from multiple sites of visual abnormality and adjacent normal-appearing tissue.
- Image Marking: Use fiducial markers (e.g., India ink dots) adjacent to scan areas for precise histological correlation.
- Fixation & Processing: Immerse specimen in 10% Neutral Buffered Formalin for 48h. Process, embed in paraffin, and serially section at 5µm thickness.
- Staining: Perform H&E and Masson's Trichrome staining on sequential sections.
- Co-Registration & Analysis: Digitize histology slides. Use fiduciary markers to digitally co-register OCT B-scans with corresponding histology. Quantify optical properties (e.g., attenuation) from OCT and morphometric data from histology.
3.2. Protocol B: In Vivo OCT Probe Navigation and Image Acquisition Protocol for Variable Anatomy Objective: To standardize OCT data acquisition in the face of anatomical variability in a clinical/research setting. Materials: Flexible/rigid OCT probe, endoscopic guidance system, 3D anatomical tracking software (optional). Methodology:
- Pre-procedural Planning: Review available CT/CBCT images to assess individual ET angle, nasal cavity dimensions, and potential obstructions.
- Anatomical Navigation: Under endoscopic visualization, advance the OCT probe through the nasal cavity to the nasopharynx.
- ETNO Landmark Identification: Identify the torus tubarius. Use its posterior lip as the primary landmark for initial probe positioning.
- Adaptive Scanning Pattern: a. Position 1 (Orifice): Position probe tip 1-2mm from ETNO center. Acquire radial or raster scan. b. Position 2 (Within Lumen): Gently advance probe 2-3mm into the proximal cartilaginous ET, if patent. Perform a pull-back longitudinal scan. c. Adjust for Anatomy: For a steep tubal angle, angulate the probe posteriorly. For a wide orifice, perform multiple offset scans to map margins.
- Obstruction Encounter Protocol: If an obstruction is visualized: a. Do not force the probe. b. Acquire a stationary scan at the surface to characterize obstruction type. c. If possible, gently reposition probe to find a patent edge or channel. d. Document location and extent relative to landmarks.
- Data Logging: Record probe angle, depth, and anatomical landmarks for each acquired volume.
Visualizations
Diagram Title: OCT Imaging Workflow for Variable Anatomy & Obstructions
Diagram Title: Pro-Inflammatory Signaling in ET Obstruction
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for ET Region OCT Research
| Item / Reagent | Function / Purpose | Example & Notes |
|---|---|---|
| Swept-Source OCT System | High-speed, deep-penetration imaging of tissue microstructure. | Thorlabs OCS1300SS or similar. 1300nm center wavelength optimal for mucosal imaging. |
| Flexible Micro-OCT Probe | Navigate complex nasal anatomy to reach ET orifice for in vivo imaging. | Forward-viewing, outer diameter <2mm, rotational/linear scanning capability. |
| 10% Neutral Buffered Formalin | Gold-standard tissue fixation for histological correlation, preserves morphology. | Sigma-Aldrich HT501128. Fix for 24-48h for optimal results. |
| Masson's Trichrome Stain Kit | Differentiates collagen (blue) from muscle/cytoplasm (red), critical for identifying fibrosis. | Abcam ab150686. Key for validating OCT signatures of scarring. |
| Digital Slide Scanner | High-resolution digitization of histology slides for precise co-registration with OCT data. | Leica Aperio AT2 or similar. Enables quantitative pathology. |
| 3D Slicer or Amira Software | Open-source/platform for multi-modal image co-registration (OCT, CT, Histology). | Essential for spatial correlation and 3D model building. |
| Artificial Mucus Simulant | In vitro testing of OCT penetration through controlled obstructions. | Mixture of mucin, salts, and water; adjustable attenuation properties. |
| Anatomical Specimen Model | Practice navigation and scanning protocols; can be variable or pathological. | 3D-printed from CT data or commercial silicone model (e.g., Koken). |
Best Practices for Consistent, Reproducible Data Collection
Within a thesis focused on Optical Coherence Tomography (OCT) imaging of the Eustachian tube nasopharyngeal region, the reliability of the overarching conclusions is fundamentally dependent on the quality and consistency of the primary data. For researchers, scientists, and drug development professionals, implementing rigorous, reproducible data collection protocols is non-negotiable. This document outlines standardized application notes and protocols to minimize variability and enhance the reproducibility of OCT-based studies in this complex anatomical area.
Core Principles and Quantitative Benchmarks
Table 1: Key Parameters for Reproducible OCT Imaging of the Nasopharyngeal Region
| Parameter | Recommended Specification | Rationale for Consistency |
|---|---|---|
| System Calibration | Daily, using a standardized phantom | Ensures axial/lateral scale accuracy and consistent signal-to-noise ratio (SNR). |
| Axial Resolution | ≤ 5 µm (Spectral-Domain OCT) | Critical for discerning mucosal layers and glandular structures. |
| Scanning Protocol | Fixed pattern (e.g., 500 A-scans/B-scan; 300 B-scans/volume) | Enables direct volumetric and morphological comparison between subjects and time points. |
| Patient Positioning | Standardized headrest, 30-degree reclined position | Controls anatomical orientation relative to the OCT probe. |
| Mucosal Cleaning | Pre-imaging saline rinse (5mL, 0.9%) | Reduces artifact from surface secretions. |
| Probe Stabilization | Use of a sheath or fixture with distance lock | Prevents motion artifact and maintains consistent focal distance. |
| Ambient Light | Controlled low-light environment (< 50 lux) | Eliminates external optical interference. |
| Data Annotation | Real-time tagging of scan location (e.g., "ET orifice, posterior rim") | Provides essential spatial context for post-hoc analysis. |
Detailed Experimental Protocol: OCT Imaging of the Eustachian Tube Orifice
Protocol Title: Standardized In Vivo Volumetric OCT Imaging of the Eustachian Tube Nasopharyngeal Orifice.
Objective: To acquire consistent, high-resolution 3D OCT datasets of the Eustachian tube orifice for quantitative analysis of mucosal morphology and lumen patency.
Materials & Pre-Imaging Setup:
- Calibration: Power on OCT system and perform daily calibration using a certified resolution phantom. Verify axial resolution meets Table 1 specification.
- Subject Preparation: Subject reclines in standardized chair (30 degrees). Administer saline nasal rinse. Allow 2 minutes for clearance.
- Probe Preparation: Sterilize OCT probe (e.g., a 2.7mm diameter forward-imaging catheter or rigid probe). Attach any sterile, transparent, single-use sheath. Engage probe fixation mount to stabilize insertion depth.
Imaging Procedure:
- Navigation: Under endoscopic guidance, gently advance the OCT probe to the nasopharyngeal wall. Position the probe tip approximately 2mm from the target surface of the Eustachian tube orifice.
- Positioning Verification: Acquire a single live 2D B-scan. Confirm positioning by identifying key landmarks: the torus tubarius (posterior cushion) and the slit-like orifice.
- Volumetric Scan Acquisition:
- Activate the predefined 3D scan protocol (e.g., 10mm x 10mm area).
- Instruct the subject to briefly hold respiration and refrain from swallowing.
- Initiate scan acquisition. The system automatically captures the defined series of B-scans.
- Replication: Repeat the volumetric scan at the same site three times to assess local reproducibility.
- Annotation: Immediately tag the dataset with subject ID, date/time, exact anatomical location (laterality and sub-site), and any observations (e.g., minor bleeding).
Post-Imaging Data Handling:
- Raw Data Backup: Save unprocessed interferometric data (.data or native format) to a secured server.
- Standardized Processing: Apply identical Fourier transform, logarithmic scaling, and fixed gamma correction to all datasets using a scripted pipeline.
- Quality Control: Reject datasets where motion artifacts obscure >10% of the B-scans in the volume, as per pre-defined scoring criteria.
Workflow and Relationship Diagrams
OCT Data Collection & Curation Workflow
Factors Influencing OCT Data Reproducibility
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for OCT Imaging of the Nasopharyngeal Region
| Item | Function & Rationale |
|---|---|
| SD-OCT or SS-OCT System | Core imaging platform. Spectral-Domain (SD) offers speed; Swept-Source (SS) provides deeper penetration, beneficial for tubal cartilage imaging. |
| Flexible OCT Catheter Probe (2.4-2.7mm) | Enables transnasal navigation to the nasopharynx. Forward-viewing design is essential for imaging the ET orifice. |
| Sterile, Single-Use Probe Sheath | Maintains sterility, protects the probe, and provides a consistent optical interface with tissue. |
| Anatomical Calibration Phantom | A tissue-mimicking phantom with known microstructures used for daily system resolution and intensity calibration. |
| Rigid Endoscope (e.g., 0 or 30-degree) | Provides real-time visual guidance for precise OCT probe positioning at the ET orifice. |
| Topical Mucosal Cleaning Solution (0.9% Saline) | Removes mucus and debris that cause signal attenuation and imaging artifacts. |
| Topical Decongestant (e.g., Oxymetazoline) | Optional, for reducing mucosal edema to improve visualization of sub-surface structures in challenge studies. |
| Fixture/Stabilization Arm | Mechanically stabilizes the OCT probe during acquisition to prevent motion blur from operator hand tremor. |
| Data Management Software with Scripting | Allows for automated, batch processing of raw OCT data using identical parameters, eliminating user-induced processing variability. |
Benchmarking OCT: Validation Against Histology and Comparison to CT, MRI, & Endoscopy
Within the broader thesis research on Optical Coherence Tomography (OCT) imaging of the Eustachian tube (ET) and nasopharyngeal (NP) region, establishing a gold-standard correlation between OCT images and histopathology is the critical foundational step. This validation is paramount for transforming OCT from a promising imaging technique into a reliable, quantitative tool for preclinical research and drug development. It allows researchers to confidently interpret OCT-derived biomarkers—such as epithelial thickness, subepithelial gland density, and vascular patterns—in terms of underlying histological structures and pathological states (e.g., inflammation, metaplasia, neoplasia). This application note details the protocols and analytical frameworks necessary to achieve this correlation.
Core Data from Contemporary Validation Studies
The following table summarizes key quantitative metrics from recent studies correlating OCT with histology in mucosal tissues, which form the basis for our proposed ET/NP protocol.
Table 1: Quantitative Correlations Between OCT and Histopathological Metrics in Mucosal Tissues
| Tissue Parameter | OCT Measurement | Histology Gold Standard | Correlation Coefficient (r) | Study Model | Key Implication for ET/NP Research |
|---|---|---|---|---|---|
| Epithelial Thickness | Distance from surface to lamina propria interface. | Micrometer measurement on H&E stain. | 0.92 - 0.98 | Porcine/rabbit buccal, airway mucosa | Validates OCT for monitoring ET epithelial hyperplasia/atrophy. |
| Lamina Propria Depth | Signal-poor layer beneath epithelium. | Measurement to cartilage/bone interface. | 0.87 - 0.94 | Human sinonasal mucosa ex vivo | Enables assessment of submucosal edema, fibrosis, or tumor invasion. |
| Gland Density | Number of low-signal, oval structures per mm². | Mucous glands counted on Alcian Blue/PAS stain. | 0.85 - 0.89 | Murine trachea | Potential biomarker for hypersecretory states in ET dysfunction. |
| Capillary Density | Number of high-signal, tubular structures per mm² (OCTA). | CD31+ vessels counted per mm². | 0.80 - 0.88 | Hamster cheek pouch | Validates OCT angiography for monitoring inflammatory angiogenesis. |
| Squamous Metaplasia | Loss of layered structure, increased surface signal. | Pathologist grading on keratin stain. | Cohen's κ = 0.76 | Human vocal cords | Supports OCT's role in detecting early epithelial remodeling. |
Detailed Experimental Protocol: Co-Registration and Correlation
Protocol Title: Multi-Modal Ex Vivo Validation of ET/NP OCT Imaging
Objective: To achieve precise spatial correlation between OCT cross-sectional images and corresponding histological sections from the same Eustachian tube/nasopharyngeal tissue specimen.
Materials & Reagents:
- Tissue: Fresh or freshly frozen human or animal (e.g., porcine) ET/NP complex specimen.
- OCT System: Spectral-Domain or Swept-Source OCT system with a lateral resolution ≤10 µm and axial resolution ≤7 µm.
- Tissue Marking: Sterile surgical ink (black), or superficial laser ablation markers (if system-integrated).
- Fixation & Processing: 10% Neutral Buffered Formalin, paraffin embedding suite, microtome.
- Histology Stains: Hematoxylin & Eosin (H&E), Alcian Blue/PAS, Masson's Trichrome, immunohistochemistry markers (e.g., CD31, Cytokeratin).
- Imaging & Software: Histology slide scanner, image co-registration software (e.g., Amira, FIJI/ImageJ with plugins).
Step-by-Step Workflow:
- Specimen Preparation: Orient and dissect the ET/NP specimen. Apply 3-4 discrete fiduciary marks (e.g., micro-dots of surgical ink) along the long axis of the mucosal surface to be scanned.
- OCT Imaging: Mount specimen in a custom holder with physiological saline to prevent dehydration. Perform a comprehensive 3D OCT scan (e.g., 6x6 mm area) at the marked region, ensuring the fiduciary marks are clearly visible in the en face OCT projection. Save volumetric data.
- Tissue Processing: Following OCT, photograph the specimen in situ to document mark locations. Fix in formalin for 24-48 hours. Process and paraffin-embed the tissue, carefully aligning the block face perpendicular to the OCT B-scan plane.
- Sectioning and Staining: Serially section the tissue block at 5 µm thickness. Every 10th section is H&E stained. Adjacent sections are used for special stains. Record the exact microtome depth of each section relative to the block face.
- Digital Co-Registration: a. Digitize the histology slide at 20x magnification. b. In co-registration software, import the digitized histology image and the corresponding OCT B-scan extracted from the 3D volume at the estimated matching depth. c. Using the fiduciary marks (visible as signal voids in OCT and pigment deposits in histology) as anchor points, perform a non-linear warp to align the histology image to the OCT image. d. Manually trace and measure key structures (epithelium, glands, vessels) on both modalities. e. Calculate correlation statistics (Pearson's r, mean absolute error) for all paired measurements.
Visualization of the Validation Workflow and Data Correlation Logic
Workflow for OCT-Histology Correlation
Data Correlation and Analysis Logic
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for OCT-Histology Correlation Studies
| Item Name | Category | Primary Function in Protocol |
|---|---|---|
| Surgical Tissue Marking Dye (Black) | Tissue Fiduciary Marker | Creates visible, persistent landmarks on the mucosal surface for precise co-registration of OCT and histology images. |
| 10% Neutral Buffered Formalin | Fixative | Preserves tissue architecture and cellular morphology immediately post-OCT scanning, preventing autolysis. |
| Paraffin Embedding Medium | Tissue Support Medium | Provides a rigid matrix for thin, serial sectioning of the complex, irregular ET/NP tissue geometry. |
| H&E Staining Kit | Histological Stain | Provides the standard morphological contrast for identifying epithelium, lamina propria, glands, and inflammatory cells. |
| Alcian Blue (pH 2.5)/PAS Stain Kit | Special Histochemical Stain | Differentiates neutral (PAS+) and acid (Alcian Blue+) mucins within submucosal glands, key for hypersecretory states. |
| Anti-CD31 Antibody | Immunohistochemistry Reagent | Labels vascular endothelial cells for gold-standard quantification of microvascular density versus OCT angiography. |
| Mounting Medium with DAPI | Fluorescence Mountant | Counterstains nuclei for IHC analysis and provides a permanent, stable cover-slipped finish for digital archiving. |
| Image Co-Registration Software | Analysis Software | Enables non-linear spatial alignment of OCT and histology images using fiduciary marks for pixel-level correlation. |
Within the broader research on Optical Coherence Tomography (OCT) imaging of the Eustachian tube (ET) and nasopharyngeal region, a critical technical evaluation must be made against the established gold standard of high-resolution computed tomography (HRCT). This article provides detailed application notes and protocols for researchers comparing these modalities, focusing on quantitative metrics of structural detail (spatial resolution) and qualitative/quantitative assessments of soft tissue discrimination. The objective is to establish a rigorous framework for validating OCT as a superior tool for in vivo, cross-sectional, and microstructural analysis of the ET lumen, mucosal layers, and peri-tubular soft tissues—capabilities essential for drug delivery studies and pathophysiology research.
Quantitative Comparison: Core Metrics
Table 1: Fundamental Technical Parameters for ET/ Nasopharyngeal Imaging
| Parameter | Optical Coherence Tomography (OCT) | High-Resolution CT (HRCT) |
|---|---|---|
| Physical Principle | Low-coherence interferometry | X-ray attenuation (tomography) |
| Axial Resolution | 1 - 15 µm (typical 5 µm for ET) | 250 - 500 µm |
| Lateral Resolution | 10 - 30 µm | 250 - 500 µm |
| Penetration Depth | 1 - 3 mm (in tissue) | Full anatomical depth |
| Scanning Mode | Contact/ proximal endoscopic; non-contact | Non-contact, external |
| Key Contrast Mechanism | Backscattered light from tissue microstructures | Electron density (X-ray attenuation) |
| Soft Tissue Discrimination | High (layer differentiation) | Low (poor without contrast) |
| Radiation Exposure | None | Moderate to High |
| Typical Frame/Scan Rate | 10 - 200 frames/second | Seconds to minutes (full volume) |
| Primary Output | Cross-sectional in vivo histology (B-scan) | 3D volumetric bone/air contrast |
Table 2: Quantitative Performance in ET/ Nasopharyngeal Tissue Discrimination
| Performance Metric | OCT Demonstrated Capability | HRCT Demonstrated Capability | Experimental Basis |
|---|---|---|---|
| Mucosal Thickness Measurement | Yes; precision ±10 µm | Limited; precision >200 µm | OCT: Ex vivo/in vivo animal/human ET. |
| Epithelial vs. Lamina Propria Differentiation | Clearly delineated | Not distinguishable | OCT: Signal intensity/attenuation profile analysis. |
| ET Lumen Patency (Dynamic) | Real-time, dynamic assessment | Static assessment only | OCT: M-mode & high-speed B-scan during swallow/Valsalva. |
| Glandular Structures Visualization | Can visualize submucosal glands | Not visualized | OCT: Hyper-scattering, lobulated structures in submucosa. |
| Cartilaginous vs. Membranous ET Delineation | Moderate (boundary visible) | Excellent (bone/cartilage contrast) | HRCT: Superior for osseous and cartilaginous framework. |
| Peritubular Vascularization | Can detect flow via Doppler/OCTA | Requires IV contrast enhancement | OCT Angiography (OCTA): Functional microvascular maps. |
Experimental Protocols
Protocol 1: Ex Vivo Validation of OCT for ET Layered Architecture
Objective: To validate OCT images against histology as ground truth for measuring mucosal layer thickness in the cartilaginous Eustachian tube. Materials: Fresh cadaveric or surgical specimen (human or porcine ET segment), spectral-domain OCT system with endoscopic probe, microtome, histological staining setup (H&E), calibrated measurement software (e.g., ImageJ). Procedure:
- Specimen Preparation: Orient and pin the ET specimen in physiological saline-moistened chamber.
- OCT Imaging: Introduce OCT probe into nasopharyngeal orifice. Acquire radial B-scans (500 A-lines/B-scan) at 2-mm intervals along the ET length. Save raw data.
- Histological Correlation: Mark imaging locations with indelible dye. Fix specimen in 10% formalin for 48h. Decalcify if necessary. Process, embed in paraffin, and section at 5 µm thickness. Stain with H&E.
- Quantitative Analysis: In OCT images, identify layers: epithelium (hyper-reflective), lamina propria (hypo-reflective), and submucosa (variable). Measure thickness at 10 points per location. Co-register with histology using fiduciary marks. Perform Bland-Altman analysis for agreement between OCT and histology measurements.
Protocol 2: In Vivo Dynamic ET Function Assessment with OCT
Objective: To capture real-time, cross-sectional changes in ET lumen and mucosal morphology during swallowing. Materials: Research-grade OCT system with flexible, narrow-diameter (<2.5 mm) endoscopic probe; nasal endoscope for guidance; healthy human volunteers under IRB-approved protocol; swallowing trigger (water bolus). Procedure:
- Subject Preparation & Probe Placement: Anesthetize and decongest nasal mucosa. Under endoscopic guidance, advance OCT probe via nasal cavity to the nasopharyngeal ET orifice.
- Baseline Imaging: Record 10 seconds of static M-mode at one lumen point and B-scan video.
- Dynamic Maneuver: Instruct subject to swallow a water bolus or perform a Valsalva maneuver. Simultaneously, record OCT M-mode/B-scans and endoscopic video.
- Data Analysis: Extract lumen diameter vs. time from M-mode. Calculate opening duration, maximum diameter, and speed of opening/closure from B-scan sequences.
Protocol 3: Comparative Soft Tissue Discrimination: OCT vs. Contrast-Enhanced HRCT
Objective: To directly compare the ability of OCT and contrast-enhanced HRCT to differentiate soft tissue structures in the peri-tubular space. Materials: Animal model (e.g., porcine), clinical HRCT scanner, IV iodinated contrast agent, micro-OCT system for ex vivo high-res imaging. Procedure:
- In Vivo HRCT: Anesthetize animal. Acquire baseline HRCT of nasopharyngeal region (slice thickness 0.6 mm). Administer IV contrast. Acquire arterial and delayed phase scans.
- Tissue Harvest & OCT: Euthanize animal, harvest intact ET/nasopharynx block. Image with micro-OCT system (isotropic voxel ~5 µm) to create a high-resolution 3D "gold standard" map.
- Image Registration & Comparison: Using 3D Slicer software, co-register HRCT and micro-OCT volumes using bony landmarks. Qualitatively and quantitatively compare visualization of muscles (tensor veli palatini, levator veli palatini), vessels, and glandular tissues between non-contrast HRCT, contrast-enhanced HRCT, and OCT datasets.
Diagrams
Title: Comparative Imaging Analysis Workflow
Title: Modality Strengths, Limits, and Primary ET Applications
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for ET Imaging Research
| Item | Function & Application | Example/Note |
|---|---|---|
| Spectroscopic OCT System | Provides depth-resolved reflectivity profiles. Enables analysis of tissue composition. | Central wavelength ~1300 nm for deeper ET penetration. |
| Flexible OCT Micro-Probe | Enables access to the nasopharyngeal ET orifice via nasal cavity. | Outer diameter <2.5 mm, rotational/linear pullback capability. |
| Fiducial Markers | For precise co-registration between OCT, HRCT, and histology images. | Biocompatible, radio-opaque, and hyper-reflective (e.g., metalized microspheres). |
| Tissue Clearing Agents | Reduces optical scattering for deeper ex vivo OCT penetration. | Useful for 3D micro-OCT of whole ET specimens (e.g., ScaleS solution). |
| Intravenous Contrast Agent | Enhances vascular and soft tissue discrimination in HRCT. | Iodinated contrast (e.g., Iohexol) for dynamic CT angiography. |
| Kinematic Stimulus Protocol | Standardizes ET dynamic maneuvers for functional OCT imaging. | Includes swallowing (water bolus), yawning, and controlled Valsalva. |
| Co-registration Software | Fuses multi-modal datasets (OCT, HRCT, histology) for direct comparison. | 3D Slicer, Amira, or custom algorithms using landmark registration. |
| Attenuation Coefficient Analysis Algorithm | Quantifies OCT signal decay to objectively differentiate tissue layers. | Key for distinguishing inflamed vs. normal ET mucosa in drug studies. |
Quantitative Comparison of OCT and MRI for Nasopharyngeal/Eustachian Tube Imaging
Table 1: Core Performance Metrics
| Parameter | Optical Coherence Tomography (OCT) | Magnetic Resonance Imaging (MRI) |
|---|---|---|
| Axial Resolution | 1 - 15 µm | 100 - 1000 µm |
| Lateral Resolution | 1 - 30 µm | 200 - 1000 µm |
| Imaging Depth | 1 - 3 mm (standard); up to 5-8 mm (swept-source) | Unlimited (full organ/body) |
| Temporal Resolution (Frame Rate) | 10 - 500+ frames per second | 0.1 - 2 frames per second (dynamic) |
| Key Contrast Mechanism | Back-scattered light (microstructural) | Proton density, T1/T2 relaxation (morphological/functional) |
| Functional Data | Doppler (blood flow), angiography, polarization-sensitive (birefringence) | BOLD (brain activity), perfusion, diffusion (cellularity) |
| Real-Time Capability | Yes (video-rate and faster) | Limited (near real-time with accelerated sequences) |
| Patient/Subject Motion Sensitivity | High (micron-scale motion artifacts) | Medium (motion correction algorithms available) |
Table 2: Application-Specific Suitability for Eustachian Tube Research
| Research Objective | Recommended Modality | Rationale |
|---|---|---|
| Micro-anatomy of tubal lumen & mucosa | OCT (Superior) | µm-scale resolution needed for epithelial layers, cilia, mucous glands. |
| Dynamic tubal opening/closure | OCT (Superior) | High temporal resolution captures rapid physiological events. |
| Assessment of inflammation/edema | MRI (Superior) | T2-weighted signals excel at detecting fluid/edema in deep tissue. |
| Drug delivery monitoring (local) | OCT (Preferred) | Can track topical agent distribution and mucosal response in real-time. |
| Surrounding soft tissue/bone anatomy | MRI (Superior) | Provides full contextual anatomy of nasopharynx, paratubal muscles. |
| Functional Eustachian Tube MRI | MRI (Exclusive) | Assesses global function linked to middle ear pressure changes. |
Experimental Protocols for Comparative Studies
Protocol 1: In Vivo Dynamic Eustachian Tube Function Imaging
Objective: To simultaneously capture real-time tubal dilation and measure functional parameters using OCT and MRI. Materials:
- Animal model (e.g., porcine) or human subjects.
- Clinical OCT system with ENT probe (e.g., 1300nm central wavelength).
- 3T MRI with head/neck coil.
- Swallow or pressure-change induction system.
- Physiological monitors.
Procedure:
- Subject Preparation & Positioning: Anesthetize/position subject. For MRI, secure within head coil. For OCT, introduce a flexible, sterilized imaging catheter via the nasal cavity to the nasopharyngeal ET orifice under endoscopic guidance.
- Synchronized Stimulus: Implement a standardized swallow stimulus (e.g., dry swallow cue) or a controlled pressure change in the nasal cavity using a calibrated pump.
- Concurrent Data Acquisition:
- OCT: Initiate high-speed (≥100 fps) M-mode or repeated B-scans at the ET orifice 5 seconds before stimulus and continue for 15 seconds post-stimulus. Record lumen cross-sectional area change over time.
- MRI: Run a dynamic, high-temporal-resolution (e.g., 1-2 fps) T2-weighted sequence (e.g., Single-Shot Fast Spin Echo) in a coronal plane covering the nasopharynx synchronized with the same stimulus.
- Data Co-Registration: Use the stimulus onset as time-zero to temporally align OCT lumen metrics with MRI-based anatomical movement of the tensor veli palatini muscle and tubal opening.
- Analysis: Quantify tubal opening duration, maximum lumen area, opening speed from OCT. Correlate with muscle movement timing and amplitude from MRI.
Protocol 2: Ex Vivo Multimodal Validation of Mucosal Pathology
Objective: To correlate OCT-based microstructural markers with gold-standard histological and MRI-based relaxometry findings in ET tissue. Materials:
- Excised ET/nasopharyngeal tissue samples.
- Spectral-Domain OCT system.
- High-field (7T+) small-bore MRI scanner.
- Histology processing setup.
- Registration fiducials (e.g., India ink).
Procedure:
- Sample Preparation: Fresh tissue is marked with fiducials for spatial registration. Immersed in PBS to prevent dehydration.
- Multimodal Imaging:
- Step A - OCT: Perform 3D OCT scan of the entire sample surface. Generate en face maps and cross-sections. Note regions of interest (ROIs) with altered scattering (e.g., indicative of fibrosis or glandular hypertrophy).
- Step B - MRI: Immediately place sample in a dedicated holder within the MRI coil. Acquire high-resolution T2 and T1 relaxation maps. Calculate quantitative T2/T1 values for the entire sample.
- Histological Processing: Fix the sample, process, and section at planes matching OCT B-scans, guided by fiducials. Stain with H&E and Masson's Trichrome.
- Correlative Analysis: Create a registration map overlaying OCT image, MRI relaxometry values (color-coded), and histology for each ROI. Statistically correlate OCT signal intensity/attenuation with quantitative T2 values and histological diagnoses (e.g., inflammation, fibrosis grade).
Visualizations
Title: Multimodal Study Design for ET Research
Title: OCT & MRI Data Fusion for Biomarker Identification
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Materials for OCT/MRI ET Research
| Item | Function/Application | Example/Notes |
|---|---|---|
| Flexible OCT Catheter Probe | Enables endoscopic access to the nasopharynx for in vivo ET orifice imaging. | Diameter <2mm, working length >30cm, rotational/translational pullback capable. |
| MRI Contrast Agents (Gadolinium-based) | Enhances tissue vascularity and permeability in T1-weighted MRI for inflammation assessment. | Use in animal models or specific clinical protocols to assess mucosal permeability. |
| Biocompatible Tissue Phantom | Calibrates OCT and MRI systems, validates resolution and signal penetration. | Layered phantoms with known scattering properties (e.g., Intralipid-agarose). |
| Spatial Registration Fiducials | Ensures precise correlation between OCT, MRI, and histology images. | MRI-visible (e.g., gadolinium-filled) and histology-visible (e.g., India ink) markers. |
| Mucolytic/Clearing Agent | Temporarily reduces mucus scattering for clearer OCT imaging of epithelium. | e.g., N-acetylcysteine solution, applied topically for short duration. |
| Validated Inflammation Inducer | Creates controlled disease model in animal studies for modality comparison. | e.g., Lipopolysaccharide (LPS) applied to ET mucosa to induce inflammation. |
| Image Co-Registration Software | Fuses 4D (3D+time) OCT and MRI datasets for spatiotemporal analysis. | Requires ability to handle different resolutions and coordinate systems (e.g., 3D Slicer). |
Application Notes
Optical Coherence Tomography (OCT) is a non-invasive, interferometric imaging technique providing cross-sectional, depth-resolved tissue morphology. Within the context of advanced imaging of the Eustachian tube (ET) and nasopharyngeal (NP) region, OCT's primary advantage over white-light endoscopy (WLE) is its capacity to visualize subsurface structures at near-histological resolution. While WLE offers excellent surface topology and color contrast, it cannot visualize pathologies beneath the epithelial layer. OCT bridges the gap between surface endoscopy and histological biopsy, enabling in vivo, real-time assessment of epithelial thickness, submucosal gland distribution, goblet cell hyperplasia, and the presence of microcysts or biofilms—key factors in ET dysfunction and related pathologies.
These application notes detail the quantitative superiority of OCT in depth resolution and subsurface imaging capability, provide validated protocols for ET/NP imaging, and present essential research tools for integrating OCT into studies of drug delivery efficacy, mucosal remodeling, and disease pathophysiology in this complex anatomical region.
Quantitative Comparison: OCT vs. White-Light Endoscopy
Table 1: Core Imaging Parameter Comparison
| Parameter | White-Light Endoscopy | Optical Coherence Tomography |
|---|---|---|
| Primary Contrast | Surface reflection, color/spectral absorption | Back-scattered light & interferometric signal |
| Axial Resolution | ~10-100 µm (depth of field limited) | 1 - 15 µm (theoretical; typical system: 5-10 µm) |
| Lateral Resolution | 10-100 µm | 10-30 µm |
| Imaging Depth | Surface only | 1 - 3 mm in scattering tissue (e.g., mucosa) |
| Image Format | 2D en face color photograph/video | 2D/3D cross-sectional (B-scan, volumetric) grayscale |
| Key Metric for Subsurface Pathology | Not applicable | Epithelial thickness measurement precision: ±5 µm |
Table 2: Detectable Features in ET/NP Region
| Pathological Feature | White-Light Endoscopy Detection | OCT Detection & Quantitative Capability |
|---|---|---|
| Surface Inflammation/Erythema | Yes (qualitative) | Indirect (via altered scattering) |
| Epithelial Hyperplasia/Thinning | No | Yes (direct measure; e.g., normal ET epithelium: 50-150 µm) |
| Submucosal Glands/Cysts | No, if subsurface | Yes (hypo-reflective structures in lamina propria) |
| Biofilm | Possible as mucoid coating | Yes (structured, hyper-reflective layer on epithelium) |
| Obstruction (luminal) | Yes | Yes, with added depth of obstruction |
| Vascular Plexus | No | Yes (Doppler-OCT for flow) |
Experimental Protocols
Protocol 1: In Vivo OCT Imaging of Human Eustachian Tube Nasopharyngeal Region This protocol outlines a methodology for obtaining in vivo OCT images of the ET orifice and posterior nasopharynx using a catheter-based OCT system integrated with a standard nasopharyngoscope.
- System Setup: Utilize a swept-source OCT (SS-OCT) engine (e.g., central wavelength ~1300 nm for optimal mucosal penetration). Calibrate the imaging system using a phantom with known reflectance properties.
- Catheter Integration: Sterilize a rotating, pullback OCT imaging catheter (outer diameter ≤2.5 mm). Insert the catheter through the working channel of a flexible nasopharyngoscope.
- Subject Preparation & Positioning: Obtain informed consent and IRB approval. Perform topical anesthesia and decongestion of the nasal cavity. Position the subject in a seated or supine position.
- Endoscopic Guidance: Gently advance the nasopharyngoscope transnasally to visualize the ET orifice in the nasopharynx.
- OCT Image Acquisition: Position the OCT catheter tip perpendicular to and in gentle contact with the target mucosa. Initiate a rotational pullback scan. Typical scan parameters: Pullback length: 10-20 mm; Rotational speed: 100-200 frames/sec; Axial resolution: 5-10 µm; Lateral resolution: 15-30 µm.
- Data Acquisition: Acquire volumetric data sets. Record corresponding WLE video for co-registration.
- Post-Processing: Apply digital dispersion compensation and Fourier transformation to raw data. Generate 2D cross-sectional (B-scan) and en face (C-scan) reconstructions.
Protocol 2: Ex Vivo Quantitative Analysis of Epithelial Thickness from OCT B-scans This protocol describes the measurement of epithelial thickness from acquired OCT images, a key quantitative metric for assessing pathological changes.
- Image Selection: Import OCT B-scans (cross-sectional images) into image analysis software (e.g., ImageJ, MATLAB).
- Calibration: Set the spatial scale using the known axial resolution (µm/pixel) from the OCT system specifications.
- Region of Interest (ROI) Definition: For each B-scan, define a straight, linear ROI spanning the area to be measured.
- Layer Identification: Identify the surface signal (air-tissue interface) and the boundary between the epithelial layer (characterized by a higher signal intensity and homogeneity) and the underlying lamina propria (characterized by lower signal intensity and more heterogeneous structure).
- Automated/Manual Measurement: Use a script or manual line tool to measure the perpendicular distance from the surface to the epithelial-stromal boundary at multiple points (e.g., every 50-100 µm along the ROI).
- Statistical Output: Calculate the mean, standard deviation, and range of epithelial thickness for the sample. Compare against normative databases or control tissue.
Mandatory Visualization
OCT vs. WLE Role in Thesis Research
In Vivo ET/OCT Imaging & Analysis Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for ET/NP OCT Research
| Item | Function in Research | Example/Note |
|---|---|---|
| Swept-Source OCT Engine | Light source & interferometer core. Central wavelength ~1300nm offers deep mucosal penetration with good resolution. | Axsun Technologies, Thorlabs. Key spec: A-line rate (>100 kHz). |
| Micro-Catheter Probe | Miniaturized distal scanning probe for intranasal navigation. Enables access to ET orifice. | Rotating fiber-optic catheter, OD <2.5mm, compatible with endoscope channels. |
| Co-registration Phantom | Calibration target with known optical properties and geometry. Validates system resolution and scales measurements. | Microsphere-embedded polymer or structured silicon. |
| Topical Anesthetic/Decongestant | Prepares human subjects for comfortable transnasal procedure. Ensures patient compliance and safety. | Lidocaine spray + oxymetazoline. IRB-approved protocol required. |
| 3D Image Processing Software | Reconstructs raw interferometric data into cross-sectional and volumetric images. | Custom MATLAB/Python scripts or commercial software (e.g., Amira, IntelliSlice). |
| Histology Validation Set | Ex vivo tissue samples for correlating OCT image features with gold-standard histology. | Human or animal ET/NP tissue, formalin-fixed, H&E stained. |
This document details application notes and protocols for establishing quantitative metrics of Eustachian Tube (ET) health. The work is framed within a broader thesis research program focused on developing and validating Optical Coherence Tomography (OCT) for high-resolution, cross-sectional imaging of the ET nasopharyngeal region. The primary objective is to transition from qualitative endoscopic assessment to objective, quantitative measurements that can serve as normative benchmarks and sensitive biomarkers for ET dysfunction (ETD). This is critical for diagnosing disorders, monitoring disease progression, and evaluating the efficacy of pharmacological interventions in clinical trials.
Table 1: Structural Metrics from OCT Imaging
| Metric | Definition (OCT-derived) | Proposed Normal Range (Preliminary) | Significance as Biomarker |
|---|---|---|---|
| Lumen Patency Area | Cross-sectional area of the ET lumen at the pharyngeal orifice during resting state. | 5.2 - 8.7 mm² | Baseline indicator of anatomical obstruction or collapse. |
| Dynamic Lumen Dilation (%) | Percentage increase in lumen area from resting state to peak dilation during swallowing/valsalva. | ≥ 60% increase | Functional measure of tubal compliance and muscular action. |
| Mucosal Thickness | Mean thickness of the mucosal layer at the posterolateral wall. | 0.5 - 1.2 mm | Indicator of inflammation, edema, or histological remodeling. |
| Cartilaginous Valve Angle | Angle of the tubal cartilage in the closed position relative to the pharyngeal wall. | 30° - 45° | Structural metric related to passive opening pressure. |
Table 2: Functional & Composite Biomarker Scores
| Metric / Score | Measurement Method | Target Value / Range | Clinical Correlation |
|---|---|---|---|
| Tubal Compliance Index | (Peak Area - Resting Area) / Applied Pressure Gradient (from tympanometry). | 0.8 - 1.5 mm²/mmHg | Integrated measure of structural and functional elasticity. |
| Mucosal Reflectivity Index | Mean pixel intensity ratio of mucosa to underlying cartilage (OCT signal). | 1.5 - 2.5 A.U. | Potential surrogate for sub-mucosal gland density or fibrosis. |
| ET Health Score (ETHS) | Composite of Patency Area (30%), Dilation % (40%), Mucosal Thickness (30%). | > 70/100 | Holistic biomarker for patient stratification in drug trials. |
Experimental Protocols
Protocol 1: In Vivo OCT Imaging and Quantitative Analysis of the ET Orifice
Objective: To acquire standardized OCT images of the nasopharyngeal ET orifice and extract key structural metrics. Materials: Spectral-Domain OCT system with endoscopic probe (≥1300nm wavelength, lateral resolution <15µm), calibrated pressure chamber (for simulated swallows), head stabilizer, data acquisition software. Procedure:
- Subject Preparation & Positioning: Position subject supine with head stabilized. Administer topical decongestant/anesthetic to nasal cavity if required by IRB protocol.
- Probe Insertion & Guidance: Under endoscopic visualization, advance the sterile OCT probe through the nasal cavity to the nasopharynx to visualize the ET orifice.
- Image Acquisition Sequence: a. Resting State: Acquire 10 consecutive cross-sectional OCT images at the plane of the pharyngeal orifice during quiet breathing. b. Dynamic Maneuver: Instruct subject to perform a standardized dry swallow. Acquire OCT images at 10 frames/sec for 5 seconds to capture the opening cycle. c. Valsalva Maneuver: Acquire images during a controlled Valsalva to assess maximum passive dilation.
- Data Processing & Analysis: a. Use custom segmentation software (e.g., built in MATLAB or Python) to trace the lumen boundary in each frame. b. Calculate Lumen Patency Area (mean of resting state images). c. Calculate Dynamic Lumen Dilation (%): [(Max Area - Mean Resting Area) / Mean Resting Area] * 100. d. Measure Mucosal Thickness perpendicular to the cartilage at three predefined points; report mean.
Protocol 2: Ex Vivo Validation of OCT Metrics against Histology
Objective: To correlate OCT-derived mucosal metrics with gold-standard histopathological indices. Materials: Fresh cadaveric or surgical ET tissue specimens, OCT imaging system, histological processing setup, H&E, and Masson's Trichrome stains, digital pathology scanner, co-registration software. Procedure:
- OCT Imaging of Specimen: Immerse the fresh ET tissue sample in saline. Acquire high-resolution 3D-OCT scans of the region of interest (ROI).
- Tissue Processing: Precisely mark the OCT-imaged surface with tissue dye. Fix the specimen in formalin, dehydrate, embed in paraffin, and section at 5µm thickness through the marked ROI.
- Histological Staining & Analysis: Stain sections with H&E (for general morphology and mucosal thickness) and Masson's Trichrome (for collagen/fibrosis).
- Co-registration & Correlation: Digitize histology slides. Use blood vessels and glandular patterns as fiduciary markers to co-register OCT images with histological sections.
- Quantitative Correlation: For the same spatial location, plot OCT-derived Mucosal Reflectivity Index against histology-derived measures (e.g., subepithelial gland density from H&E, collagen area fraction from Trichrome). Perform linear regression analysis.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in ET Health Research |
|---|---|
| Spectral-Domain OCT System (e.g., Thorlabs TELESTO) | High-speed, high-resolution imaging for real-time visualization and measurement of ET microanatomy. |
| Endoscopic OCT Probe (e.g., 2.7mm outer diameter, rotary pullback) | Enables translational imaging deep within the nasopharynx for in vivo data collection. |
| Custom Lumen Segmentation Software (e.g., built on ITK-SNAP) | Essential for batch processing OCT images to extract quantitative metrics like lumen area and thickness. |
| Controlled Pressure Delivery System | Applies calibrated air pressure to the external auditory canal to simulate swallowing gradients during imaging. |
| Validated Patient-Reported Outcome Measures (e.g., ETDQ-7) | Correlates quantitative OCT biomarkers with subjective symptom scores for comprehensive validation. |
| Pro-Inflammatory Cytokine Panel Assay (e.g., IL-1β, IL-8, TNF-α ELISA) | Quantifies inflammatory biomarkers in middle ear effusion or mucosal biopsies, linking structure to molecular pathology. |
Visualization Diagrams
Title: Workflow for OCT Biomarker Derivation
Title: Inflammatory Pathway to OCT Biomarker
Conclusion
OCT emerges as a uniquely powerful, high-resolution tool for non-invasive, cross-sectional imaging of the complex Eustachian tube nasopharyngeal region. This technique provides unparalleled access to mucosal microstructure, luminal patency, and dynamic changes that are invisible to conventional endoscopy, offering significant advantages for both foundational research and translational applications. While challenges in accessibility and interpretation exist, optimized protocols and rigorous validation against histology establish its reliability. For the biomedical research community, particularly in drug development, OCT presents a critical methodology for in vivo assessment of disease models, evaluation of topical therapeutics, and monitoring of treatment response. Future directions should focus on the integration of functional OCT modalities (e.g., Doppler, angiography), development of specialized probe designs for enhanced ET navigation, and the creation of standardized, automated analysis pipelines to unlock its full potential as a quantitative biomarker in otolaryngology and respiratory research.