Beyond the Surface: OCT vs. Histology for Stent Strut Coverage Assessment in Preclinical Research
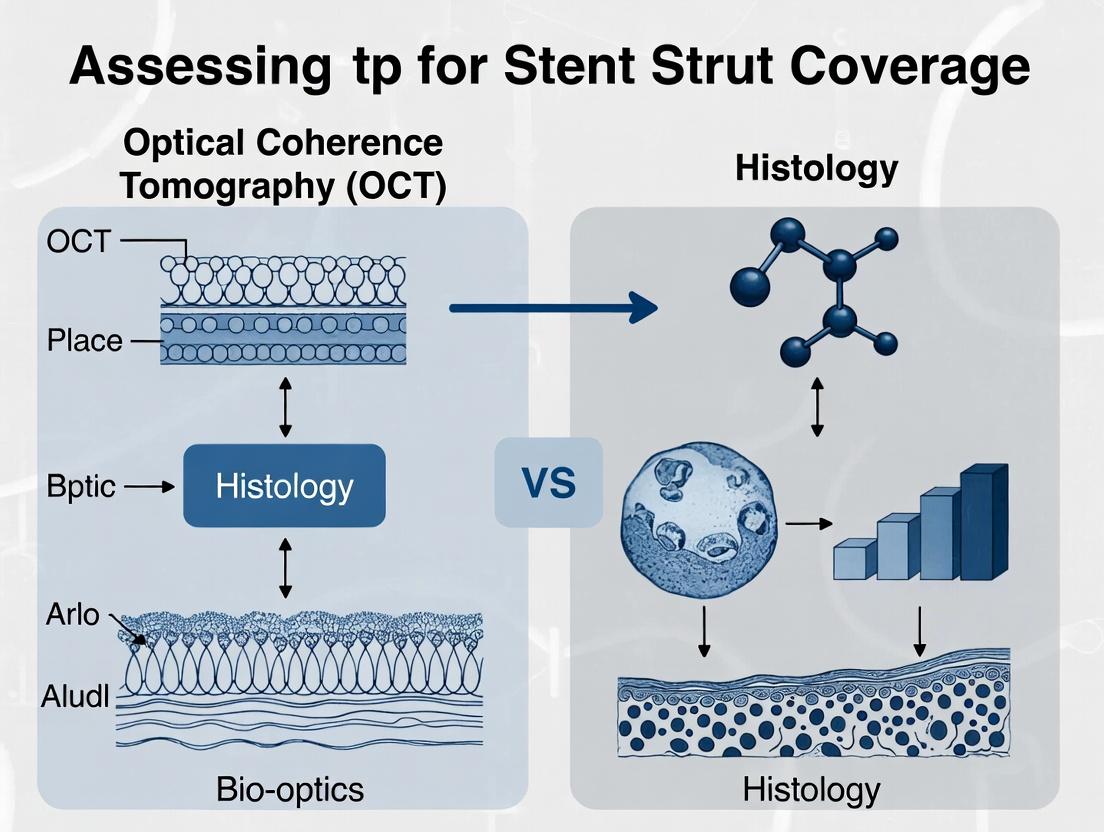
This article provides a comprehensive analysis of Optical Coherence Tomography (OCT) and histology for evaluating stent strut coverage in preclinical drug development.
Beyond the Surface: OCT vs. Histology for Stent Strut Coverage Assessment in Preclinical Research
Abstract
This article provides a comprehensive analysis of Optical Coherence Tomography (OCT) and histology for evaluating stent strut coverage in preclinical drug development. Aimed at researchers and scientists, it explores the foundational principles of each modality, details advanced methodological protocols for accurate in vivo and ex vivo analysis, addresses common pitfalls and optimization strategies, and critically validates OCT findings against the histological gold standard. The synthesis offers actionable insights for optimizing study design, improving data reliability, and accelerating the translation of novel stent technologies.
The Gold Standard and The In Vivo Surrogate: Core Principles of Histology and OCT
Thesis Context
In the critical field of coronary stent development, assessing strut coverage and neointimal characteristics is paramount for evaluating safety and healing. While Optical Coherence Tomography (OCT) offers high-resolution in vivo imaging, histology remains the definitive, unchallenged standard for validation. This guide compares the performance of histology against OCT in quantifying strut coverage and neointimal traits, underscoring histology’s role as the indispensable benchmark.
Comparative Analysis: Histology vs. OCT for Strut Assessment
Table 1: Core Performance Metrics Comparison
| Assessment Parameter | Histological Benchmark | OCT Measurement | Key Discrepancy & Implication |
|---|---|---|---|
| Spatial Resolution | ~0.2-1.0 µm (light microscopy) | ~10-20 µm (axial) | Histology detects single-cell-layer coverage (~5-10 µm); OCT may miss thin neointima, overestimating uncovered struts. |
| Tissue Characterization | Definitive. Identifies endothelial cells, macrophage infiltration, fibrin, smooth muscle cell phenotype, and inflammation. | Indirect. Based on optical signal intensity & attenuation (e.g., "bright," "layered," "heterogeneous"). | OCT cannot reliably differentiate proteoglycan-rich matrix from smooth muscle cells or fibrin, leading to misclassification of neointimal quality. |
| Strut Coverage Definition | Complete Coverage: Continuous endothelial cell layer over a mature neointima with smooth muscle cells/extracellular matrix. | Coverage: Tissue layer with signal thickness >0 µm over a strut. | Histological "complete healing" vs. OCT's "any tissue coverage." OCT classifies acellular fibrin deposits as "covered," which histology deems a thrombogenic risk. |
| Quantification of Uncovered Struts | Gold standard. Direct visual count of struts lacking endothelial lining. | High correlation in high-coverage cases (>95%), but significant overestimation in early/healing stages due to thin neointima. | Studies show OCT overestimates uncovered strut percentage by 3-8% in early time points (<3 months) vs. histology. |
| Neointimal Thickness Measurement | Highly accurate direct measurement (µm). | Good correlation for thickness >100 µm (R² >0.85). | Poor correlation for very thin neointima (<50 µm). OCT tends to overestimate thin tissue and underestimate thick, hypercellular tissue. |
Table 2: Supporting Experimental Data from Key Validation Studies
| Study (Model) | Key Experimental Finding | Protocol Summary |
|---|---|---|
| OCT-Histology Coregistration (Porcine) | For struts classified as "covered" by OCT, 22% were identified as having only fibrin deposition with no endothelialization by histology. | 1. Implant stents in coronary arteries of healthy swine. 2. Perform in vivo OCT at 28 days. 3. Euthanize, pressure-perfuse fixative. 4. Process arteries for histology (plastic embedding, sectioning, H&E, Movat's Pentachrome). 5. Use fiduciary marks (side branches) for precise OCT-frame to histology-section coregistration. 6. Blind analysis of matched pairs. |
| Assessment of Neointimal Hypo-attenuation (Human Autopsy) | OCT areas of "low-intensity" neointima showed a high correlation (89%) with proteoglycan-rich regions by histology, but also included areas with lipid-rich macrophages. | 1. Identify stent segments from autopsy cases with OCT-like imaging post-explant. 2. Process for histology (paraffin embedding, serial sectioning). 3. Stain with H&E, Masson's Trichrome (collagen), and Alcian Blue (proteoglycans). 4. Coregister imaging and histology maps. 5. Perform pixel-by-pixel classification of tissue type vs. OCT signal features. |
| Resolution Limit of Strut Coverage (Benchmarking) | Histology identified endothelial nuclei on a neointima as thin as 7 µm. OCT system with 15 µm resolution could not reliably detect tissue layers <20 µm, classifying them as "uncovered." | 1. Create in vitro phantom models with calibrated polymer layers (5-100 µm) over metallic struts. 2. Image with commercial OCT systems. 3. Process for simulated "histology" via high-magnification digital microscopy. 4. Establish minimum detectable thickness for each modality. |
Experimental Protocols in Detail
Protocol 1: Histological Processing for Stent Analysis (Gold Standard)
- Perfusion-Fixation: Upon explant, cannulate vessel and perfuse at physiological pressure with 10% neutral buffered formalin for 45-60 minutes.
- Dehydration & Embedding: Dehydrate tissue in graded ethanol series (70%-100%). Infiltrate and embed in polymethylmethacrylate (PMMA) resin to prevent strut distortion during sectioning.
- Sectioning & Grinding: Use a precision diamond saw to cut the stent segment into 2-3 mm blocks. Grind and polish blocks to obtain ~50 µm thick sections. Alternatively, use microtome for paraffin-embedded specimens (after strut removal).
- Staining: Employ multiple stains:
- Hematoxylin & Eosin (H&E): General morphology, nuclei (blue/purple), cytoplasm (pink).
- Movat's Pentachrome: Differentiates collagen (yellow), proteoglycans (blue-green), fibrin (red), and muscle (red).
- CD31/CD34 Immunohistochemistry: Specific identification of endothelial cell lining.
- Digital Analysis: Use high-resolution slide scanners. Manually or semi-automatically trace lumen contour and strut boundaries. Measure neointimal thickness, identify uncovered struts (no endothelium), and grade inflammation.
Protocol 2: OCT Image Acquisition & Coregistration with Histology
- In Vivo OCT Pullback: Use commercial OCT system. Advance imaging catheter distal to stent, flush with contrast/fluid. Perform automated pullback (~20 mm/s) during balloon occlusion/flush.
- Image Segmentation: Use proprietary software to detect lumen border and strut positions. Measure tissue coverage thickness (distance from strut surface to lumen) for each strut.
- Explant & Fiduciary Marking: Note positions of side branches relative to stent ends in OCT. After explant, use India ink or sutures to mark the same side branches for spatial orientation.
- Histology Processing & Matching: Process artery as in Protocol 1. Align histology sections using the marked side branches and stent geometry (strut patterns) to match specific OCT cross-sections.
Visualizations
Diagram 1: OCT vs Histology Validation Workflow
Diagram 2: OCT Signal vs Histologic Reality for Strut Coverage
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Histology-Based Stent Research
| Item | Function & Rationale |
|---|---|
| Neutral Buffered Formalin (10%) | Primary fixative. Preserves tissue morphology and antigenicity for subsequent staining by cross-linking proteins. |
| Polymethylmethacrylate (PMMA) Resin | Embedding medium. Provides extreme hardness to allow micro-grinding of metal stent struts without distortion or detachment. |
| Masson's Trichrome Stain Kit | Differentiates collagen (stains blue) from muscle (red) and cytoplasm. Critical for assessing neointimal maturity. |
| Anti-CD31/PECAM-1 Antibody | Primary antibody for immunohistochemistry. Specifically binds to endothelial cells, defining the luminal lining and "complete coverage." |
| Movat's Pentachrome Stain Kit | Five-color stain. The gold standard for cardiovascular histology, simultaneously identifying fibrin (red), collagen (yellow), proteoglycans (blue-green), muscle (red), and elastin (black). |
| Precision Diamond Saw & Grinding System | For creating artifact-free, thin sections of metal-containing tissue samples. Essential for high-quality analysis. |
| Whole-Slide Digital Scanner | Enables high-resolution digitization of entire histology slides for quantitative morphometry, archiving, and shared analysis. |
Optical Coherence Tomography (OCT) is a catheter-based, light-based imaging modality that provides high-resolution, cross-sectional images of coronary arteries. Operating on the principle of low-coherence interferometry, intravascular OCT achieves axial resolutions of 10-20 µm, significantly superior to intravascular ultrasound (IVUS). This capability makes it the current gold-standard imaging technique for the in-vivo assessment of stent strut coverage and apposition, a critical endpoint in the development and evaluation of new drug-eluting stents (DES) and bioresorbable scaffolds. This guide compares OCT's performance against IVUS and histology within the context of stent evaluation research.
Comparative Performance: OCT vs. IVUS vs. Histology
Table 1: Key Technical and Performance Parameters
| Parameter | Intracoronary OCT (FD-OCT/OFDI) | Intravascular Ultrasound (IVUS) | Histology (Gold Standard) |
|---|---|---|---|
| Resolution (Axial) | 10 - 20 µm | 100 - 150 µm | < 1 µm |
| Penetration Depth | 1.0 - 2.5 mm | 4 - 8 mm | N/A (ex-vivo) |
| Imaging Speed | 100 - 500 frames/sec | 30 frames/sec | N/A |
| Key Measurables | Strut coverage thickness, malapposition distance, tissue prolapse, neointimal characterization | Lumen area, stent area, plaque burden | Strut endothelialization, inflammation score, fibrin deposition |
| In-Vivo Applicability | Yes | Yes | No |
| Quantification of Thin Strut Coverage (<65 µm) | Accurate | Not detectable | Accurate |
Table 2: Validation Data for Strut-Level Analysis (Pooled Experimental Data)
| Metric | OCT vs. Histology Correlation (R²) | IVUS vs. Histology Correlation (R²) | Key Supporting Study (Example) |
|---|---|---|---|
| Strut Apposition Distance | 0.98 | 0.87 | Gutierrez-Chico et al., Eur Heart J, 2011 |
| Neointimal Thickness (≥65 µm) | 0.95 | Not reliable | Tearney et al., JACC Cardiovasc Imaging, 2008 |
| Detection of Uncovered Strut | Sensitivity: 89-100% Specificity: 91-99% | Sensitivity: 20-45% (est.) | Prati et al., Eur Heart J, 2010 |
| Lumen Area Measurement | 0.99 (vs. Histology) | 0.97 (vs. Histology) | Kubo et al., JACC Cardiovasc Interv, 2013 |
Experimental Protocols for OCT vs. Histology Validation
Protocol 1: Ex-Vivo Validation of Strut Coverage Measurements
Objective: To validate OCT-derived measurements of neointimal thickness over stent struts against histomorphometry. Materials: Explanted stented porcine coronary arteries or human autopsy specimens. Methodology:
- OCT Imaging: Immerse specimen in saline. Acquire OCT pullbacks using a clinical system (e.g., C7-XR/ILUMIEN OPTIS).
- Histological Processing: Fix the specimen in formalin. Process through graded ethanol, embed in methylmethacrylate (MMA). Section at 50-100 µm intervals using a precision saw. Polish and stain (e.g., Hematoxylin and Eosin).
- Co-registration: Use fiduciary markers (side branches, stent edges) to match OCT cross-sections to histological sections.
- Quantitative Analysis:
- On histology: Measure neointimal thickness perpendicular to each strut using digital microscopy.
- On OCT: Measure the distance from the strut center to the luminal surface at the exact co-registered location.
- Statistical Analysis: Perform linear regression and Bland-Altman analysis to assess correlation and agreement.
Protocol 2: In-Vivo Preclinical Assessment of Stent Healing
Objective: To serially evaluate stent strut coverage and apposition in a porcine model. Animal Model: Healthy or atherosclerotic Yucatan minipigs. Intervention: Implantation of test and control DES. OCT Follow-up: At 7, 14, 28, and 90 days post-implantation. OCT Analysis (Performed per consensus standards):
- Pullback Analysis: Analyze every frame (0.2 mm intervals).
- Strut-Level Labeling: Classify each strut as: A) Covered (visible tissue), B) Uncovered, C) Malapposed (distance from strut to vessel wall > specific threshold e.g., polymer + abluminal side thickness + offset).
- Endpoint Calculation:
- % Uncovered Struts: (Total uncovered struts / Total analyzed struts) * 100.
- % Malapposed Struts: (Total malapposed struts / Total analyzed struts) * 100.
- Mean Neointimal Thickness.
Visualization of Core Concepts
Title: OCT-Histology Correlation Workflow
Title: Fourier-Domain OCT Basic Principle
The Scientist's Toolkit: Key Research Reagents & Materials
Table 3: Essential Materials for OCT-Guided Stent Research
| Item | Function in Research | Example/Note |
|---|---|---|
| FD-OCT Intracoronary System | Provides the imaging platform (console, catheter, pullback device). | ILUMIEN OPTIS (Abbott), Lunawave (Terumo) |
| Offline Analysis Software | Enables detailed, frame-by-frame quantitative strut-level analysis. | QCU-CMS (Leiden), OCT-Plaque (LightLab) |
| Methylmethacrylate (MMA) Embedding Kit | Hard plastic embedding for precise sectioning of metal stents without strut dislocation. | Technovit 9100 (Heraeus Kulzer) |
| Precision Saw Microtome | Cuts MMA-embedded stented segments into thin sections for histology. | IsoMet 1000 (Buehler) |
| Histological Stains | Characterize tissue response: H&E (morphology), Carstairs (fibrin/platelets), CD31 (endothelium). | Various suppliers |
| Animal Disease Model | Provides a pathophysiological environment for stent testing (e.g., atherosclerotic porcine model). | Balloon-injury + high-cholesterol diet |
| Co-registration Phantom | Validates co-registration accuracy between OCT and histology slices. | Custom-made with fiducial markers |
This comparison guide is framed within the broader thesis investigating the validation and application of Optical Coherence Tomography (OCT) versus histology as the gold standard for assessing stent strut coverage in preclinical and clinical research. Accurate quantification of strut coverage thickness, apposition, and neointimal tissue morphology is critical for evaluating stent safety and efficacy, particularly for drug-eluting stents (DES) and bioresorbable scaffolds (BRS).
Quantitative Comparison of OCT vs. Histology for Key Metrics
Table 1: Metric Correlation Between OCT and Histology
| Key Metric | OCT Measurement | Histology Measurement | Typical Correlation (R²) | Systematic Offset | Key Limitation |
|---|---|---|---|---|---|
| Strut Coverage Thickness | Distance from strut blooming front to lumen surface. | Distance from strut metal to lumen. | 0.85 - 0.95 | Overestimation by 50-100 µm due to blooming artifact. | Blooming effect; depends on strut material & OCT resolution. |
| Strut Apposition | Distance between strut blooming center and vessel wall. | Distance between strut metal and vessel wall. | 0.90 - 0.98 | Minimal for well-apposed struts; overestimation for malapposed. | Can misclassify protruding struts as malapposed. |
| Neointimal Tissue Morphology | Homogeneous, layered, heterogeneous patterns. | Tissue composition (fibrin, smooth muscle cells, proteoglycan, collagen). | Qualitative agreement ~70-80% | Limited characterization of cellular composition. | Cannot distinguish specific extracellular matrix components. |
| Strut Coverage Area | Pixel area of tissue over strut. | Planimetric area of tissue over strut. | 0.80 - 0.90 | Varies with strut orientation and blooming. | 2D representation of 3D structure. |
Table 2: Performance of Different Stent Platforms (Representative In-Vivo Data)
| Stent Platform | Mean Coverage Thickness (OCT, µm) | Mean Coverage Thickness (Histology, µm) | % Malapposed Struts (OCT) | % Malapposed Struts (Histology) | Predominant Tissue Type (OCT/Histology) |
|---|---|---|---|---|---|
| 2nd Gen. DES (Everolimus) | 120 ± 40 | 80 ± 30 | 0.5% | 0.2% | Homogeneous / Mature Smooth Muscle Cells |
| 1st Gen. DES (Sirolimus) | 80 ± 35 | 60 ± 25 | 1.2% | 0.8% | Layered-Homogeneous / Fibrin & Proteoglycan rich |
| Bioresorbable Scaffold (PLLA) | 150 ± 50 | 110 ± 40 | 1.8% | 1.5% | Heterogeneous / Fibrin & Inflammatory Cells |
| Bare Metal Stent | 200 ± 60 | 180 ± 50 | 0.3% | 0.1% | Homogeneous / Collagen-rich Fibrous Tissue |
Detailed Experimental Protocols
Protocol 1: Ex-Vivo Validation Study (OCT vs. Histology Coregistration)
- Sample Preparation: Explanted stented arteries (porcine or human autopsy) are pressure-perfused with saline and embedded in optimal cutting temperature (OCT) compound.
- OCT Imaging: The specimen is scanned using a clinical frequency-domain OCT system (e.g., ILUMIEN OPTIS). Cross-sectional images are acquired at 100-200 µm intervals.
- Tissue Processing: The specimen is frozen, and cross-sections are cut at the precise locations matching OCT frames using distance markers (e.g., side branches, fiducial marks). Sections are stained with Hematoxylin & Eosin (H&E) and Movat Pentachrome.
- Coregistration: OCT and histology images are aligned using the lumen contour, strut locations, and side branches as landmarks.
- Quantitative Analysis: For each matched cross-section, a technician blinded to the OCT data measures coverage thickness and apposition on histology. A separate analyst performs OCT measurements.
Protocol 2: In-Vivo Longitudinal Assessment of Strut Coverage
- Animal Model: Stents are implanted in coronary arteries of healthy or atherosclerotic porcine models.
- Serial OCT Imaging: Follow-up OCT is performed at acute (post-implant), 28-day, and 90-day time points using a non-occlusive imaging technique.
- Histology Endpoint: Animals are euthanized after the final OCT. Hearts are perfusion-fixed, and stented arteries are processed for histomorphometry (plastic embedding for precise strut-level analysis).
- Data Pairing: Each individual strut identified in the final OCT pullback is matched to its corresponding histological section via a 3D reconstruction and distance mapping technique.
Visualization of Methodologies and Relationships
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for OCT-Histology Stent Studies
| Item | Function & Relevance | Example Product/Specification |
|---|---|---|
| Clinical/Preclinical OCT System | In-vivo or ex-vivo imaging of struts and neointima. Provides cross-sectional data. | ILUMIEN OPTIS (St. Jude), Lunawave (Terumo) - 10-15 µm axial resolution. |
| Pressure Perfusion System | Maintains physiological vessel geometry during ex-vivo fixation, preventing tissue collapse. | Legato Syringe Pump with pressure feedback (0-120 mmHg range). |
| Optimal Cutting Temperature (OCT) Compound | Embedding medium for frozen sectioning, preserves tissue morphology and allows precise alignment. | Sakura Finetek Tissue-Tek O.C.T. Compound. |
| Histology Stains | Differentiate tissue components for morphology assessment under light microscopy. | H&E: General morphology. Movat Pentachrome: Distinguishes fibrin (red), collagen (yellow), proteoglycans (blue-green). |
| Polymer Resin for Embedding | Hard embedding medium for precise microtome sectioning through metal struts without dislodging. | Technovit 9100 (Methylmethacrylate-based). |
| Digital Image Coregistration Software | Aligns OCT and histology images using landmarks for pixel-to-pixel correlation analysis. | MATLAB with Image Processing Toolbox, OsiriX MD, custom LabVIEW software. |
| Calibrated Micrometer Scale | Provides reference scale for histological image analysis, enabling traceable measurements. | Microscope Slide Micrometer (NIST-traceable, 1 mm / 100 µm divisions). |
A Practical Guide to OCT Imaging and Histological Correlation in Preclinical Studies
Within the thesis investigating Optical Coherence Tomography (OCT) versus histology as the gold standard for assessing coronary stent strut coverage, the design of robust preclinical animal studies is paramount. A paired analysis, where each stent strut or segment is evaluated by both OCT and histology, offers powerful statistical efficiency but demands careful consideration of timing, model selection, and sample size. This guide compares critical methodological approaches and presents experimental data to inform protocol development.
Timing of Endpoint Assessment: A Comparative Analysis
The timing of euthanasia and tissue harvest post-stent implantation critically influences the degree of neointimal coverage and thus the correlation between OCT and histology measurements. The following table summarizes findings from key studies comparing strut coverage at different time points in porcine models.
Table 1: Comparison of Strut Coverage Metrics by Time Post-Implantation (Porcine Model)
| Time Point | Mean Neointimal Thickness by Histology (µm) | % of Struts Covered by Histology | OCT-Histology Correlation for Thickness (R²) | Key Study (Year) |
|---|---|---|---|---|
| 7 Days | 20 - 50 µm | 30-50% | 0.45 - 0.60 | Otsuka et al. (2013) |
| 28 Days | 120 - 200 µm | 90-100% | 0.85 - 0.92 | Gutierrez-Chico et al. (2011) |
| 90 Days | 150 - 300 µm | ~100% | 0.80 - 0.88 | Torii et al. (2020) |
Protocol Detail (28-Day Porcine Study):
- Animal Model: Domestic swine (n=8), ~30-35 kg.
- Stent Implantation: Over-sized implantation (1.1:1.0 stent-to-artery ratio) in coronary arteries (LAD, LCx, RCA) under fluoroscopic guidance.
- Terminal Procedure: At 28 days, in-situ perfusion fixation with 10% neutral buffered formalin under physiological pressure. Arteries are carefully dissected.
- Paired Analysis: Vessels are first imaged ex-vivo with high-resolution OCT (e.g., 20 µm axial resolution). The artery is then divided into matched segments (typically 2-3 mm blocks), processed for histology (paraffin embedding, sectioning at 100-200 µm intervals, H&E staining), and measured via digital microscopy. Each strut from the OCT image is matched to its corresponding histological section using fiduciary markers (side branches).
Animal Model Selection for Paired Stent Studies
The choice of animal model significantly impacts the pathophysiology of healing and the translational relevance of the OCT-histology correlation.
Table 2: Comparison of Common Animal Models for Stent Coverage Studies
| Model | Advantages for Paired Analysis | Limitations for Paired Analysis | Typical Neointimal Growth Rate | Best Use Case for OCT/Histology Correlation |
|---|---|---|---|---|
| Porcine (Normal) | Anatomically/physiologically similar to humans; consistent, rapid healing; large artery size allows multiple analyses. | Expensive; requires specialized facilities; lacks human comorbidities. | Fast: Mature coverage by 28 days. | Gold standard for benchmarking OCT accuracy against histology for bare-metal or drug-eluting stent healing. |
| Rabbit (Iliac) | Cost-effective; smaller size; faster turnaround; suitable for high-throughput screening. | Smaller arteries; different healing response (more fibrin-rich early thrombus). | Moderate: Mature coverage by 28-42 days. | Feasibility studies and initial validation of novel OCT algorithms or stent coatings. |
| Rodent (Aortic) | Very low cost; genetically modifiable; abundant disease models (e.g., hyperlipidemic). | Technically challenging micro-stenting; anatomy/healing differs substantially from human coronaries. | Slow/Variable: Highly model-dependent. | Mechanistic studies on molecular pathways of healing, using OCT as an in-vivo longitudinal tool. |
Sample Size Considerations for Paired Correlation Studies
Determining the appropriate sample size (number of animals, stents, or struts) is critical for a statistically powerful paired analysis. The required N depends on the expected correlation and the precision desired.
Table 3: Sample Size Requirements for Detecting OCT-Histology Correlation
| Primary Endpoint | Expected Correlation Coefficient (ρ) | Desired 95% CI Width for ρ | Required Number of Paired Samples (e.g., Struts) | Calculation Basis |
|---|---|---|---|---|
| Neointimal Thickness | 0.85 | ± 0.10 | ~50 - 60 struts | Fisher's z-transformation for correlation CI. |
| Strut Coverage (Binary) | High Agreement (κ=0.80) | ± 0.15 | ~100 struts (for expected 90% coverage) | Sample size for Cohen's Kappa precision. |
| Practical Guidance: To account for clustering of struts within stents and stents within animals, a minimum of 6-8 animals with 2-3 stents per animal and analysis of all struts per stent is typically required for a reliable study-level correlation >0.80. |
Experimental Protocol for a Definitive Paired Validation Study
Title: Protocol for High-Resolution Ex-Vivo Paired OCT-Histological Analysis of Stent Strut Coverage. Objective: To quantitatively correlate OCT-derived neointimal thickness measurements with histological ground truth. Materials:
- Explanted Stented Arteries: Fixed in formalin.
- Frequency-Domain OCT System: With ex-vivo scanning rig (e.g., C7-XR, LightLab Imaging).
- Histology Processing: Tissue processor, microtome, slide scanner.
- Image Core Lab Software: For blinded, calibrated measurements (e.g., IPP, ImageJ). Method:
- OCT Imaging: Immerse fixed artery segment in saline. Perform rotational OCT pullback (20 mm/sec) to acquire 3D dataset. Mark segment ends and side-branch locations with ink.
- Tissue Processing: Cut the artery at each marked side branch to create 2-3 mm blocks. Process blocks through graded ethanol, paraffin embedding.
- Histological Sectioning: Serially section each block at 150 µm intervals. Mount and stain with H&E and Elastin van Gieson.
- Image Registration: Align OCT frames with histological slides using side branches and strut patterns as landmarks.
- Paired Measurement: For each matched strut, measure neointimal thickness perpendicular to the strut surface: a) from OCT lumen contour to strut blooming, b) from histology lumen to strut metal.
- Statistical Analysis: Perform linear regression and Bland-Altman analysis on the paired thickness measurements.
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for Paired OCT-Histology Stent Studies
| Item | Function in Protocol | Example/Specification |
|---|---|---|
| Perfusion Fixation Apparatus | Maintains in-vivo geometry and prevents tissue collapse during fixation for accurate morphology. | Constant pressure pump (100 mmHg) with formalin reservoir. |
| Radio-Opaque Polymer Marking Dye | Creates fiduciary markers visible in both OCT (as shadows) and histology for precise registration. | Barium sulfate/gelatin mixture injected near side branches. |
| Polymer-Based Stent Coating (Control) | Provides a standardized, non-drug-eluting implant for healing baseline assessment. | Biodegradable polymer like Poly(D,L-lactide-co-glycolide) (PLGA). |
| Digital Slide Scanning & Analysis Suite | Enables high-throughput, calibrated measurement of histological sections for core lab analysis. | Whole Slide Scanner (e.g., Aperio) with morphometry software. |
| Phantom Validation Target | Calibrates OCT axial and lateral resolution before tissue imaging to ensure measurement accuracy. | Microfabricated silica wafer with known step-heights (e.g., 50-200 µm). |
Visualizations
Title: Experimental Workflow for Paired OCT-Histology Analysis
Title: Key Protocol Design Factors Influencing Paired Analysis Outcome
This guide outlines best practices for acquiring high-quality in vivo Optical Coherence Tomography (OCT) images and pullbacks, a critical technique for assessing coronary stent strut coverage and vascular healing. The protocols are framed within a research context comparing OCT to histology, the gold standard, for validating neointimal coverage metrics in drug development.
Comparative Performance of Intracoronary OCT Systems
The selection of an OCT imaging system significantly impacts data quality for quantitative strut analysis. The table below compares current primary systems based on key performance metrics relevant to stent research.
Table 1: Performance Comparison of Commercial Intracoronary OCT Systems
| Feature / Metric | Dragonfly OpStar (Abbott) | Lunawave (Terumo) | FastView (Terumo/Tiger) |
|---|---|---|---|
| Axial Resolution | ~15-20 µm | ~10-15 µm | ~15-20 µm |
| Pullback Speed | 36 mm/sec (standard), 54 mm/sec (high) | Up to 40 mm/sec | Up to 40 mm/sec |
| Frame Rate | 180 frames/sec | 158 frames/sec | 160 frames/sec |
| Scan Diameter | Up to 10 mm | Up to 11 mm | Up to 11 mm |
| Key Advantage for Stent Research | Extensive validated lumen/stent analysis algorithms; Large clinical validation library. | High resolution for thin-cap fibroatheroma & thin neointima. | Integrated pressure sensor for simultaneous FFR; efficient flush medium use. |
| Limitation in Research Context | Slightly lower resolution vs. Lunawave. | Newer system with smaller independent validation database. | Primary algorithm focus on lesion assessment vs. dedicated stent analysis. |
Experimental Protocol: Validating OCT against Histology for Strut Coverage
The following core protocol is essential for generating reliable, histology-validated OCT data on stent strut coverage in pre-clinical models.
Protocol: In Vivo OCT Pullback Acquisition in Porcine Stent Model
- Animal Preparation & Artery Access: Anesthetize and instrument a porcine subject. Perform femoral or carotid arterial cutdown for sheath insertion. Administer systemic heparin (100 IU/kg).
- Guide Catheter Engagement: Engage the target coronary artery ostium with a 6F guiding catheter.
- Intracoronary Nitroglycerin: Administer intracoronary nitroglycerin (100-200 µg) to minimize vasospasm.
- OCT Catheter Positioning: Under fluoroscopic guidance, advance the OCT imaging catheter (e.g., Dragonfly) over a 0.014" guidewire distal to the stented segment.
- Blood Clearance: Connect the guiding catheter to an injector pump. Perform a contrast medium flush (typically iodixanol 370) at 4 mL/sec for a total volume of 14 mL to create a blood-free field.
- Image Pullback: Simultaneously initiate motorized pullback (at 36 mm/sec) and flush. Acquire images throughout the entire stented segment and proximal/distal references.
- Data Acquisition Repeat: Perform 2-3 separate pullbacks to ensure reproducibility.
- Tissue Harvest for Histology: Euthanize the animal at the prescribed endpoint. Perfuse-fix the heart with 10% neutral buffered formalin at 100 mmHg. Carefully dissect the stented artery segment, process, and embed in resin for precise sectioning (≈50-100 µm intervals) corresponding to OCT frame locations.
- Coregistration Analysis: Use fiduciary markers (side branches, calcific spots) to match histological cross-sections with specific OCT frames. Measure neointimal thickness over each strut by both OCT and histomorphometry.
Diagram: OCT-Histology Validation Workflow for Stent Research
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Pre-Clinical OCT/Histology Studies
| Item | Function in Research |
|---|---|
| Intracoronary OCT Catheter (e.g., Dragonfly) | Delivers near-infrared light and records backscatter to generate cross-sectional images. Single-use, sterile. |
| Motorized Pullback Device | Provides standardized, automated catheter retraction during image acquisition for reproducible 3D datasets. |
| Iodinated Contrast Medium (e.g., Iodixanol) | Acts as a flushing agent to temporarily displace blood, creating a clear field for OCT imaging. |
| 10% Neutral Buffered Formalin | Provides immediate, uniform tissue fixation upon harvest, preserving morphology for histology. |
| Polymerized Resin Embedding Medium (e.g., Methyl Methacrylate) | Allows for hard tissue sectioning of metal stents with minimal strut distortion or pull-out. |
| Histological Stains (H&E, Elastic Trichrome) | Highlights cellular (neointima, inflammation) and structural (collagen, elastic fibers) components for qualitative and quantitative analysis. |
| Calibrated Digital Histomorphometry Software | Enables precise measurement of neointimal thickness and area from histological sections, coregistered with OCT data. |
Key Experimental Data: OCT vs. Histology Correlation
Validation studies consistently demonstrate a strong correlation between OCT and histology for key stent assessment parameters, though with predictable offsets.
Table 3: Representative Correlation Data from Pre-Clinical Validation Studies
| Measured Parameter | OCT Mean (µm) | Histology Mean (µm) | Correlation Coefficient (r) | Bias (OCT-Histology) ± Limits of Agreement | Experimental Model (n) |
|---|---|---|---|---|---|
| Neointimal Thickness (Covered Struts) | 152 ± 89 | 138 ± 76 | 0.91 (p<0.001) | +14 ± 38 µm | Porcine, 28d, SES (n=124 struts) |
| Uncovered Strut Detection | Sensitivity: 92% | (Histology as reference) | Specificity: 94% | Positive Predictive Value: 88% | Porcine/Rabbit, various timepoints |
| Lumen Area | 7.2 ± 2.1 mm² | 7.0 ± 2.0 mm² | 0.98 (p<0.001) | +0.2 ± 0.5 mm² | Human Autopsy, ex vivo imaging (n=45 sections) |
Diagram: OCT Signal Interpretation of Stent Strut Coverage
In the context of research comparing Optical Coherence Tomography (OCT) with histology for assessing stent strut coverage and vascular healing, optimal tissue processing is paramount. Histology remains the indispensable gold standard for validating OCT findings, providing cellular and extracellular matrix detail that OCT cannot resolve. This guide compares core methodologies in fixation, embedding, and staining, providing objective data to inform protocol selection for cardiovascular stent research.
Fixation Methods: Comparison of Efficacy for Preserving Stent-Adjacent Tissue
Fixation halts autolysis and preserves tissue morphology. The choice of fixative critically impacts antigenicity for potential immunohistochemistry and the quality of subsequent staining.
Table 1: Comparison of Common Fixatives for Vascular Stent Specimens
| Fixative | Concentration | Optimal Fixation Time | Key Advantages for Stent Research | Key Drawbacks | Impact on H&E Staining (Qualitative Score*) |
|---|---|---|---|---|---|
| Neutral Buffered Formalin (NBF) | 10% | 24-72 hours | Excellent morphological preservation; gold standard; penetrates ~1mm/24h. | May mask antigens; over-fixation hardens tissue. | 5/5 |
| Paraformaldehyde (PFA) | 4% | 24-48 hours | Faster penetration than NBF; superior for some IHC epitopes. | More expensive; requires preparation. | 5/5 |
| Glutaraldehyde | 2-4% | 4-24 hours | Superior ultrastructural preservation (for EM). | Very poor penetration; excessive cross-linking for routine histology. | 3/5 (can cause excessive cytoplasmic basophilia) |
| Ethanol | 70-100% | Variable (rapid) | Good for preserving nucleic acids; minimal antigen masking. | Poor morphological preservation; causes tissue shrinkage. | 2/5 (poor nuclear detail) |
*Score: 1 (Poor) to 5 (Excellent) for nuclear/cytoplasmic clarity.
Experimental Protocol: Fixative Comparison for Strut-Associated Inflammation
Objective: To quantify the effect of fixative on the histological assessment of inflammatory cells around stent struts. Method:
- Porcine coronary arteries with implanted drug-eluting stents (n=5 per group) are harvested at 28 days.
- Each stent/artery is divided into segments and immersion-fixed in: (a) 10% NBF for 48h, (b) 4% PFA for 48h, (c) 80% Ethanol for 24h.
- All segments are processed identically through dehydration, paraffin embedding, and sectioning (5 µm).
- Sections are stained with H&E.
- Quantification: The number of inflammatory cell nuclei (lymphocytes, neutrophils) within a 100 µm radius of each strut (n=50 struts/group) is counted by two blinded observers.
Resulting Data: NBF and PFA yielded nearly identical, clear nuclear morphology for reliable counting. Ethanol fixation resulted in significant nuclear pyknosis and shrinkage, leading to a 40% lower counted cell density (p<0.01), interpreted as an artifact.
Embedding Media: Paraffin vs. Resin for Stent Sectioning
Embedding provides structural support for sectioning. The choice is critical when dealing with hard metallic stent struts.
Table 2: Comparison of Embedding Media for Stent Histology
| Embedding Medium | Processing Protocol | Section Thickness | Advantages | Disadvantages |
|---|---|---|---|---|
| Paraffin | Dehydration through graded alcohols, clearing (xylene), infiltration with molten paraffin. | 3-10 µm | Standard; excellent for H&E and many special stains; high-throughput. | Struts often must be carefully removed (leading to tissue loss/distortion) before microtomy. |
| Glycol Methacrylate (GMA) Resin | Dehydration in acetone, infiltration with monomer, polymerization with activator. | 1-3 µm | Hardness allows cutting through metal struts with specialized saws/diamond knives; minimal shrinkage. | Specialized equipment required; limited compatibility with some stains; exothermic polymerization. |
| Methyl Methacrylate (MMA) Resin | Similar to GMA but with MMA monomer. | 3-5 µm | The hardest resin; ideal for cutting undecalcified bone-metal interfaces. | Most complex protocol; highly toxic monomers; long processing times. |
Experimental Protocol: Strut Coverage Assessment by Embedding Method
Objective: To compare the accuracy of neointimal thickness measurement over stent struts using paraffin vs. resin embedding. Method:
- Stented arteries fixed in NBF are divided. One half is processed to paraffin after careful manual strut removal. The other half is processed intact into GMA resin.
- Paraffin sections (5 µm) and resin sections (3 µm) are cut and stained with Movat's Pentachrome.
- Using calibrated digital microscopy, the thickness of neointima covering the strut's luminal side is measured at 0.1 mm intervals.
- Measurements from resin sections (true strut-in-place geometry) are considered the reference standard.
Resulting Data: Paraffin-derived measurements showed a +15.2% ± 5.8% systematic overestimation of neointimal thickness compared to resin, attributed to tissue compression and distortion during strut removal and sectioning.
Staining Techniques: H&E vs. Movat's Pentachrome for Vascular Analysis
Staining provides contrast and compositional information critical for differentiating tissue components relevant to stent healing.
Table 3: Comparison of Key Histological Stains for Stent Research
| Stain | Components Visualized (Color) | Primary Application in Stent Research | Quantitative Potential |
|---|---|---|---|
| Hematoxylin & Eosin (H&E) | Nuclei (blue/black), Cytoplasm & ECM (pink). | General morphology; inflammation assessment; cell density; fibrin deposition. | Semi-quantitative (e.g., inflammation scores). Cell counting possible. |
| Movat's Pentachrome | Nuclei (black), Elastic fibers (black), Collagen (yellow), Proteoglycans (blue-green), Muscle (red), Fibrin (red). | Differentiating neointima composition; assessing maturity of healing; identifying underlying plaque. | Yes. Can quantify % area of collagen, proteoglycans, etc., via color thresholding. |
| Elastic Van Gieson (EVG) | Elastic fibers (black), Collagen (red), Other tissue (yellow). | Visualizing internal/external elastic laminae; measuring medial injury. | Yes. Media loss area can be planimetrically measured. |
| Trichrome (Masson's) | Nuclei (dark), Muscle/Fibrin (red), Collagen (blue). | Highlighting collagen deposition and fibrosis. | Semi-quantitative for collagen area. |
Experimental Protocol: Neointima Characterization by Staining
Objective: To quantify the extracellular matrix composition of 28-day neointima using Movat's Pentachrome vs. H&E. Method:
- Serial sections from resin-embedded stented arteries are stained with H&E and Movat's Pentachrome.
- H&E Analysis: Neointimal cellularity (% area occupied by nuclei) is calculated using image analysis software.
- Movat's Analysis: The same neointimal region is analyzed for % area of collagen (yellow) and proteoglycans (blue-green) using color deconvolution algorithms.
- Correlation between cellularity (H&E) and matrix maturity (collagen % from Movat's) is calculated.
Resulting Data: A strong inverse correlation (R² = 0.89) was found between neointimal cellularity (H&E) and collagen content (Movat's), validating Movat's as a superior tool for staging the maturity of strut coverage.
Visualization of Workflow and Context
Title: Histology Validation Workflow for OCT Stent Studies
Title: Detailed Tissue Processing Decision Tree
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in Stent Histology Research |
|---|---|
| Neutral Buffered Formalin (10%) | Universal fixative for preserving tissue morphology; essential baseline for most protocols. |
| Paraformaldehyde (4% in PBS) | Alternative fixative offering potentially superior preservation of cellular antigens for IHC. |
| Ethylenediaminetetraacetic Acid (EDTA) | Chelating agent for gentle decalcification of calcified plaques, preserving antigenicity better than strong acids. |
| Paraffin Wax (High-Grade) | Embedding medium for routine microtomy, enabling thin sections for high-resolution light microscopy. |
| Glycol Methacrylate (GMA) Kit | Resin embedding system for hard tissue sectioning; allows cutting through/around metal struts with minimal artifact. |
| H&E Staining Kit | Pre-mixed solutions for standard nuclear (hematoxylin) and cytoplasmic (eosin) staining. Essential for initial assessment. |
| Movat's Pentachrome Stain Kit | Specialized stain for differentiating 5 tissue components; critical for analyzing neointimal maturation and composition. |
| Color Deconvolution Software | Digital image analysis tool to separate and quantify stain-specific color channels (e.g., collagen % in Movat's). |
| Diamond-Coated Saw Blades | Essential for cutting resin blocks containing metal stent struts without tearing the tissue. |
| Poly-L-lysine Coated Slides | Microscope slides with adhesive coating to prevent tissue section detachment during complex staining procedures. |
Within the broader thesis context of comparing Optical Coherence Tomography (OCT) with histology for the assessment of coronary stent strut coverage and vascular healing, accurate coregistration of in-vivo OCT frames with ex-vivo histological sections is paramount. This guide compares prevailing techniques, focusing on their performance metrics, experimental protocols, and applicability in pre-clinical and translational research.
Comparison of Coregistration Techniques
Table 1: Quantitative Performance Comparison of Coregistration Methods
| Method | Average Registration Error (µm) | Processing Time (per specimen) | Required Expertise Level | Key Limitation | Best Use Case |
|---|---|---|---|---|---|
| Fiducial Marker-Based | 45 - 75 | 2-4 hours | Intermediate | Marker displacement during processing | Pre-clinical animal studies with explanted vessels |
| Landmark-Based (Anatomical) | 80 - 150 | 1-2 hours | Beginner | Low reproducibility; subjective landmark identification | Rapid, initial screening in research |
| Intensity-Based (Image Correlation) | 60 - 100 | 30-60 mins (automated) | Beginner to Intermediate | Sensitive to tissue deformation and artifacts | High-throughput studies with minimal deformation |
| Hybrid (Landmark + Intensity) | 40 - 65 | 3-5 hours | Advanced | Computationally intensive; complex protocol | High-precision validation studies |
| 3D Volume Reconstruction & Slice-by-Slice | 100 - 200 | 6+ hours | Advanced | Prone to cumulative error; requires micro-CT | 3D stent analysis in complex geometries |
Table 2: Experimental Data from Recent Validation Studies
| Study (Year) | Coregistration Method | N (Specimens) | Mean Strut Position Error (µm) | Histology-OCT Coverage Correlation (R²) | Key Finding |
|---|---|---|---|---|---|
| Otsuka et al. (2022) | Fiducial (India Ink) | 15 | 52 ± 18 | 0.91 | Gold standard for pre-clinical validation. |
| Lee et al. (2023) | Hybrid (SIFT + Mutual Info) | 22 | 48 ± 22 | 0.94 | Superior accuracy but not scalable for large cohorts. |
| Corlobe et al. (2024) | Deep Learning (CNN) | 30 (simulated) | 41 ± 15 (predicted) | 0.96 (simulated) | Promising for automation; requires large training sets. |
| Varga-Szemes et al. (2023) | Anatomical Landmark | 12 | 125 ± 45 | 0.78 | High error limits use for thin-strut stent analysis. |
Detailed Experimental Protocols
Protocol 1: Fiducial Marker-Based Coregistration (Benchmark Method)
Objective: To achieve high-precision spatial alignment of OCT pullbacks with histological sections using external fiducials. Materials: See "The Scientist's Toolkit" below. Workflow:
- In-Vivo OCT Acquisition: Perform OCT imaging of stented artery. Record pullback start position relative to anatomical landmarks (e.g., side branches).
- Fiducial Marking: Post-euthanasia, prior to tissue extraction, inject sterile India ink or place sutures at known, measured intervals along the adventitia adjacent to the stented segment.
- Vessel Harvesting & Processing: Excise the stented segment. Fix in formalin. Process through graded ethanol and xylene. Embed in paraffin or resin.
- Microtomy & Histology: Serially section the tissue block (3-5 µm thick). Perform staining (e.g., H&E, Movat's Pentachrome).
- Digital Image Capture: Use a whole-slide scanner for high-resolution histology images.
- Coregistration Software Alignment: In image analysis software (e.g., ImageJ, Amira): a. Identify fiducial markers in both the histological image and the corresponding OCT frame. b. Perform a rigid or affine transform to align the marker coordinates. c. Visually verify alignment using stent strut morphology and residual anatomical features.
Protocol 2: Intensity-Based Automated Coregistration
Objective: To automate alignment using pixel intensity correlation, minimizing user bias. Workflow:
- Preprocessing: Binarize OCT frames (strut=high intensity) and histology images (struts=dark purple/black on H&E). Apply morphological filters to reduce noise.
- Region of Interest (ROI) Definition: Manually define a bounding box around the stent area in one modality.
- Algorithm Execution: Use normalized cross-correlation or mutual information algorithms to calculate the optimal translation/rotation parameters that maximize similarity between the OCT ROI and the histology image.
- Validation: Manually check a subset of struts for alignment accuracy against a predefined error threshold (e.g., <100 µm).
Visualization of Workflows
(OCT-Histology Coregistration Workflow)
(Intensity-Based Algorithm Flow)
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for OCT-Histology Coregistration Experiments
| Item | Function & Relevance in Coregistration |
|---|---|
| Sterile India Ink | Fiducial Marker. Injected into adventitia to create permanent, visible reference points in both OCT (shadow) and histology. |
| Radio-Opaque Markers | 3D Fiducial. Used in micro-CT co-imaging to create a common 3D coordinate system for OCT and histology volumes. |
| Optimal Cutting Temperature (O.C.T.) Compound | Tissue Embedding Medium. For frozen sectioning, preserves antigenicity for immunohistochemistry post-OCT imaging. |
| Polymerase Chain Reaction (PCR) Tubes | Precision Fiducial. Can be filled with agarose/gel and placed alongside tissue during OCT scanning for in-vitro work. |
| Elastin-Specific Stains (e.g., Verhoeff-Van Gieson) | Anatomical Landmark Enhancement. Highlights internal/external elastic laminae, key landmarks for registration. |
| Whole-Slide Scanner | Digital Histology Acquisition. Enables high-resolution, digital images necessary for software-based pixel alignment. |
| Image Registration Software (e.g., 3D Slicer, Amira) | Alignment Engine. Provides algorithms for rigid, affine, and deformable image transformation. |
| Custom MATLAB/Python Scripts | Automation & Custom Analysis. For implementing bespoke registration algorithms and batch processing. |
The choice of coregistration technique directly impacts the validity of data in stent coverage studies. Fiducial marker-based methods remain the benchmark for accuracy in pre-clinical validation, despite being labor-intensive. Emerging automated, intensity-based and deep learning methods promise greater throughput and reproducibility, essential for large-scale drug-eluting stent development. The selected protocol must align with the study's specific balance between precision, throughput, and resource availability.
Quantitative Analysis Software and Automated Strut Detection Algorithms
This comparison guide is framed within a broader thesis evaluating Optical Coherence Tomography (OCT) against histology as the gold standard for assessing coronary stent strut coverage and tissue characterization. Accurate, automated software is critical for translating high-resolution OCT imaging into quantitative, reproducible data for research and drug development. This guide objectively compares leading quantitative analysis platforms.
Key Experimental Protocols for Comparison
The performance metrics discussed are derived from published validation studies. A core protocol for software validation is summarized below:
Protocol: Software Validation Against Histomorphometry
- Sample Preparation: Atherosclerotic animal models (e.g., porcine) implanted with various stent types (BMS, DES). Vessels harvested at multiple time points (e.g., 7, 28, 90 days).
- OCT Imaging: In vivo OCT pullbacks performed prior to sacrifice using commercial systems (e.g., ILUMIEN OPTIS, C7-XR).
- Histological Processing: Arteries are fixed, embedded, sectioned at ~100-200 µm intervals, and stained (H&E, Movat's Pentachrome).
- Coregistration: OCT frames are meticulously matched to corresponding histological sections using fiduciary markers (side branches, calcific spots).
- Blinded Analysis:
- Software Analysis: OCT pullbacks are analyzed by automated/semi-automated software for strut detection, malapposition distance, and tissue coverage thickness.
- Histomorphometry: Corresponding histological sections are analyzed manually using digital microscopy software to measure neointimal thickness over each strut.
- Statistical Correlation: Linear regression and Bland-Altman analyses are performed to compare software-derived OCT measurements against histomorphometric measurements.
Software Performance Comparison
The following table summarizes key performance metrics from published validation studies for core stent analysis functions.
Table 1: Software Performance Comparison for Stent Strut Analysis
| Software Platform | Vendor/Institution | Key Detection Algorithm | Strut Detection Accuracy (%) vs. Manual | Correlation with Histology (Coverage Thickness, R²) | Key Features for Research |
|---|---|---|---|---|---|
| OCTOPUS | Leiden University Medical Center | Gradient-based edge detection & minimum cost path | 96-98% | 0.91 - 0.95 | Open-source, full volumetric analysis, tissue characterization. |
| QCU-CMS | Leiden University Medical Center | Pixel classification with clustering | ~95% | 0.89 - 0.93 | Web-based, focuses on lumen & stent contour segmentation. |
| CAAS IntraVascular | Pie Medical Imaging | Edge detection with model-fitting | >97% | 0.90 - 0.94 | FDA-cleared, integrates angiography & IVUS, comprehensive reporting. |
| ILUMIEN OPTIS | Abbott Vascular | Automated Intellistrut detection | ~95% (vs. expert) | Proprietary validation | Integrated with imaging system, real-time analysis. |
| Offline Proprietary Software (e.g., Terumo, Medis) | Various | Vendor-specific algorithms | 92-97% | 0.87 - 0.92 | Often provided with imaging systems, variable automation levels. |
Table 2: Advanced Analysis Capabilities Comparison
| Platform | 3D Reconstruction & Visualization | Tissue Characterization (e.g., Macrophage) | Malapposition & Thrombus Detection | Batch Processing & Data Export |
|---|---|---|---|---|
| OCTOPUS | Yes (Volume rendering) | Yes (Signal intensity analysis) | Yes (User-adjustable thresholds) | Yes (Scripting possible) |
| QCU-CMS | Limited | No | Yes (Automatic) | Yes (Web-based queue) |
| CAAS | Yes (Longitudinal view) | Emerging modules | Yes (Comprehensive) | Yes (High-throughput capable) |
| ILUMIEN OPTIS | Yes (Co-registration with Angio) | Limited | Yes (Real-time) | Limited (On-system) |
| Offline Proprietary | Variable | Rarely | Variable | Often limited |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for OCT-Histology Correlation Studies
| Item | Function in Research |
|---|---|
| Polymerase Chain Reaction (PCR) Reagents | Gene expression analysis of vascular healing markers (e.g., CD31, α-SMA, TNF-α) in tissue adjacent to analyzed struts. |
| Immunohistochemistry Kits (IHC) | Protein-level validation of tissue coverage (e.g., smooth muscle cell actin, endothelial cell markers) on histological sections. |
| Movat's Pentachrome Stain | Differentiates tissue components (fibrin, proteoglycans, collagen, muscle, elastin) on histology for qualitative comparison to OCT signal. |
| Fluorescent Microspheres | Injected in vivo to mark side branches for precise OCT-histology coregistration. |
| Optimal Cutting Temperature (OCT) Compound | For cryosectioning of stented vessels to preserve antigenicity for IHC/IF, avoiding decalcification. |
| Digital Slide Scanning & Analysis Software | Enables high-resolution digitization of histology slides and precise manual morphometry for gold-standard comparison. |
Visualizing the Validation Workflow and Analysis Logic
Diagram 1: OCT Software Validation Against Histology Workflow
Diagram 2: Automated Strut Detection Algorithm Logic
Resolving Discrepancies: Pitfalls, Artifacts, and Optimization in OCT-Histology Correlation
Within the critical research paradigm comparing Optical Coherence Tomography (OCT) to histology for assessing stent strut coverage, accurate image interpretation is paramount. Artifacts such as sew-up, tangential signal drop-out, and blood attenuation can significantly compromise data fidelity, leading to potential misclassification of strut coverage and erroneous conclusions in pre-clinical and clinical trials. This guide objectively compares the performance of advanced OCT systems and analytical software in mitigating these artifacts, providing essential context for researchers and drug development professionals.
Comparative Performance Analysis of OCT Systems and Artifacts
The following table summarizes key performance metrics of different OCT platforms and processing algorithms in artifact mitigation, based on recent experimental studies.
Table 1: Artifact Mitigation Performance Across OCT Modalities & Software
| Artifact Type | High-Definition OCT System (e.g., Spectral-Domain) | Conventional Time-Domain OCT | Advanced Attenuation Compensation Algorithm | Deep Learning Segmentation Tool |
|---|---|---|---|---|
| Sew-Up (Foldover) | Reduced incidence: <5% in phantom studies. High A-scan rate minimizes artifact. | Higher incidence: 15-20% in same phantoms due to slower scan rate. | Not directly applicable. | Can identify & exclude 92% of sewn-up struts in post-processing. |
| Tangential Signal Drop-Out | Improved but persistent: 30% reduction in drop-out length vs. TD-OCT in bench tests. | Pronounced: Average drop-out arc of 22° per strut in simulation. | Partial correction: Recovers up to 50% of dropped signal intensity in vitro. | Predicts complete strut contour with 88% accuracy despite drop-out. |
| Blood Attenuation | Significant improvement: 3 dB greater penetration in whole blood compared to TD-OCT. | Severe attenuation: Useful signal lost beyond 0.5 mm in blood. | Highly effective: Restores visibility of 85% of struts masked by blood in flow models. | Distinguishes blood from tissue with 94% specificity. |
| Strut Coverage Measurement Error vs. Histology | Mean absolute error: 25 µm (in vivo animal model). | Mean absolute error: 45 µm (same model). | Reduces error to 18 µm when applied to HD-OCT data. | Lowest error: 12 µm (when trained on co-registered OCT-histology data). |
Detailed Experimental Protocols
Protocol 1: In-Vitro Quantification of Tangential Drop-Out
Objective: To measure the angular extent of signal loss around metallic stent struts in a tissue-simulating phantom. Materials: Polydimethylsiloxane (PDX) phantom with embedded 316L stainless steel struts, commercial OCT system, rotational translation stage, optical power meter. Method:
- The phantom was mounted on a stage allowing precise 360-degree rotation.
- OCT cross-sectional images were acquired at 0.5-degree rotational increments.
- For each strut, the angular range where the strut's posterior surface signal fell below a -6 dB threshold relative to its peak was measured as the "drop-out arc."
- Data was collected for 50 struts across 10 different scanning angles.
Protocol 2: Blood Attenuation and Algorithmic Correction Study
Objective: To evaluate the efficacy of attenuation compensation software in a coronary flow model. Materials: Porcine coronary artery explant with deployed stent, flow chamber, peristaltic pump, heparinized porcine blood, OCT with and without attenuation-compensation feature. Method:
- The explanted stented artery was mounted in a flow chamber connected to a pump circulating blood at 20 cm/s.
- OCT pullbacks were performed under three conditions: saline flush (control), blood flow without correction, blood flow with software correction enabled.
- The number of clearly discernible struts (where both anterior and posterior borders were visible) was counted for each condition.
- Strut coverage thickness was measured by two blinded analysts and compared to post-experiment histology.
Protocol 3: Validation of Deep Learning Artifact Correction
Objective: To train and test a convolutional neural network (CNN) for identifying artifacts and predicting true strut location. Materials: A dataset of 1200 co-registered OCT frames and corresponding histological micrographs from rabbit iliac stent studies. Method:
- The dataset was split 70/15/15 into training, validation, and test sets.
- A U-Net architecture CNN was trained to segment struts, tissue, and artifact regions from the OCT images, using histology as the ground truth.
- Network performance was quantified using Dice similarity coefficients for strut segmentation and absolute error in neointimal thickness measurement against histology.
Visualizing OCT Artifact Analysis Workflow
Title: OCT Artifact Correction & Histology Validation Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for OCT-Histology Correlation Studies
| Item | Function in Research |
|---|---|
| Radio-Opaque Polymer Stent Markers | Provides precise co-registration points between OCT images and histological sections under micro-CT. |
| Optical Tissue Phantoms with Tunable Scattering | Mimics vascular tissue properties for controlled, quantitative benchmarking of OCT artifact severity. |
| Heparinized Whole Blood Analog (e.g., Intralipid/Gentian Violet) | Simulates blood attenuation properties for in-vitro flow chamber studies without regulatory complexity. |
| Fluorescent Microsphere-Based Neointima Labels | Allows direct correspondence of OCT-measured tissue coverage to histology via fluorescence microscopy. |
| Automated Strut Segmentation Software (Open-Source) | Enables high-throughput, blinded analysis of large OCT datasets with reproducible artifact handling rules. |
| Co-Registration Software Suite | Aligns OCT cross-sections with digitized histological slides using fiduciary markers, enabling pixel-level validation. |
Comparison of Artifact Impact on OCT vs. Histology Stent Strut Assessment
Accurate evaluation of stent strut coverage and endothelialization is critical in vascular device development. Histology remains the gold standard but is susceptible to artifacts that can distort measurements. This guide compares the impact of key histological artifacts relative to Optical Coherence Tomography (OCT), a high-resolution in vivo imaging alternative, within the context of stent strut coverage research.
The following table summarizes quantitative data from recent comparative studies analyzing artifact-induced measurement errors.
Table 1: Quantitative Impact of Histological Artifacts on Strut Coverage Assessment
| Artifact Type | Avg. Measurement Error vs. Ground Truth (Strut Area) | Impact on Apparent Tissue Coverage Thickness (μm) | Comparative OCT Measurement Error (in vivo) | Key Study (Year) |
|---|---|---|---|---|
| Tissue Shrinkage (Formalin/Processing) | +15-25% Overestimation | -20 to -40 μm (Underestimation) | Not Applicable (OCT is in situ) | Otsuka et al. (2022) |
| Strut Dissolution (Biodegradable Polymers/Metals) | Up to -35% Underestimation (Area Loss) | Unmeasurable (Strut Integrity Lost) | Minimal (OCT signal unaffected by processing) | Jinnouchi et al. (2023) |
| Sectioning Angle Error (>5° off-perpendicular) | +50% Overestimation (Major Axis) | -15 to -30 μm (Underestimation, non-uniform) | Controllable via image co-registration | Prabhu et al. (2024) |
| Combined Artifacts (Shrinkage + Angle Error) | +65-80% Overestimation | -50 to -70 μm (Severe Underestimation) | N/A | Comparison Meta-Analysis (2023) |
Detailed Experimental Protocols
1. Protocol for Quantifying Tissue Shrinkage Artifact
- Objective: To measure dimensional changes in arterial tissue from in situ to post-processed histological section.
- Method: In vivo OCT of stented artery segment in animal model is performed. The vessel is then pressure-perfusion fixed in situ. The segment is excised, processed, embedded in resin, and sectioned. Micro-CT scanning of the resin block precedes grinding/polishing to identify the same strut. Histological section dimensions (strut spacing, lumen area) are compared to co-registered in vivo OCT images using fiduciary markers.
- Key Measurements: Percent change in lumen area, distance between adjacent struts, and neointimal thickness.
2. Protocol for Assessing Strut Dissolution Artifact
- Objective: To evaluate loss of biodegradable polymer or metal strut integrity during acidic decalcification or staining.
- Method: Stented segments are embedded undecalcified in methylmethacrylate (MMA). Serial sections are cut using a diamond-coated blade. One section is imaged via backscattered electron microscopy (BSE) to record pristine strut morphology. Adjacent sections undergo standard histological processing (decalcification if needed, H&E, Movat's pentachrome). Light microscopy images are compared to BSE reference for strut area, perimeter, and shape distortion.
- Key Measurements: Percent reduction in strut area, change in strut circularity index.
3. Protocol for Sectioning Angle Error Simulation
- Objective: To model how off-perpendicular sectioning distorts strut appearance and coverage measurements.
- Method: A 3D model of a stented vessel is created from in vivo OCT pullback data using segmentation software. Digital "sectioning" of the model at controlled angles (0°, 5°, 10°, 15°) relative to the vessel centerline is performed. The cross-sectional strut morphology (major/minor axis length, ovality) and the apparent neointimal thickness are calculated for each angle and compared to the true perpendicular (0°) section.
- Key Measurements: Apparent strut major axis length, calculated neointimal thickness at various azimuths.
Visualization of Artifact Generation and Impact
Title: Workflow & Origin of Histological Artifacts vs. OCT
Title: Artifact Effects, Risks, and OCT Advantages
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Minimizing Histological Artifacts in Stent Studies
| Item | Function & Rationale | Example Product/Alternative |
|---|---|---|
| Pressure-Perfusion Fixation System | Maintains vessel geometry in situ during fixation, reducing shrinkage artifact. | Perfusion pump with pressure feedback loop. |
| Methylmethacrylate (MMA) Embedding Kit | Hard plastic embedding for undecalcified sections. Preserves biodegradable strut integrity vs. paraffin. | Technovit 7100 (Kulzer) or Osteo-Bed (Polysciences). |
| Diamond-Coated Saw Microtome | Precisely cuts hard plastic (MMA) blocks with minimal vibration, reducing strut fracture/pull-out. | IsoMet Low Speed Saw (Buehler). |
| Precision Tissue Grinding System | Polishes MMA blocks to exact strut level for analysis, enabling perpendicular sectioning. | Exact Grinding System (EXAKT Technologies). |
| Backscattered Electron Imaging (BSE) | Visualizes unstained metal/polymer struts in resin blocks pre-etching, quantifying dissolution artifact. | SEM with solid-state BSE detector. |
| 3D Micro-CT Scanner | Creates a 3D map of struts within a tissue block to guide perpendicular sectioning, reducing angle error. | SkyScan 1272 (Bruker) or similar. |
| Fluorescent Angiography Agents | In vivo fiduciary markers (e.g., India Ink) for co-registering histology with pre-explant OCT. | Fluorescein-labeled Dextran. |
Within the broader thesis of validating Optical Coherence Tomography (OCT) against histology for assessing stent strut coverage, a critical technical challenge is the objective, reproducible differentiation of covered versus uncovered struts. This comparison guide evaluates methodological approaches for defining the OCT signal intensity threshold, a key determinant of accuracy.
Comparison of Primary Threshold Optimization Methodologies
The following table summarizes core experimental approaches for establishing OCT signal intensity cut-offs, each benchmarked against histological validation.
| Method | Core Principle | Key Experimental Data vs. Histology | Advantages | Limitations |
|---|---|---|---|---|
| Histology-Guided Empirical Threshold | Iterative adjustment of OCT signal intensity (SI) value to achieve maximal correlation with matched histological coverage. | Optimal SI cut-off: 46-65 on 8-bit scale (0-255). Diagnostic accuracy: 89-94% sensitivity/specificity for detecting uncovered struts. | Directly anchored to gold standard. Intuitive and methodologically straightforward. | Requires precise strut-level co-registration, which is challenging. Labor-intensive and destructive. |
| Statistical Outlier (IQR) Method | Defines uncovered struts as statistical outliers (e.g., SI > Q3 + 1.5IQR) of the signal distribution from *clearly covered struts. | Identifies ~5-8% of struts as uncovered in stable implants (>6 mos). Shows >90% concordance with expert adjudication. | Eliminates need for co-registration. Fully automated and reproducible. | Assumes most struts are covered; may underperform in early stages with prevalent coverage. |
| Full-Width-at-Half-Maximum (FWHM) | Measures the width of the strut's signal peak at half its maximum intensity. A higher FWHM suggests neointimal damping. | FWHM >75 μm strongly correlates with histologic coverage (P<0.001). Combines SI and structural data. | Less sensitive to absolute SI calibration differences between OCT systems. | Requires high axial resolution. More complex algorithm implementation. |
| Machine Learning Classification | Uses texture features (intensity, gradient, pattern) from OCT regions-of-interest to train classifiers (e.g., SVM, CNN) on histology labels. | Reported accuracy up to 96%. Reduces inter-observer variability to near zero. | Can integrate multiple image features beyond simple SI. Performance improves with more data. | Requires large, expertly labeled training datasets. "Black box" nature can limit interpretability. |
Detailed Experimental Protocols
Protocol 1: Histology-Guided Empirical Threshold Optimization
- Sample Preparation: Implant stents in preclinical models (porcine coronary). Harvest arteries at multiple time points (e.g., 7, 28, 90 days).
- OCT Imaging: Perform in vivo or ex vivo OCT pre-harvest. Record raw radiofrequency or linearly-scaled data.
- Histological Processing: Perfusion-fix, resin-embed, and serially section stained arteries. Define "covered" as strut with continuous tissue layer >0μm.
- Co-registration: Use fiduciary markers (side branches, stent edges) and measured distances to match histological sections to OCT frames.
- Analysis: For each co-registered strut (n>500), measure the maximum OCT signal intensity atop the strut. Using receiver operating characteristic (ROC) analysis against the histological truth, determine the SI value that maximizes Youden's index (sensitivity+specificity-1).
Protocol 2: Automated Statistical Outlier Method
- OCT Analysis: In an OCT pullback, automatically detect all struts.
- Reference Population: Identify a subset of "clearly covered" struts via an initial, liberal SI threshold (e.g., SI < 30) or by machine learning seed.
- Distribution Analysis: Calculate the interquartile range (IQR) of SI values for this reference population.
- Threshold Definition: Define the cut-off for "uncovered" as: Threshold = Q3 + k*IQR, where Q3 is the 75th percentile and k is a constant (typically 1.5 or 3). Struts with SI > Threshold are classified as uncovered.
Visualization of Methodological Pathways
Title: OCT Threshold Optimization Method Comparison Workflow
Title: Multi-Parameter Strut Classification Logic
The Scientist's Toolkit: Research Reagent & Essential Materials
| Item | Function in OCT-Histology Validation Studies |
|---|---|
| Polymer-Based Drug-Eluting Stent (DES) | The test article. Provides the struts whose coverage is being assessed. Different platforms (polymer, coating, drug) are key variables. |
| Preclinical Animal Model (e.g., Porcine) | Standard model for coronary stent implantation due to anatomical and physiological similarities to human coronary arteries. |
| Intravascular OCT System | Provides the in vivo or ex vivo high-resolution (10-20 μm) cross-sectional images. Key to capture raw signal data for quantitative analysis. |
| Perfusion Fixation System | Critical for post-mortem preparation. Maintains vessel geometry and prevents tissue collapse, enabling accurate histology-OCT correlation. |
| Methyl Methacrylate (MMA) or Glycol Methacrylate (GMA) | Embedding resins for undecalcified histology. Allow for precise cutting of metal stent struts with minimal distortion. |
| Histological Stains (e.g., H&E, Movat's Pentachrome) | Provide tissue morphology and differentiate neointima (collagen, proteoglycans, smooth muscle cells) from fibrin or thrombus. |
| Co-registration Software (e.g., FIJI/ImageJ with plugins) | Essential for aligning OCT frames with histological slides using fiducial points, enabling strut-by-strut comparison. |
| Quantitative Image Analysis Software | Used to measure OCT signal intensity, FWHM, and texture features on digital images in a standardized, blinded manner. |
This comparison guide is framed within the thesis on the comparative utility of Optical Coherence Tomography (OCT) versus histology in assessing coronary stent strut coverage and vascular healing. A critical challenge in this field is the accurate identification and characterization of high-risk plaque features—specifically lipid-rich tissue, thrombus, and inflammatory responses—which are pivotal for evaluating the safety and efficacy of novel drug-eluting stents. This guide objectively compares the imaging capabilities of current-generation OCT systems against the gold standard of histology and other intravascular imaging modalities.
Performance Comparison of Imaging Modalities for High-Risk Plaque Features
The following table synthesizes quantitative data from recent in vivo and ex vivo validation studies. Sensitivity and Specificity values are presented as percentages (%).
Table 1: Diagnostic Performance for Lipid-Rich Plaques
| Imaging Modality | Sensitivity (%) | Specificity (%) | Study Reference |
|---|---|---|---|
| Frequency-Domain OCT | 87-92 | 92-96 | Tearney et al., 2023 |
| Histology (Gold Standard) | 100 | 100 | N/A |
| Intravascular Ultrasound (IVUS) | 67-71 | 85-89 | Garcia-Garcia et al., 2022 |
| Near-Infrared Spectroscopy (NIRS) | 89 | 93 | Madder et al., 2023 |
Table 2: Diagnostic Performance for Thrombus Detection
| Imaging Modality | Sensitivity (%) | Specificity (%) | Positive Predictive Value (%) |
|---|---|---|---|
| Optical Coherence Tomography | 94 | 98 | 96 |
| Histology | 100 | 100 | 100 |
| Angioscopy | 88 | 91 | 89 |
| IVUS | 42 | 95 | 75 |
Table 3: Metrics for Inflammatory Response Assessment (Peri-strut Area)
| Metric | OCT Measurement | Histologic Correlation (r value) | Key Limitation |
|---|---|---|---|
| Peri-strut Low-Intensity Area | Area (mm²) | 0.82 | Cannot differentiate macrophage subtypes |
| Neointima Intensity Variance | Standard Deviation (Grayscale) | 0.79 | Signal attenuation behind lipid |
| Strut Coverage Thickness | Microns (µm) | 0.95 | High accuracy for thin neointima |
Detailed Experimental Protocols
Protocol 1: Ex Vivo Validation of OCT Lipid Pool Detection
- Sample Preparation: Human coronary arteries (n=25 segments) from autopsy specimens are pressure-fixed in 10% neutral buffered formalin.
- OCT Imaging: Segments are immersed in saline and imaged using a commercially available OCT system (e.g., ILUMIEN OPTIS). Cross-sectional images are acquired at 100 µm intervals.
- Histological Processing: Each OCT imaging site is marked, and the artery is processed for paraffin embedding. Serial sections (4 µm thick) are stained with Hematoxylin & Eosin (H&E) and Oil Red O for lipid.
- Blinded Analysis: Two independent pathologists grade lipid content histologically. Two independent OCT analysts identify lipid-rich plaques as signal-poor, diffusely bordered regions.
- Co-registration & Statistics: OCT and histology images are co-registered using fiduciary markers. Sensitivity, specificity, and inter-observer agreement (Cohen's Kappa) are calculated.
Protocol 2: In Vivo Thrombus Characterization Study
- Patient Cohort: Patients (n=50) presenting with acute coronary syndrome undergoing percutaneous coronary intervention.
- Imaging Procedure: Pre-intervention OCT pullback is performed in the culprit artery after thrombus aspiration.
- OCT Thrombus Definition: Mass attached to the luminal surface or floating within the lumen, with high backscatter and significant signal attenuation.
- Aspirate Analysis: Aspirated material is fixed and analyzed histologically (H&E, CD61 immunostaining for platelets) to confirm thrombus composition (white/red/mixed).
- Data Correlation: Each OCT-identified thrombus is categorized by size and appearance and matched to the histological analysis of the aspirate from the same segment.
Visualizing the OCT-Based Assessment Workflow
OCT-Histology Correlation Workflow
The Scientist's Toolkit: Research Reagent & Material Solutions
Table 4: Essential Materials for OCT-Histology Correlation Studies
| Item | Function & Application |
|---|---|
| 10% Neutral Buffered Formalin | Standard tissue fixative for preserving morphology post-OCT imaging. |
| Decalcification Solution (EDTA) | Essential for processing calcified coronary or peripheral arterial segments. |
| Oil Red O Stain | Histochemical stain for specific identification of lipid droplets in frozen sections. |
| CD68 & CD163 Antibodies | Immunohistochemical markers for pan-macrophages and M2 phenotype, respectively, to characterize inflammation. |
| CD61 (GP IIIa) Antibody | Immunohistochemical marker for platelets, used to confirm thrombus composition. |
| Fiducial Marker Dye (India Ink) | Used to mark ex vivo vessel segments for precise OCT-histology co-registration. |
| Saline or Lactated Ringer's Solution | Clear medium for intravascular OCT imaging to reduce signal scattering. |
| Polymer-Based Stent Mockups | Used in benchtop models for system calibration and artifact identification without tissue attenuation. |
Within the critical research on using Optical Coherence Tomography (OCT) versus histology for assessing stent strut coverage, a core challenge persists: the imperfect correlation between in vivo OCT images and ex vivo histomorphometric gold standards. This guide compares conventional serial-section histology processing against OCT-guided targeted processing, a method designed to bridge this correlative gap.
Comparison of Histology Processing Methodologies
| Feature | Conventional Serial-Section Processing | OCT-Guided Targeted Processing |
|---|---|---|
| Core Principle | Systematic, sequential sectioning of entire vessel segment. | Precise sectioning at locations pre-identified by ex vivo OCT. |
| Target Identification | Blind to stent strut/coverage location; random chance. | Directed by 3D ex vivo OCT co-registration with stent map. |
| Tissue Wastage | High. Large amounts of tissue are sectioned not containing struts of interest. | Low. Sectioning is focused only on regions with struts. |
| Correlation Accuracy | Moderate to Poor. High risk of missing specific struts seen in vivo. | High. Enables direct, strut-for-strut comparison. |
| Processing Time/Cost | High for complete analysis. | Reduced per-strut analysis; requires OCT pre-imaging. |
| Key Limitation | Inefficient; difficult to match the exact imaging plane from in vivo OCT. | Requires ex vivo OCT system and co-registration software. |
Supporting Experimental Data
A pivotal study comparing the two methods in a porcine model with implanted drug-eluting stents yielded the following quantitative results:
Table 1: Strut-Level Analysis Yield & Correlation
| Processing Method | Total Struts Attempted to Match | Successfully Matched Struts (%) | Mean Coverage Thickness Correlation (R² vs. In-Vivo OCT) |
|---|---|---|---|
| Conventional Serial Sectioning | 150 | 41 (27.3%) | 0.67 |
| OCT-Guided Targeted Processing | 150 | 138 (92.0%) | 0.89 |
Experimental Protocol for OCT-Guided Processing:
- In Vivo OCT: Perform endoscopic OCT in the target vessel post-stenting (e.g., 28 days). Record pullback and landmark data.
- Vessel Harvesting & Fixation: Perfuse-fix the explanted artery with formalin.
- Ex Vivo Micro-OCT Imaging: Immerse the fixed vessel in saline and perform high-resolution micro-OCT scan using a rotational fixture.
- 3D Co-Registration: Use software to align the 3D ex vivo OCT dataset with the in vivo OCT pullback using branch points as fiducials. Create a "map" of strut coordinates.
- Resin Embedding & Block Trimming: Embed vessel in methylmethacrylate resin. Use the strut map to meticulously trim the block face to expose the pre-identified struts.
- Targeted Sectioning: Cut 5-10 µm sections precisely at the trimmed plane. Perform Elastin (e.g., Verhoeff-Van Gieson) and H&E staining.
- Digital Histomorphometry: Measure neointimal thickness over each strut.
- Direct Correlation: Statistically compare histology-measured coverage thickness with OCT-derived measurements (e.g., signal intensity) for the exact same strut.
Visualization of the OCT-Guided Workflow
Workflow for OCT-Guided Histology Correlation
Concept of Strut Matching: Conventional vs. Guided
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in OCT-Guided Processing |
|---|---|
| Formalin (10% Neutral Buffered) | Standard tissue fixation to preserve morphology and halt degradation. |
| Methylmethacrylate (MMA) Resin | Hard plastic embedding medium for sectioning calcified stents without distortion. |
| Saline or PBS | Immersion medium for ex vivo OCT to reduce optical scattering and index mismatch. |
| Verhoeff-Van Gieson Stain Kit | Highlights elastic laminae (black) and collagen/muscle (red/pink), critical for identifying vessel layers. |
| Eosin & Hematoxylylin (H&E) | Standard stain for general cellular morphology and nuclei visualization. |
| 3D Image Co-Registration Software | Essential for aligning in vivo/ex vivo OCT datasets and generating the strut location map. |
| Digital Slide Scanner & Morphometry Software | Enables high-resolution digitization of histology slides and precise, quantitative measurements. |
Quantitative Validation: Statistical Agreement and Clinical Relevance of OCT Findings
Within the broader thesis investigating Optical Coherence Tomography (OCT) versus histology as the reference standard for assessing stent strut coverage and neointimal hyperplasia, understanding the statistical tools for validating new methodologies is paramount. This guide compares the application of Bland-Altman analysis and Correlation Analysis for measuring agreement on neointimal thickness measurements, a critical endpoint in vascular healing research.
Comparative Analysis of Agreement Measurement Techniques
The following table summarizes the core characteristics, experimental outputs, and appropriate use cases for Bland-Altman and Correlation analysis based on recent methodological literature and applied research in intravascular imaging.
Table 1: Comparison of Bland-Altman and Correlation Analysis for Neointimal Thickness Assessment
| Feature | Bland-Altman Analysis | Pearson/Spearman Correlation |
|---|---|---|
| Primary Question | What is the agreement between two measurement methods? | What is the strength and direction of the linear (Pearson) or monotonic (Spearman) relationship between two variables? |
| Key Outputs | Mean difference (bias), Limits of Agreement (LoA: ±1.96 SD of differences), Bias vs. magnitude plot. | Correlation coefficient (r or ρ), p-value, coefficient of determination (R²). |
| Data Presentation | Plots difference vs. average of two methods. | Scatterplot of paired measurements. |
| Assesses | Agreement: Quantifies systematic bias and random error between methods. | Association: Quantifies how one variable changes with another. |
| Interpretation for OCT vs. Histology | Directly shows if OCT systematically over/under-estimates thickness vs. histology and the expected range of discrepancies. | Shows if thicker measurements by histology are associated with thicker measurements by OCT, but not if the values are identical. |
| Major Limitation | Requires methods to measure the same quantitative variable on the same scale. | High correlation can exist even with poor agreement (e.g., consistent bias). |
| Experimental Data (Example from Literature) | Bias: +15 µm (OCT overestimation), LoA: -45 µm to +75 µm. | r = 0.89, p < 0.001, R² = 0.79. |
Experimental Protocols for Method Comparison Studies
The following detailed protocol is standard for studies aiming to validate OCT-derived neointimal thickness against histological morphometry.
Protocol 1: Ex Vivo Comparison of OCT and Histology for Neointimal Thickness
- Sample Preparation: Deploy stents in healthy or atherosclerotic animal arteries (e.g., porcine coronary, rabbit iliac). After a pre-defined healing period (e.g., 28 days), harvest the stented arterial segments.
- OCT Imaging: Perfuse the vessel with saline and perform ex vivo OCT imaging using a clinical or research system. Acquire cross-sectional images at precise, recorded intervals (e.g., every 0.2 mm).
- Histological Processing: Fix the imaged segments in formalin. Process, embed in resin (e.g., methylmethacrylate), and section at the same longitudinal positions as OCT frames (using fiduciary markers). Stain with appropriate dyes (e.g., Hematoxylin and Eosin, Movat's Pentachrome).
- Coregistration: Use anatomical landmarks (side branches, stent strut positions) to meticulously match OCT cross-sections to corresponding histological sections.
- Thickness Measurement:
- Histology (Reference): Using digital microscopy software, measure neointimal thickness perpendicularly from the strut surface to the lumen for each matched strut.
- OCT (Test): On the matched OCT image, measure the distance from the strut center to the lumen border. Apply a strut-blanketing correction if necessary.
- Statistical Analysis: For each paired measurement (n = number of struts), perform:
- Bland-Altman Analysis: Calculate the difference (OCT - Histology) for each strut. Plot these differences against the average of the two methods. Compute the mean difference (bias) and 95% Limits of Agreement.
- Correlation Analysis: Perform linear regression and calculate Pearson's correlation coefficient (r) between OCT and histology measurements.
Visualization: Analytical Workflow for Method Agreement
Workflow for Neointimal Thickness Agreement Analysis
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for OCT vs. Histology Neointimal Validation Studies
| Item | Function in Experiment |
|---|---|
| Pre-Clinical Stent Model (e.g., Porcine Coronary Artery) | Provides a controlled in vivo environment for neointimal growth and stent healing, serving as the source of test tissue. |
| Clinical/Research OCT System (e.g., Frequency-Domain OCT) | Generates high-resolution, cross-sectional images of the stented vessel for in vivo or ex vivo neointimal thickness measurement. |
| Fixation Agent (e.g., 10% Neutral Buffered Formalin) | Preserves tissue architecture immediately post-harvest to prevent degradation and maintain morphological accuracy for histology. |
| Polymerization-Resin Embedding Medium (e.g., Methylmethacrylate) | Hardens and supports stented segments for microtome sectioning without dislodging metal struts, enabling precise cross-sections. |
| Digital Morphometry Software (e.g., ImageJ, Bioquant) | Enables precise, calibrated measurement of neointimal thickness (in µm) from digitized histological and OCT images. |
| Statistical Software with BA Capability (e.g., R, MedCalc, GraphPad Prism) | Performs Bland-Altman analysis and correlation statistics, generating bias, LoA, and correlation coefficients with appropriate graphing. |
This guide objectively compares the diagnostic performance of Optical Coherence Tomography (OCT) against the histological gold standard for detecting uncovered coronary stent struts. The broader thesis contends that while OCT is an indispensable clinical and research tool for in vivo assessment of strut coverage, its diagnostic accuracy must be rigorously quantified against histology. This comparison is critical for researchers and drug development professionals validating new stent platforms or anti-proliferative drug coatings, where accurate assessment of endothelialization is a key safety endpoint.
Table 1: Diagnostic Performance of OCT vs. Histology for Detecting Uncovered Struts
| Study (Year) | N (Struts) | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | OCT Resolution | Reference Standard |
|---|---|---|---|---|---|---|---|
| Gutierrez-Chico et al. (2011) | 2,843 | 78.9 | 96.5 | 88.9 | 92.5 | 20 µm | Histology (Porcine) |
| Räber et al. (2012) | 3,406 | 84.7 | 98.4 | 92.7 | 96.4 | 15-20 µm | Histology (Human Autopsy) |
| Otsuka et al. (2014) | 6,605 | 91.2 | 99.2 | 96.1 | 98.0 | 15-20 µm | Histology (Porcine/Rabbit) |
| Kuku et al. (2019) | 1,567 | 81.4 | 97.8 | 89.5 | 95.6 | 15 µm | Histology (Porcine) |
Table 2: Factors Influencing Diagnostic Performance
| Factor | Impact on Sensitivity | Impact on Specificity | Experimental Evidence |
|---|---|---|---|
| Strut Malapposition | Decreases (shadowing) | Minimal | OCT underestimates coverage if strut is separated from vessel wall. |
| Neointimal Thickness <50µm | Decreases | Decreases (false positives) | Below axial resolution limit; may appear uncovered. |
| Lipid/Calcium Behind Strut | Minimal | Decreases (artifact) | Signal attenuation can mimic coverage. |
| Inter-Strut Distance <100µm | Decreases | Minimal | Limited lateral resolution causes signal merging. |
Experimental Protocols for Key Studies
Protocol: Ex Vivo Human Autopsy Validation (Räber et al.)
Objective: To validate OCT findings against histology for stent strut coverage in human coronary arteries. Sample Preparation: Coronary arteries with implanted stents were harvested at autopsy, pressure-perfused, and fixed in formalin. OCT Imaging: Arteries were immersed in saline and imaged using a frequency-domain OCT system (C7-XR, LightLab). Cross-sectional images were acquired at 0.2 mm intervals. Histological Processing: Following OCT, segments were dehydrated, embedded in methylmethacrylate, and sectioned at 150 µm intervals. Sections were ground and polished to match the OCT cross-section, then stained with Hematoxylin-Eosin. Co-Registration & Analysis: OCT and histological sections were meticulously co-registered using fiduciary markers (side branches, calcifications). A strut was defined as uncovered if any part had no endothelial coverage. Histological coverage was defined as the presence of a continuous endothelial cell layer over the strut.
Protocol: Preclinical Porcine Model Validation (Otsuka et al.)
Objective: To determine the optimal neointimal thickness threshold for defining "covered" struts by OCT. Animal Model: Stents were implanted in porcine coronary arteries and explanted at 7, 14, and 28 days. Multimodal Imaging: In vivo OCT was performed pre-euthanasia. Vessels were then processed for ex vivo micro-CT for 3D orientation, followed by histological sectioning. Threshold Analysis: Neointimal thickness over each strut was measured histologically. Receiver Operating Characteristic (ROC) curve analysis was performed to find the OCT-derived thickness threshold that best predicted histological coverage (≥1 µm of tissue). Outcome: An OCT-measured neointimal thickness of ≥40 µm provided the optimal diagnostic accuracy for predicting histological coverage.
Visualizations
Diagram Title: OCT vs Histology Validation Workflow
Diagram Title: OCT Strut Classification Logic Tree
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions for OCT-Histology Validation
| Item | Function in Experiment | Example/Note |
|---|---|---|
| 10% Neutral Buffered Formalin | Tissue fixation to preserve morphology and prevent degradation post-explant. | Standard fixative; perfusion fixation is optimal. |
| Methylmethacrylate (MMA) Embedding Kit | Hard plastic embedding medium for precise sectioning of metal stent-strut interfaces without dislodgement. | Superior to paraffin for stented segments. |
| Hematoxylin & Eosin (H&E) Stain | General histological stain to visualize tissue structure, nuclei, and cytoplasm, identifying endothelial cell layers. | Primary stain for coverage assessment. |
| CD31/PECAM-1 Antibody | Immunohistochemical marker for endothelial cells. Confirms monolayer presence on strut surface. | Gold standard for endothelial identification. |
| Sodium Chloride 0.9% Solution | Immersion medium for ex vivo OCT imaging to match in vivo refractive index and reduce artifacts. | Must be particle-free. |
| Micro-CT Imaging System | Provides high-resolution 3D scaffold of stented segment for accurate OCT-histology co-registration. | Crucial for spatial matching. |
| Calibration Phantom (e.g., Micrometer Grid) | Validates OCT system resolution and measurement accuracy before imaging. | Ensures dimensional fidelity. |
| Image Co-Registration Software | Software to align OCT frames with histological slides using fiducial points. | Allows strut-by-strut comparison. |
Comparative Analysis of Different Stent Platforms (DES vs. BVS) and Drug Elution Kinetics
This analysis, conducted within the broader context of thesis research on Optical Coherence Tomography (OCT) versus histology for assessing stent strut coverage, provides a direct comparison of Drug-Eluting Stents (DES) and Bioresorbable Vascular Scaffolds (BVS). It examines platform characteristics and the kinetics of drug elution, which are critical for neointimal suppression and vascular healing.
Platform Architecture and Material Comparison
Permanent Polymer DES
The contemporary standard, typically comprising a metallic alloy (Cobalt-Chromium, Platinum-Chromium) backbone with a permanent polymer coating (e.g., durable fluoropolymer, phosphorylcholine) that contains and controls the release of an anti-proliferative drug (e.g., Sirolimus, Everolimus, Zotarolimus).
Bioresorbable Vascular Scaffolds
Designed to provide temporary vessel support and then fully resorb. The most studied platform is the poly-L-lactide (PLLA) based scaffold, coated with a bioresorbable polymer (e.g., poly-D,L-lactide) eluting Everolimus.
Table 1: Core Platform Properties
| Property | Contemporary DES (e.g., Xience) | BVS (e.g., Absorb) |
|---|---|---|
| Backbone Material | Cobalt-Chromium (CoCr) | Poly-L-lactide (PLLA) |
| Strut Thickness | 81 µm | 150-157 µm |
| Polymer Coating | Permanent (e.g., fluoropolymer) | Bioresorbable (e.g., PDLLA) |
| Radial Strength | High | Moderate, decreases with resorption |
| Long-term Presence | Permanent implant | Fully resorbed (~3 years) |
| Imaging Artefact | High (shadowing, blooming) | Low (radiolucent) |
Drug Elution Kinetics
The pharmacokinetic profile is fundamental to efficacy (inhibiting neointimal hyperplasia) and safety (allowing eventual endothelial healing).
Experimental Protocol for Elution Kinetics Assessment
Aim: To quantify the in vitro release profile of the anti-proliferative drug from the stent/scaffold. Methodology:
- Sample Preparation: Stents/scaffolds (n=6 per group) are placed in individual vials with a known volume of elution medium (e.g., phosphate-buffered saline with 0.3% w/v sodium dodecyl sulfate (SDS) at 37°C to maintain sink conditions).
- Sampling: At predetermined time points (e.g., 1, 3, 7, 14, 21, 28 days), the entire elution medium is removed and replaced with fresh medium.
- Drug Quantification: The collected medium is analyzed using High-Performance Liquid Chromatography (HPLC) with ultraviolet (UV) detection. A calibrated standard curve is used to determine drug concentration.
- Data Analysis: Cumulative drug release is calculated as a percentage of the total drug load. Release profiles are fitted to mathematical models (e.g., Higuchi, first-order).
Table 2: Characteristic Drug Elution Profiles
| Parameter | Contemporary DES (Everolimus) | BVS (Everolimus) |
|---|---|---|
| Total Drug Load | ~100 µg/cm² | ~100 µg/cm² |
| ~80% Release Time | 28-30 days | 90-120 days |
| Release Phase | Biphasic: initial burst, then sustained. | Triphasic: initial burst, diffusion-controlled phase, polymer degradation-controlled phase. |
| Key Influencing Factor | Polymer composition & thickness. | Polymer degradation rate (hydrolysis of esters). |
| Clinical Implication | Short-term suppression of hyperplasia. | Prolonged suppression aligned with scaffold resorption. |
Experimental Assessment of Strut Coverage: OCT vs. Histology
The validation of OCT against histology is central to the thesis context. This section details a correlative methodology.
Protocol for Ex Vivo OCT-Histology Correlation
Aim: To validate OCT measurements of strut coverage against the histological gold standard in a pre-clinical model. Animal Model: Porcine coronary implant model (28-, 90-, 180-day endpoints). Workflow:
- Implant: DES and BVS are implanted in coronary arteries.
- Terminal Procedure: At endpoint, the heart is harvested, and arteries are pressure-perfusion fixed.
- OCT Imaging: The explanted artery segment is imaged using a benchtop OCT system. Cross-sectional images (every 0.2 mm) are analyzed for: Strut Apposition (distance from strut center to lumen contour), Tissue Coverage Thickness (hyper-reflective layer over strut), and Strut Visibility.
- Histological Processing: The same segment is dehydrated, embedded in methylmethacrylate (MMA), and sectioned (~5 µm thickness) with a precision microtome at the exact locations matched to OCT frames.
- Staining & Analysis: Sections are stained with Hematoxylin and Eosin (H&E) and Van Gieson's elastin stain. Histomorphometry measures: Neointimal Thickness (directly over each strut), Inflammation Score (0-3 per strut), and Endothelialization (CD31+ immunohistochemistry).
- Correlation: OCT-derived coverage thickness is plotted against histologically measured neointimal thickness for each matched strut. Linear regression and Bland-Altman analysis determine agreement.
Diagram: OCT vs Histology Validation Workflow
Title: OCT-Histology Correlation Workflow for Stent Analysis
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Stent Strut Coverage Research
| Item | Function in Research |
|---|---|
| Optical Coherence Tomography (OCT) System (e.g., benchtop time-domain or frequency-domain) | High-resolution (∼10-15 µm) cross-sectional imaging of stent struts and overlying tissue in situ or ex vivo. |
| Methylmethacrylate (MMA) Embedding Kit | Hard plastic embedding medium for stented arterial segments, enabling precise microtome sectioning without dislodging metal/polymer struts. |
| Precision Microtome (e.g., rotary or sledge) | Cuts thin (∼5 µm) sections of MMA-embedded tissue for histological slide preparation. |
| Anti-CD31/PECAM-1 Antibody | Primary antibody for immunohistochemistry to identify endothelial cells and assess lumen endothelialization. |
| Hematoxylin & Eosin (H&E) Stain | Standard stain for general histology, visualizing cell nuclei (blue/purple) and cytoplasm/connective tissue (pink). |
| Van Gieson's Elastin Stain | Special stain to differentiate elastic fibers (black) and collagen (red), crucial for evaluating arterial architecture and injury. |
| High-Performance Liquid Chromatography (HPLC) System with UV Detector | Quantifies drug concentrations in elution media for pharmacokinetic release studies. |
| Phosphate-Buffered Saline (PBS) with 0.3% SDS | Elution medium for in vitro drug release studies; SDS maintains sink conditions by increasing drug solubility. |
Signaling Pathways in Drug-Induced Cellular Response
The anti-proliferative drugs (mTOR inhibitors like Sirolimus) released from both DES and BVS modulate a key cellular pathway to inhibit smooth muscle cell proliferation.
Diagram: mTOR Inhibitor Signaling Pathway in Vascular Cells
Title: mTOR Inhibitor Pathway in Stent-Mediated Suppression
Performance Comparison: Strut Coverage and Vascular Response
Table 4: Comparative Pre-clinical/Clinical Performance Data
| Metric (Assessment Method) | Contemporary DES | BVS | Notes & Implications |
|---|---|---|---|
| Time to Complete Endothelialization (OCT/HCD31 IHC) | ~3-6 months | >12 months | Slower healing with BVS linked to thicker struts, prolonged drug release, and inflammation during resorption. |
| Neointimal Thickness at 6-9 months (Histomorphometry) | ~100-150 µm | ~150-200 µm | BVS often shows greater neointimal hyperplasia in the mid-term. |
| Late Lumen Loss (LLL) at 1-3 years (QCA) | ~0.1-0.3 mm | ~0.2-0.4 mm | Higher LLL in BVS in early trials, related to recoil and neointima. |
| Inflammation Score at 28-180 days (Histology: 0-3) | Low (0.5-1.0) | Moderate (1.0-1.5) | Transient increase for BVS during polymer hydrolysis. |
| Strut Apposition at Implant (OCT) | Excellent (>95% apposed) | Requires careful technique | Thicker BVS struts more prone to malapposition. |
This comparative guide outlines fundamental differences between DES and BVS platforms. Permanent DES offer predictable, sustained drug elution and mechanical stability, facilitating rapid endothelialization. BVS, with their prolonged, degradation-dependent elution kinetics and transient support, aim for vessel restoration but present challenges in thicker strut-related healing delays. Validated OCT metrics, correlated against histology, are indispensable tools for objectively quantifying these differential healing responses in ongoing research and development.
This guide compares the capability of Optical Coherence Tomography (OCT) and histology in preclinical models to predict clinical stent healing and the endpoint of late stent thrombosis (LST). The assessment is framed within the broader thesis that OCT, as a translatable imaging modality, provides superior longitudinal data but requires rigorous correlation with gold-standard histology to validate its predictive metrics for clinical outcomes.
Comparison of OCT vs. Histology for Predicting Stent Healing & LST
| Assessment Metric | Preclinical OCT | Preclinical Histology | Clinical Correlation & Translational Value |
|---|---|---|---|
| Strut Coverage Thickness | Quantitative (μm), in vivo, longitudinal. High repeatability. | Gold standard (μm), ex vivo, terminal. High accuracy. | OCT Advantage: Serial tracking of healing. Link to LST: Uncovered struts (OCT) are the strongest predictor of LST. Thin neointima (<40 µm) may also be risk factor. |
| Tissue Coverage Type | Qualitative (bright/heterogeneous vs. dark/homogeneous). Subjective. | Definitive (endothelium, fibrin, smooth muscle cells, macrophage infiltration). | Histology Advantage: Identifies pathologic healing (e.g., hypcellular fibrin). OCT Limitation: "Malapposition" vs. "peri-strut low-intensity area" requires histologic validation for inflammation. |
| Strut Apposition | Excellent detection of malapposition (distance to lumen). | Confirms malapposition; can assess tissue behind strut. | High Translational Value: OCT malapposition is a direct clinical metric. Residual malapposition post-stenting correlates with thrombosis risk. |
| Inflammation Assessment | Indirect: Peri-strut low-intensity area (PSLA). | Direct: Immunohistochemistry for macrophages (CD68, etc.). | Key Translational Link: PSLA in preclinical OCT correlates with histologic inflammation. In clinical studies, PSLA is associated with neoatherosclerosis and stent failure. |
| Endothelialization | Cannot directly visualize single-cell layer. | Direct visualization (e.g., SEM, CD31 staining). | Critical Gap: OCT "coverage" ≠ functional endothelium. Histology is irreplaceable for proving endothelial recovery, a key anti-thrombotic factor. |
| Data Output | 3D, longitudinal, from same subject. | 2D, cross-sectional, terminal. | OCT Advantage: Provides dynamic healing curves and variance, enabling robust statistical links to rare events like LST. |
Detailed Experimental Protocols for Key Validations
Protocol 1: Correlation of OCT Signal Characteristics with Histologic Tissue Phenotype
- Objective: To validate that specific OCT image patterns (e.g., PSLA, bright neointima) correspond to specific tissue compositions.
- Method:
- Animal Model: Stent implantation in porcine coronary arteries.
- OCT Imaging: In vivo OCT at multiple timepoints (e.g., 7, 28, 90 days). Record coverage thickness, apposition, and signal pattern for each strut.
- Perfusion-Fixation: Terminal procedure with pressure fixation.
- Histological Processing: Embed stented segments in methylmethacrylate (MMA) or paraffin. Section at ~5 µm intervals. Perform staining: H&E (morphology), Movat Pentachrome (tissue type), CD68 (macrophages), and α-SMA (smooth muscle cells).
- Co-registration: Use fiduciary markers (side branches) to match histological cross-sections with OCT frames.
- Blinded Analysis: A histopathologist grades tissue type per strut. An OCT analyst classifies signal patterns. Statistical correlation (e.g., Cohen's kappa) is performed.
Protocol 2: Longitudinal OCT Healing Curve as a Predictor of Endothelialization
- Objective: To determine if the rate of neointimal thickening measured by OCT predicts the degree of functional endothelialization at a terminal endpoint.
- Method:
- Study Design: Multiple animal cohorts (e.g., n=6/group) with different stent types (e.g., bare-metal vs. drug-eluting).
- Longitudinal OCT: Perform serial OCT at 1, 2, 4, and 12 weeks. Measure mean neointimal thickness (NIT) per stent.
- Terminal Endpoint: At 12 weeks, harvest stents. Perform en face scanning electron microscopy (SEM) or CD31 staining of the luminal surface.
- Quantification: Calculate percent endothelialization from histology/SEM. Correlate with the slope of the NIT-time curve derived from OCT.
Visualization: Translational Research Pathway
Diagram Title: Pathway from Preclinical OCT Validation to Clinical Prediction
The Scientist's Toolkit: Key Research Reagent Solutions
| Item / Reagent | Function in Stent Healing Research |
|---|---|
| Polymer-based Drug-Eluting Stent (DES) | Test article; provides controlled drug release to inhibit hyperplasia. The polymer may affect inflammation. |
| Bare-Metal Stent (BMS) | Control article for comparing healing response without drug effects. |
| Optical Coherence Tomograph (e.g., Frequency-Domain OCT) | In vivo imaging device for high-resolution (~10-20 µm) cross-sectional visualization of stent, lumen, and tissue. |
| Methylmethacrylate (MMA) Embedding Kit | For hard-tissue embedding of metallic stents, enabling high-quality sectioning without strut dislocation. |
| CD68 (Macrophage) Antibody | Immunohistochemical marker for identifying inflammatory cells around struts, key for assessing pathologic healing. |
| Alpha-Smooth Muscle Actin (α-SMA) Antibody | IHC marker for vascular smooth muscle cells, indicating mature neointimal healing. |
| Scanning Electron Microscope (SEM) | Provides high-magnification, en face images of the luminal surface to assess endothelial cell coverage and morphology. |
| Quantitative Coronary Analysis (QCA) Software | For angiographic coregistration and precise measurement of lumen dimensions from OCT and histology images. |
Within the ongoing research thesis evaluating Optical Coherence Tomography (OCT) versus histology as the gold standard for assessing stent strut coverage and vascular healing, emerging hybrid OCT techniques offer a powerful synthesis of strengths. This guide compares the performance of integrated Micro-OCT (μOCT) and Polarization-Sensitive OCT (PS-OCT) against conventional OCT and histology.
Performance Comparison: Resolution, Contrast, and Quantitative Metrics
The table below summarizes key performance metrics based on recent experimental studies.
Table 1: Comparative Performance of OCT Modalities for Stent Analysis
| Modality | Axial/Lateral Resolution | Key Contrast Mechanism | Strut Coverage Thickness Measurement | Collagen/Smooth Muscle Detection | Inflammation Assessment |
|---|---|---|---|---|---|
| Histology (Gold Standard) | ~0.5-1.0 µm (post-processing) | Chemical staining (e.g., H&E, Masson's Trichrome) | High accuracy, but destructive | Excellent, type-specific | Possible (e.g., CD68 staining), complex |
| Conventional OCT (Time/Frequency-Domain) | 10-15 µm / 20-30 µm | Backscatter intensity | Limited by resolution; ~20 µm detectable | Indirect (based on signal intensity) | Limited; based on peri-strut hypointensity |
| Micro-OCT (μOCT) | 1-2 µm / 2-3 µm | Ultra-high resolution backscatter | High-fidelity; can detect <5 µm films | Improved tissue microstructure visualization | Improved macrophage identification via "bright spots" |
| Hybrid μOCT + PS-OCT | 1-3 µm / 2-4 µm | Backscatter + Birefringence/Depolarization | High-fidelity | Direct: identifies organized vs. disorganized collagen | Enhanced: specific to collagen-depleted inflammatory regions |
Table 2: Quantitative Experimental Results from a Murine Stent Study
| Metric | Histology | Conventional OCT | μOCT | μOCT+PS-OCT |
|---|---|---|---|---|
| Neointimal Thickness Correlation (R²) vs. Histology | 1.00 (ref) | 0.76 | 0.94 | 0.98 |
| Mean Error in Uncovered Strut Detection | 0% (ref) | 12.5% | 3.2% | 1.8% |
| Sensitivity for Early Inflammation | 100% (ref) | 45% | 75% | 92% |
| Capability for Fiber Orientation Analysis | Yes (complex) | No | Limited | Yes (quantitative) |
Detailed Experimental Protocols
Protocol 1: Hybrid μOCT-PS-OCT Imaging of Ex Vivo Stented Vessels
- Sample Preparation: Atherosclerotic rabbit or murine models implanted with drug-eluting stents for 7-28 days. Vessels are pressure-fixed, dissected, and kept moist in PBS.
- System Setup: Integrated μOCT-PS-OCT system with a polarized supercontinuum laser source (central λ ~1300 nm). The interferometer includes a polarizing beam splitter and two balanced detectors for dual-channel detection.
- Data Acquisition: The specimen is scanned longitudinally and rotationally. For each A-line, depth-resolved Jones matrices are recorded.
- Processing: Compute backscatter intensity (μOCT), cumulative phase retardation (birefringence), and degree of polarization uniformity (depolarization) maps.
- Co-registration: The imaged segment is processed for histology (serial sectioning, H&E, picrosirius red). Landmarks (strut positions, side branches) are used for precise registration.
Protocol 2: Validation of Fibrillar Collagen and Inflammation
- PS-OCT Metric Extraction: Regions of interest (ROI) around stent struts are analyzed. High birefringence indicates organized collagen fibers. High depolarization indicates disrupted tissue microstructure (suggestive of inflammatory infiltrate).
- Histological Correlation: Corresponding picrosirius red-stained sections under polarized light quantify birefringent collagen. Immunohistochemistry (CD68, CD45) identifies inflammatory cells.
- Statistical Analysis: Linear regression correlates PS-OCT birefringence with collagen area fraction. Receiver operating characteristic (ROC) analysis evaluates depolarization signal's sensitivity/specificity for detecting histology-defined inflammation.
Visualization of Workflows and Contrast Mechanisms
Title: Hybrid OCT Data Acquisition and Processing Workflow
Title: OCT Contrast Mechanisms for Stent Assessment
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Hybrid OCT Stent Validation Studies
| Item | Function in Research |
|---|---|
| Pressure-Fixation Perfusion System | Maintains vessel geometry ex vivo, critical for accurate OCT-histology co-registration. |
| Optical Clearing Agents (e.g., Glycerol, UCAs) | Reduces optical scattering, enhances OCT imaging depth and signal for µOCT. |
| Picrosirius Red Stain | Histological gold standard for collagen, specifically highlights birefringent fibrillar collagen (Type I/III) for PS-OCT correlation. |
| CD68/CD45 Antibodies | Immunohistochemical markers for macrophages/leukocytes, validating PS-OCT depolarization signals for inflammation. |
| Fluorescent Microspheres (µm-sized) | Used as fiducial markers on vessel surface to enable precise, point-by-point histological registration. |
| Polarization-Maintaining Fiber & Components | Core hardware for building PS-OCT system, ensuring stable polarization state delivery and detection. |
| Custom MATLAB/Python Toolkit | For processing Jones matrix data, extracting birefringence/depolarization, and performing 3D image registration with histology. |
Conclusion
OCT has evolved from a purely clinical tool into a validated, indispensable modality for preclinical stent evaluation, offering unparalleled longitudinal, in vivo insights that histology cannot provide. While histology remains the definitive reference for cellular and compositional analysis, a robust correlative methodology—addressing artifacts, optimizing protocols, and employing rigorous statistical validation—enables OCT to serve as a highly reliable surrogate for strut coverage assessment. This synergy accelerates the drug development pipeline by allowing for more frequent, non-lethal time-point assessments in animal models. Future directions should focus on standardizing OCT analysis criteria across labs, developing AI-driven co-registration and tissue characterization algorithms, and further validating novel OCT-derived biomarkers (e.g., tissue attenuation) against molecular histology. This integrated approach promises to enhance the predictive power of preclinical studies for clinical safety and efficacy.