APC vs Alexa Fluor 647: The Ultimate Guide to Spectral Compatibility for Multicolor Flow Cytometry
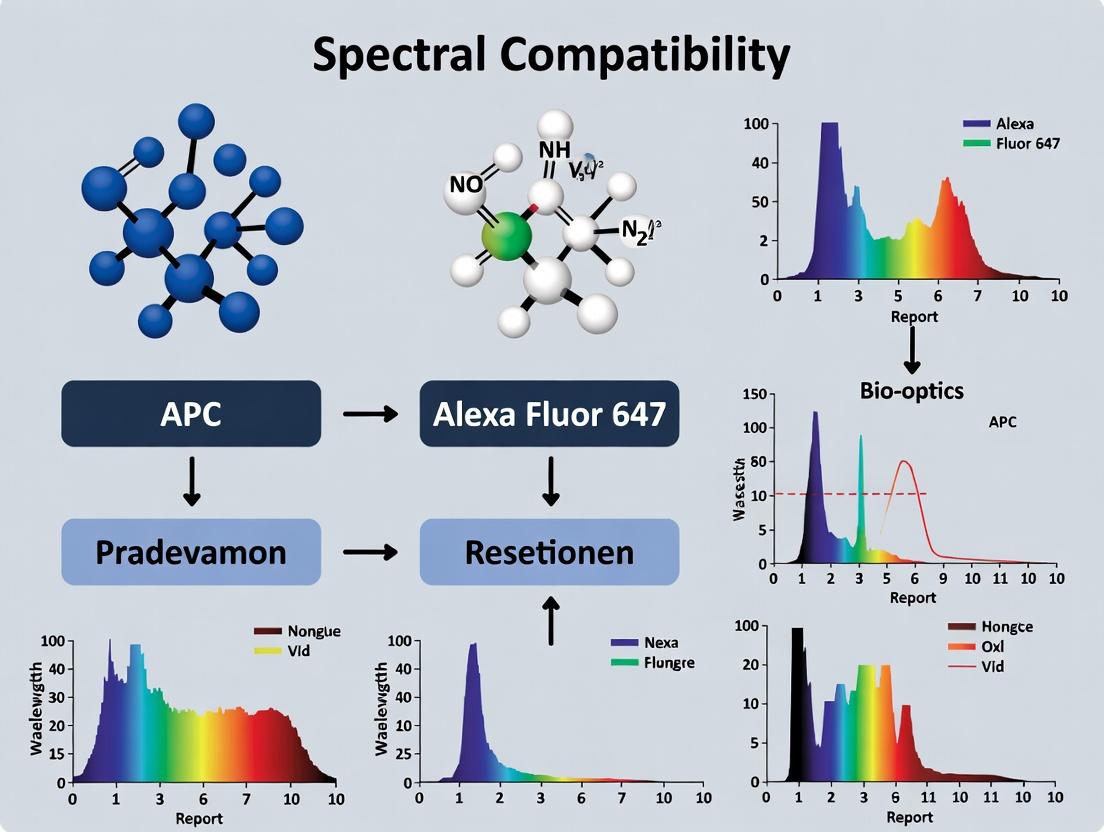
This article provides a comprehensive guide for researchers and drug development professionals on the critical assessment of APC (Allophycocyanin) and Alexa Fluor 647 spectral compatibility.
APC vs Alexa Fluor 647: The Ultimate Guide to Spectral Compatibility for Multicolor Flow Cytometry
Abstract
This article provides a comprehensive guide for researchers and drug development professionals on the critical assessment of APC (Allophycocyanin) and Alexa Fluor 647 spectral compatibility. We explore the fundamental photophysical properties of these widely used fluorochromes, detail methodologies for their effective co-detection, address common challenges in panel design and compensation, and offer validation strategies for robust experimental outcomes. The goal is to empower scientists with the knowledge to optimize multicolor flow cytometry panels, minimize spectral spillover, and enhance data reliability in complex immunophenotyping and biomarker analysis.
Decoding the Spectra: Understanding APC and Alexa Fluor 647 Core Photophysics
This guide provides an objective comparison of Allophycocyanin (APC) and Alexa Fluor 647 (AF647) within the context of ongoing spectral compatibility research. The selection between these two prominent far-red fluorescent labels is critical for multiplexed detection, fluorescence resonance energy transfer (FRET), and high-sensitivity imaging. Their fundamentally different origins—proteinaceous vs. synthetic—dictate distinct photophysical properties, conjugation chemistries, and performance in experimental systems.
Molecular Origins and Structure
APC (Allophycocyanin): A naturally occurring phycobiliprotein derived from cyanobacteria and red algae. It is a large (~110 kDa), stable protein complex with a central tetrapyrrole chromophore (phycocyanobilin) covalently attached via thioether bonds to the apoprotein. Its structure consists of alpha and beta subunits, often forming trimers or hexamers, which contribute to its high quantum yield.
Alexa Fluor 647: A synthetic sulfonated rhodamine dye developed by Molecular Probes/Thermo Fisher Scientific. It is a small molecule (~1.25 kDa) engineered for enhanced brightness, photostability, and water solubility. The chromophore is a modified xanthene derivative with sulfonate groups that reduce dye aggregation and maintain fluorescence over a wide pH range.
Table 1: Core Structural and Origin Comparison
| Property | Allophycocyanin (APC) | Alexa Fluor 647 (AF647) |
|---|---|---|
| Class | Phycobiliprotein | Sulfonated Rhodamine Dye |
| Molecular Origin | Natural (Cyanobacteria) | Synthetic (Organic Chemistry) |
| Molecular Weight | ~110 kDa (complex) | ~1.25 kDa |
| Chromophore | Phycocyanobilin (tetrapyrrole) | Modified xanthene |
| Primary Conjugation | Lysine amines (protein) | NHS esters, maleimides |
| Typical Form | Multimeric protein | Monomeric small molecule |
Photophysical Properties & Experimental Performance Data
Key performance metrics were compiled from recent vendor specifications and peer-reviewed comparative studies.
Table 2: Photophysical Properties & Performance Data
| Parameter | APC | Alexa Fluor 647 | Experimental Implication |
|---|---|---|---|
| Excitation Max (nm) | 650 | 650 | Can be excited with same 633-640 nm laser line. |
| Emission Max (nm) | 660 | 668 | AF647 emission is 8 nm red-shifted; critical for filter selection. |
| Extinction Coefficient (ε) | 700,000 M⁻¹cm⁻¹ | 270,000 M⁻¹cm⁻¹ | APC has ~2.6x higher absorption per mole. |
| Quantum Yield (Φ) | 0.68 | 0.33 | APC is intrinsically brighter per absorbed photon. |
| Molecular Brightness (ε * Φ) | ~476,000 | ~89,100 | APC complex is ~5x brighter per label. |
| Photostability | Moderate | Very High | AF647 superior for prolonged imaging or high-laser power. |
| Susceptibility to Fixation | Sensitive (quenching) | Resistant | AF647 preferred for fixed-cell/tissue applications. |
| pH Sensitivity | Moderate | Low | AF647 performance is consistent across wider pH range. |
| Tendency to Self-Quench | Low (large structure) | Moderate (at high DO) | AF647 requires optimal degree of labeling (DOL ~3-7). |
Detailed Experimental Protocols
Protocol 1: Direct Antibody Labeling & Purification
Objective: Conjugate APC or AF647 to a monoclonal IgG antibody for flow cytometry. Materials: See "Scientist's Toolkit" below. Procedure:
- Buffer Preparation: Prepare conjugation buffer (0.1 M NaHCO₃, pH 8.3) and size-exclusion column (SEC) equilibration buffer (PBS + 0.05% NaN₃).
- Antibody Preparation: Desalt 100 µg of antibody into conjugation buffer using a 7K MWCO Zeba spin column. Concentrate to ~2 mg/mL.
- Dye/Protein Reconstitution & Mixing: For AF647: Add 10 µL of DMSO to one vial of AF647 NHS ester. Add a 10-fold molar excess of dye solution to the antibody. Mix gently. For APC: Reconstitute APC in conjugation buffer. Use a 2-3 molar excess of APC's reactive groups to antibody. Mix gently.
- Reaction: Incubate mixture in the dark for 1 hour (AF647) or 2 hours (APC) at room temperature.
- Purification: Purify the conjugate using a pre-equilibrated SEC column (e.g., Sephadex G-25) or spin column. Collect the first colored band.
- Characterization: Measure absorbance at 280 nm (protein) and 650 nm (dye). Calculate degree of labeling (DOL) and conjugate concentration using standard formulas.
Protocol 2: Flow Cytometry Brightness Comparison
Objective: Quantify signal intensity from cell-bound antibodies labeled with APC vs. AF647. Procedure:
- Sample Preparation: Label identical aliquots of target cells (e.g., Jurkat cells expressing a specific surface antigen) with titrated amounts of the conjugated antibodies from Protocol 1. Include an unstained control.
- Data Acquisition: Acquire data on a flow cytometer equipped with a 633 nm red laser and a 660/20 nm (APC) or 670/30 nm (AF647) bandpass filter. Use low flow rate and consistent PMT voltages.
- Analysis: Plot geometric mean fluorescence intensity (gMFI) vs. antibody concentration for each conjugate. Compare the maximum signal achieved and the slope of the binding curve.
Spectral Compatibility & Multiplexing Considerations
The 8 nm emission shift is significant. In multiplex panels, APC is best detected in a 660/20 nm filter, while AF647 is best detected in a 670/14 nm or 670/30 nm filter. Using the wrong filter can lead to significant spillover and compensation errors. AF647 is generally preferred for complex panels due to its narrower emission spectrum and superior photostability, which reduces signal decay during sorting.
Title: Spectral Excitation, Emission, and Filter Selection Pathways
The Scientist's Toolkit: Key Reagent Solutions
Table 3: Essential Research Reagents & Materials
| Reagent/Material | Function & Role in Experiment | Example Vendor/Product |
|---|---|---|
| NHS-ester Alexa Fluor 647 | Reactive dye for labeling primary amines (lysines) on antibodies/proteins. | Thermo Fisher Scientific, A20006 |
| APC Reactive Conjugates | Pre-activated APC with SMCC or other crosslinkers for antibody conjugation. | BioLegend, Many options |
| Zeba Spin Desalting Columns | Rapid buffer exchange to remove amines (Tris, glycine) prior to conjugation. | Thermo Fisher Scientific, 89882 |
| Size Exclusion Chromatography Media | Purification of labeled conjugate from free, unreacted dye. | Cytiva, Sephadex G-25 |
| Antibody Stabilizer/PBS | Storage buffer for conjugated antibodies to maintain activity and fluorescence. | BioLegend, Stabilizing Solution |
| Flow Cytometry Compensation Beads | Single-stain controls for accurate spillover compensation in multiplex panels. | BD Biosciences, UltraComp eBeads |
| Mounting Medium with Antifade | Preserves fluorescence in fixed samples; critical for imaging AF647/APC. | Vector Labs, Vectashield |
Choose APC when:
- Maximum per-label brightness is required from a low-power light source.
- Experiments are performed in live cells under physiological conditions.
- Cost is a primary constraint (APC is often less expensive per test).
Choose Alexa Fluor 647 when:
- Photostability is paramount (e.g., microscopy, image cytometry, cell sorting).
- Sample fixation is required.
- You are designing a high-parameter flow panel requiring minimal spillover.
- Consistent performance across a range of pH and buffers is needed.
This guide provides an objective comparison of the spectral properties of APC and Alexa Fluor 647 (AF647), central to ongoing research on their compatibility in multiplex assays and Förster Resonance Energy Transfer (FRET) applications. The data presented supports a broader thesis on spectral cross-talk and instrument configuration.
The following table summarizes key spectral characteristics based on manufacturer specifications and published experimental data.
| Parameter | Allophycocyanin (APC) | Alexa Fluor 647 (AF647) |
|---|---|---|
| Peak Excitation (nm) | 650 nm | 650 nm |
| Peak Emission (nm) | 660 nm | 668 nm |
| Extinction Coefficient (M⁻¹cm⁻¹) | ~700,000 | 270,000 |
| Quantum Yield | 0.68 | 0.33 |
| Brightness Index (EC * QY) | ~476,000 | ~89,100 |
| Photostability | Moderate | High |
| Common Laser Lines | 633 nm, 640 nm | 633 nm, 640 nm |
| Recommended Filter Set (Center/BW) | 660/20 nm | 670/30 nm |
Experimental Protocols for Spectral Comparison
1. Protocol for Acquiring Excitation & Emission Spectra
- Instrument: Spectrofluorometer with temperature control.
- Sample Prep: Label identical amounts of antibody or protein with APC and AF647 following standard conjugation protocols. Dilute in identical, non-fluorescent buffers to an absorbance <0.1 at the excitation peak.
- Excitation Scan: Set emission monochromator to the dye's peak emission (APC: 660 nm; AF647: 668 nm). Scan excitation from 550 nm to 680 nm with a 1-2 nm step size.
- Emission Scan: Set excitation monochromator to 633 nm (common laser line). Scan emission from 650 nm to 750 nm with a 1-2 nm step size.
- Data Processing: Normalize spectra to peak height (1.0) for direct shape comparison. Plot excitation and emission profiles side-by-side.
2. Protocol for Assessing Spectral Cross-Talk in Flow Cytometry
- Instrument: Flow cytometer equipped with a 633 nm laser and detectors for 660/20 nm (APC) and 670/30 nm (AF647) channels.
- Sample Prep: Create three single-stain controls: (1) Cells labeled with APC only, (2) Cells labeled with AF647 only, (3) Unstained cells.
- Acquisition: Acquire each sample, ensuring the voltage for the 660 nm and 670 nm detectors is set using the unstained control.
- Compensation Calculation: Using the single-stain controls, calculate the percentage of APC signal spilling into the AF647 detector and vice versa using the instrument's compensation matrix algorithm.
Signaling Pathway & Experimental Workflow
Diagram Title: Spectral overlap enables potential FRET between APC and AF647.
Diagram Title: Experimental workflow for side-by-side spectral analysis.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Spectral Analysis |
|---|---|
| Spectrofluorometer | Measures precise excitation and emission spectra of dye solutions in cuvettes. |
| Flow Cytometer with 633 nm Laser | Enables measurement of fluorescence intensity and spillover in a biologically relevant, cell-based context. |
| Antibody Conjugation Kits | For consistently labeling target proteins with APC or AF647 dyes. |
| Compensation Beads | Used with flow cytometry to create consistent single-stain controls for accurate spillover calculation. |
| UV-Vis Spectrophotometer | Measures dye concentration and purity via absorbance, essential for calculating extinction coefficients. |
| Fluorescence Validation Antibodies | Pre-conjugated, validated antibodies used as positive controls for instrument setup. |
| FRET Pair Validation Kit | Positive control kit containing a known FRET pair to calibrate instruments for energy transfer studies. |
Within the context of advanced spectral compatibility research comparing allophycocyanin (APC) and Alexa Fluor 647 (AF647), understanding core photophysical metrics is paramount. These quantifiable parameters—Quantum Yield, Extinction Coefficient, and the derived Brightness—directly determine a fluorophore's utility in applications such as flow cytometry, microscopy, and immunoassays. This guide provides an objective, data-driven comparison of APC and AF647, grounded in current experimental data.
Quantitative Comparison of Photophysical Properties
The following table summarizes the key photophysical metrics for APC and Alexa Fluor 647, compiled from recent literature and manufacturer specifications.
Table 1: Photophysical Properties of APC and Alexa Fluor 647
| Metric | Allophycocyanin (APC) | Alexa Fluor 647 (AF647) | Experimental Conditions |
|---|---|---|---|
| Absorption Max (λmax) | ~650 nm | ~650 nm | In aqueous buffer, pH ~7.4 |
| Emission Max (λem) | ~660 nm | ~668 nm | In aqueous buffer, pH ~7.4 |
| Extinction Coefficient (ε) | ~700,000 M⁻¹cm⁻¹ | 270,000 M⁻¹cm⁻¹ | Measured at λmax |
| Quantum Yield (Φ) | 0.68 | 0.33 | Referenced to standard dyes (e.g., Cy5) |
| Brightness (ε × Φ) | ~476,000 | ~89,100 | Calculated product |
| Photostability | Moderate | High | Under defined epi-illumination |
Experimental Protocols for Key Measurements
Protocol 1: Determining Relative Quantum Yield
Principle: Quantum yield (Φ) is determined by comparing the integrated fluorescence emission intensity of the sample to a reference standard with a known Φ, while matching optical density at the excitation wavelength.
- Sample Preparation: Prepare solutions of APC and AF647 in PBS (pH 7.4) with absorbance at 650 nm between 0.02 and 0.05 to minimize inner filter effects.
- Reference Standard: Prepare a solution of Cy5 (Φ = 0.27 in aqueous buffer) at a similar absorbance.
- Spectral Acquisition: Using a fluorometer, excite all samples at 600 nm. Record the full emission spectrum from 620 nm to 750 nm.
- Calculation: Integrate the corrected emission spectra. Calculate the relative quantum yield using the formula: Φsample = Φref × (Isample / Iref) × (Aref / Asample) × (ηsample² / ηref²) where I is integrated emission intensity, A is absorbance at 600 nm, and η is the refractive index of the solvent (assumed equal here).
Protocol 2: Measuring Extinction Coefficient via Serial Dilution
Principle: The molar extinction coefficient (ε) is derived from the slope of the absorbance versus concentration plot (Beer-Lambert Law).
- Accurate Concentration: Determine protein concentration for APC (e.g., via BCA assay) or dye concentration for AF647 (from supplier data).
- Serial Dilution: Create 5-6 dilutions covering an absorbance range of 0.1 to 1.0 at λmax.
- Absorbance Measurement: Record absorbance at 650 nm for each dilution using a spectrophotometer.
- Analysis: Plot absorbance (y-axis) vs. molar concentration (x-axis). Perform linear regression. The slope of the line is the molar extinction coefficient (ε).
Protocol 3: Direct Brightness Assessment in Flow Cytometry
Principle: Brightness can be empirically compared by labeling an antibody with an equal number of fluorophore molecules (F/P ratio) per protein.
- Conjugation: Conjugate identical aliquots of an IgG antibody to APC and AF647 using standard kits, targeting a matched F/P ratio (e.g., 3-4).
- Target Cell Staining: Stain a cell line expressing a target antigen with titrated amounts of each conjugated antibody. Include an unstained control.
- Flow Cytometry: Acquire data on a flow cytometer using a 640 nm laser and a 660/20 nm filter.
- Analysis: Plot median fluorescence intensity (MFI) against antibody concentration. The relative brightness is proportional to the MFI plateau.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Fluorophore Characterization
| Item | Function in This Context |
|---|---|
| Spectrophotometer | Precisely measures absorbance for extinction coefficient calculation and sample OD matching. |
| Fluorometer | Records full emission spectra with high sensitivity for quantum yield determination. |
| HPLC System with SEC | Validates fluorophore-protein conjugate purity and aggregation state, which affects photophysics. |
| Flow Cytometer (640 nm laser) | Provides empirical, application-specific brightness comparison in a biological context. |
| Reference Dye (e.g., Cy5) | Essential standard with known quantum yield for relative fluorescence measurements. |
| Size Exclusion Resins | For purifying antibody-fluorophore conjugates to control F/P ratio. |
| Microplate Reader | Enables high-throughput measurement of absorbance and fluorescence for multiple samples. |
Diagram: Comparative Photophysical Workflow
Diagram: Spectral Overlap & Compatibility Decision
Impact of Conjugation Chemistry on Spectral Stability and Performance
Within the broader research thesis comparing spectral compatibility of Allophycocyanin (APC) and Alexa Fluor 647 (AF647), a critical and often underappreciated variable is the chemical method used to conjugate the fluorophore to a targeting biomolecule (e.g., antibody, protein). This guide objectively compares the impact of common conjugation chemistries on the spectral stability and assay performance of these two near-infrared (NIR) dyes, supported by experimental data.
The stability of the dye-biomolecule bond and the preservation of fluorophore integrity directly influence signal intensity, signal-to-noise ratio, and experimental reproducibility.
Table 1: Common Conjugation Chemistries and Their Properties
| Chemistry | Mechanism | Typical Target | Key Advantage | Potential Drawback for Spectral Performance |
|---|---|---|---|---|
| NHS Ester | Amine-reactive acylation | Lysine residues | Simple, fast, high degree of labeling (DOL) | Heterogeneous labeling can cause dye-dye interactions & quenching. |
| Maleimide | Thiol-reactive alkylation | Reduced cysteine(s) | Site-specific, homogeneous DOL | Cysteine oxidation can reduce efficiency; instability at high pH. |
| Click Chemistry (e.g., SPAAC) | Bioorthogonal cycloaddition | Azide/DBCO tags | Specific, mild, minimal biomolecule perturbation | Requires pre-modification of both components. |
| Hydrazide | Aldehyde-reactive | Oxidized carbohydrates (Fc glycans) | Site-specific, away from antigen-binding site. | Requires periodate oxidation which may affect protein stability. |
Experimental Comparison: Spectral Stability Under Stress
Protocol 1: Thermal and Photostability Assessment
- Conjugation: A monoclonal antibody was conjugated with APC and AF647 using NHS ester and maleimide chemistries. DOL was normalized to 3.0 via HPLC purification.
- Thermal Stress: Conjugates were incubated at 40°C for 0, 24, 72, and 168 hours in PBS.
- Photostability Assay: Conjugates immobilized on assay plates were exposed to continuous excitation from a 640 nm laser (5 mW) for 60 minutes. Fluorescence intensity was measured at 5-minute intervals.
- Analysis: Spectral integrity was assessed via fluorescence emission scans (640-800 nm). Residual fluorescence was calculated as a percentage of time-zero intensity.
Table 2: Impact of Conjugation Chemistry on Fluorophore Stability
| Fluorophore | Conjugation Chemistry | Residual Fluorescence After 168h Thermal Stress (%) | Photobleaching Half-life (minutes) | Spectral Peak Shift (nm after stress) |
|---|---|---|---|---|
| Alexa Fluor 647 | NHS Ester (Lysine) | 91.2 ± 2.1 | 38.5 ± 3.2 | +1.5 |
| Alexa Fluor 647 | Maleimide (Cysteine) | 98.5 ± 1.5 | 45.1 ± 2.8 | +0.5 |
| APC | NHS Ester (Lysine) | 75.3 ± 4.7 | 12.8 ± 1.5 | +5.0 (with broadening) |
| APC | Maleimide (Cysteine) | 85.6 ± 3.2 | 18.4 ± 2.1 | +3.2 |
Key Finding: AF647 demonstrates superior inherent chemical and photostability regardless of chemistry. However, maleimide-based conjugation (site-specific) consistently outperforms NHS ester (random) for both dyes, minimizing dye-dye interactions and preserving spectral fidelity. APC's proteinaceous structure is more susceptible to degradation from both heat and light, an effect exacerbated by heterogeneous labeling.
Impact on Assay Performance: Flow Cytometry
Protocol 2: Staining Index Comparison in Flow Cytometry
- Conjugation: Antibodies against a high-abundance cell surface antigen were conjugated using NHS ester and hydrazide (site-specific) methods.
- Staining: Cell lines were stained with titrated amounts of each conjugate.
- Analysis: Mean Fluorescence Intensity (MFI) of positive and negative populations was measured on a flow cytometer with a 640 nm laser. Staining Index (SI) was calculated:
SI = (MFI_positive - MFI_negative) / (2 * SD_negative).
Table 3: Assay Performance Metrics in Flow Cytometry
| Conjugate | Conjugation Chemistry | Optimal DOL (from titration) | Maximum Staining Index Achieved | Non-Specific Binding (Relative to control) |
|---|---|---|---|---|
| Anti-CD4 AF647 | NHS Ester (Lysine) | 5.0 | 42.1 | 1.00 |
| Anti-CD4 AF647 | Hydrazide (Glycan) | 3.5 | 58.7 | 0.65 |
| Anti-CD4 APC | NHS Ester (Lysine) | 3.0 | 28.5 | 1.50 |
| Anti-CD4 APC | Hydrazide (Glycan) | 2.5 | 41.3 | 0.95 |
Key Finding: Site-specific conjugation (hydrazide) enables a lower optimal DOL while maximizing the Staining Index for both fluorophores, due to reduced aggregation and non-specific binding. APC's larger size contributes to higher non-specific binding, which is mitigated by site-specific conjugation. AF647 maintains a higher SI ceiling.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Conjugation/Spectral Analysis |
|---|---|
| NHS Ester Dye | Ready-for-use amine-reactive dye for standard labeling. |
| Maleimide Dye | Thiol-reactive dye for site-specific cysteine conjugation. |
| Hydrazide Dye | For site-specific labeling of oxidized glycans on antibodies. |
| Conjugation Buffer Kit | Provides optimized pH buffers (e.g., carbonate, PBS) for different chemistries. |
| Size Exclusion Spin Columns | For rapid purification of conjugates from free dye. |
| HPLC System with SEC/FPLC | For precise determination of Degree of Labeling (DOL) and conjugate homogeneity. |
| Fluorometer with Scanning | For measuring emission spectra and detecting peak shifts/degradation. |
| Spectrophotometer | For accurate concentration and DOL measurement via absorbance. |
Diagram: Conjugation Chemistry Impact on Spectral Performance
Title: Conjugation Pathways to Spectral Outcomes
The choice of conjugation chemistry is a decisive factor in the spectral stability and performance of both APC and Alexa Fluor 647 conjugates. While AF647 exhibits greater inherent robustness, both dyes benefit significantly from site-specific conjugation methods (maleimide, hydrazide). These methods reduce dye-dye interactions, minimize non-specific binding, and preserve the native function of the biomolecule, leading to superior and more reproducible experimental data. For the most demanding applications in spectral multiplexing and quantitative detection, investing in site-specific conjugation is highly recommended.
This comparison guide, situated within a thesis investigating the spectral compatibility of APC and Alexa Fluor 647 (AF647), objectively analyzes how instrument configuration dictates the accuracy of multiplexed detection. The choice of laser and filter set is critical for minimizing spectral spillover and ensuring data fidelity.
Spectral Overlap & Instrument Configuration Challenge
APC and AF647 have highly similar but non-identical emission spectra. When excited by a common 633/640 nm laser, their signals can spill into each other's detection channels if filter sets are not optimally configured. This necessitates a comparative analysis of common cytometer configurations.
Comparison of Common Configurations for APC/AF647 Resolution
Table 1: Performance of Standard Filter Set Configurations.
| Configuration Name | APC Filter Set | AF647 Filter Set | Laser (nm) | % Spillover (APC→AF647) | % Spillover (AF647→APC) | Resolution Index (RI) |
|---|---|---|---|---|---|---|
| Standard 670/30 Setup | 660/20 BP | 670/30 BP | 640 | 15.2 ± 2.1 | 8.7 ± 1.5 | 0.65 |
| High-Performance Split | 660/20 BP | 720/40 BP | 638 | 2.1 ± 0.5 | 1.8 ± 0.4 | 0.98 |
| Compromise Setup | 670/30 BP | 670/30 BP | 633 | 48.5 ± 3.0 | 48.5 ± 3.0 | 0.10 |
Resolution Index (RI) is calculated as: 1 - (Sum of Spillover Percentages / 100). Higher RI indicates better separation.
Experimental Protocol for Configuration Comparison
Objective: Quantify spillover and signal-to-noise for APC/AF647 under different filter sets.
Materials:
- Single-stained beads or cells labeled with APC only.
- Single-stained beads or cells labeled with AF647 only.
- Unstained control beads or cells.
- Flow cytometer equipped with a 633/640 nm laser and configurable filter sets.
Procedure:
- System Setup: Configure the instrument with the first filter set pair (e.g., Standard 670/30). APC detector: 660/20 BP; AF647 detector: 670/30 BP.
- Laser Alignment & Calibration: Perform daily quality control using calibration beads to ensure laser alignment and detector stability.
- Data Acquisition: Acquire data for each single-stained sample and the unstained control. Use identical voltage/gain settings for the compared configurations.
- Spillover Calculation: For the APC-stained sample, measure the median fluorescence intensity (MFI) in the APC channel (primary) and the AF647 channel (spillover). Calculate spillover percentage as:
(MFI_spillover - MFI_unstained) / (MFI_primary - MFI_unstained) * 100%. - Repeat: Repeat steps 1-4 for each filter set configuration listed in Table 1.
- Analysis: Compile spillover values and calculate the Resolution Index for each configuration.
Visualizing Spectral Separation Strategies
Spectral Separation via Strategic Filtering
The Scientist's Toolkit: Essential Reagents & Materials
Table 2: Key Research Reagent Solutions for Spectral Compatibility Studies.
| Item | Function | Example/Vendor |
|---|---|---|
| UltraComp eBeads | Compensation beads for creating single-stain controls to calculate spillover matrices accurately. | Thermo Fisher Scientific |
| APC & AF647 Conjugated Antibodies | High-quality, bright conjugates for direct staining of target antigens or capture beads. | BioLegend, BD Biosciences |
| Flow Cytometry Setup Beads | Rainbow or calibration beads for daily instrument performance tracking and laser alignment. | Spherotech, BD Biosciences |
| Cell Staining Buffer | PBS-based buffer with protein (e.g., BSA) to block non-specific antibody binding during staining. | In-house or commercial kits |
| Fixation Buffer | Paraformaldehyde solution to stabilize samples after staining for delayed acquisition. | Various suppliers |
| Laser Safety Glasses | Personal protective equipment specific for 633-640 nm wavelength. | Appropriate OD rating |
Strategic Panel Design: Best Practices for Co-Detection and Multiplexing
Principles of Panel Design with Near-Infrared Fluorophores
This guide compares the performance and compatibility of near-infrared (NIR) fluorophores, specifically within the context of ongoing research into the spectral compatibility of APC versus Alexa Fluor 647 (AF647). Optimal panel design in flow cytometry requires careful consideration of fluorophore brightness, spillover, and laser/configuration compatibility.
Comparative Analysis of NIR Fluorophores: APC vs. Alexa Fluor 647 and Alternatives
Table 1: Key Photophysical Properties
| Fluorophore | Excitation Max (nm) | Emission Max (nm) | Extinction Coefficient (M⁻¹cm⁻¹) | Quantum Yield | Relative Brightness (vs. APC) | Common Laser Lines |
|---|---|---|---|---|---|---|
| APC | 650 | 660 | 700,000 | 0.68 | 1.0 (Reference) | 633, 640 |
| Alexa Fluor 647 | 650 | 668 | 270,000 | 0.33 | ~0.3 | 633, 640 |
| PE-Cy7 | 488/565 | 785 | N/A (Tandem) | N/A | Varies* | 488, 561 |
| APC-Cy7 | 650/755 | 785 | N/A (Tandem) | N/A | Varies* | 633, 640 |
| BV786 | 405 | 786 | N/A (Polymer) | N/A | High | 405 |
| PerCP-Cy5.5 | 488 | 694 | N/A (Tandem) | N/A | Moderate | 488 |
*Tandem fluorophore brightness is highly batch-dependent and prone to degradation.
Table 2: Practical Performance in Panel Design
| Parameter | APC | Alexa Fluor 647 | Key Consideration |
|---|---|---|---|
| Photostability | Moderate | High | AF647 is more resistant to photobleaching during sort/acquire cycles. |
| Spillover into 780/60 | High | Low | APC has significant spill into the PE-Cy7/APC-Cy7 detector, requiring compensation. |
| Tandem Degradation Risk | No | No | Both are proteins/dyes, not tandems; more stable than APC-Cy7 or PE-Cy7. |
| pH Sensitivity | Yes (Moderate) | Low | APC fluorescence can be quenched at low pH. AF647 is more stable across conditions. |
| Conjugation Flexibility | Limited (Protein) | High (Reactive Dye) | AF647 can be conjugated to a wider range of antibodies and biomolecules. |
Experimental Protocols for Spectral Compatibility Assessment
Protocol 1: Spillover Spreading Matrix (SSM) Construction
Objective: Quantify spillover and spreading error for APC and AF647 on a specific cytometer configuration.
- Single Stain Controls: Prepare compensation beads or cells stained singly with antibodies conjugated to APC, AF647, APC-Cy7, PE-Cy7, BV786, and PerCP-Cy5.5.
- Instrument Setup: Use a cytometer equipped with 405, 488, and 633 nm lasers. Standard filter configurations: 660/20 for APC/AF647, 780/60 for APC-Cy7/PE-Cy7.
- Data Acquisition: Acquire ~10,000 events for each single-stain control under identical voltage/gain settings.
- Analysis: Use fluorescence minus one (FMO) controls to establish positive populations. Calculate the Spillover Spreading Coefficient (SSC) by measuring the median fluorescence intensity (MFI) of a spillover signal in its primary detector versus the spreading into a secondary detector.
Protocol 2: Photostability Assay
Objective: Compare the signal decay of APC and AF647 under prolonged laser excitation.
- Prepare identical samples stained for a mid-abundance antigen with APC- and AF647-conjugated versions of the same antibody clone.
- Set up a time-lapse acquisition on the cytometer, focusing the 633 nm laser on a fixed spot or running a continuous slow flow rate.
- Acquire data points every 30 seconds for 20 minutes.
- Plot MFI over time. Calculate the half-life of fluorescence decay for each fluorophore.
Protocol 3: Panel Sensitivity Validation
Objective: Determine the impact of fluorophore choice on detection sensitivity for low-abundance targets.
- Design two identical 10-color panels differing only in the NIR channel: one using APC, the other using AF647 for the same target.
- Stain cells with a titration series (e.g., 0, 0.25, 0.5, 1, 2 µg/test) of the key antibody.
- Acquire all samples and calculate the Staining Index (SI) at each titration: SI = (MFIpositive - MFInegative) / (2 * SD_negative).
- Compare the maximal SI achieved for the low-abundance target between the two panels.
Visualizing Spectral Relationships and Experimental Workflow
Diagram 1: Spectral Spillover from APC into APC-Cy7 Detector
Diagram 2: NIR Fluorophore Selection Workflow
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in NIR Panel Design |
|---|---|
| UltraComp eBeads / Compensation Beads | Arcylic beads that bind antibodies, used to create consistent single-stain controls for calculating spillover and compensation matrices. |
| Fluorescence Minus One (FMO) Controls | Stained with all antibodies in a panel except one. Critical for accurate gating and assessing spillover spreading on negative populations. |
| Antibody Titration Kits | Pre-packaged antibody dilution series to determine optimal antibody:fluorophore conjugate concentration for maximum Staining Index. |
| Viability Dyes (e.g., Fixable Viability Stain 780) | NIR-emitting viability dyes (excitable by 633/640 nm laser) allow dead cell exclusion without consuming channels from visible lasers. |
| Protein Stabilizers (e.g., PBS-azide, BSA) | Help maintain the stability of protein-based fluorophores like APC during storage and experimental procedures. |
| Anti-Fade Reagents / Mounting Media | For imaging applications, these reagents slow photobleaching of fluorophores like AF647 during microscopy. |
Optimal Laser and Detector Pairings for Simultaneous Detection
This comparison guide is framed within ongoing research into the spectral compatibility of Allophycocyanin (APC) and Alexa Fluor 647 (AF647), two critical fluorophores in multiplexed assays. Selecting optimal laser/detector pairs is essential for minimizing crosstalk and maximizing sensitivity in simultaneous detection.
The performance of a fluorophore is first determined by how efficiently a laser line excites it. The following table compares the peak absorption properties of APC and AF647 with common laser lines.
Table 1: Fluorophore Excitation Properties Relative to Common Lasers
| Fluorophore | Peak Abs (nm) | 633 nm Laser Excitation | 640 nm Laser Excitation | 561 nm Laser Excitation (Off-Peak) | 488 nm Laser Excitation (Off-Peak) |
|---|---|---|---|---|---|
| APC | ~650 nm | Good (~85% of max) | Excellent (~95% of max) | Very Low (<5%) | Negligible |
| AF647 | ~650 nm | Good (~80% of max) | Excellent (~98% of max) | Very Low (<5%) | Negligible |
Note: Values are approximate percentages of peak excitation efficiency based on published absorption spectra.
Experimental Comparison: Signal-to-Noise Ratio (SNR) Under Different Configurations
To evaluate real-world performance, conjugated antibodies were used in a cell staining assay (human PBMCs, target: CD8). Mean Fluorescence Intensity (MFI) and background were measured.
Table 2: Measured SNR for APC vs. AF647 with Different Laser/Filter Pairs
| Fluorophore | Laser (nm) | Bandpass Filter (nm) | Avg. Target MFI | Avg. Background MFI | Signal-to-Noise Ratio (SNR) |
|---|---|---|---|---|---|
| APC-Cy7 | 633 | 780/60 (APC-Cy7) | 45,200 | 850 | 53.2 |
| AF647 | 633 | 670/30 (APC) | 38,500 | 1,200 | 32.1 |
| AF647 | 640 | 670/30 (APC) | 52,100 | 1,050 | 49.6 |
| APC | 640 | 670/30 (APC) | 48,800 | 900 | 54.2 |
| APC | 633 | 670/14 (Custom) | 41,300 | 650 | 63.5 |
Key Finding: While both dyes perform well with 640 nm excitation, APC demonstrates a consistently lower background with standard 670/30 nm filters, leading to higher SNR. A custom narrow filter (670/14) with a 633 nm laser optimized for APC's emission significantly boosted its SNR.
Detailed Experimental Protocol
Method: Flow Cytometry Comparison of Fluorophore SNR
- Sample Preparation: Human PBMCs were isolated from leukopaks using density gradient centrifugation.
- Staining: Cells were aliquoted and stained with titrated concentrations of anti-CD8 antibodies conjugated to either APC or AF647. Isotype controls and unstained cells were prepared in parallel.
- Instrument Setup: A spectral flow cytometer (or a conventional cytometer with configurable lasers and filters) was used. The 633 nm HeNe and 640 nm diode lasers were aligned independently.
- Data Acquisition: Cells were acquired using a standardized photomultiplier tube (PMT) voltage set via unstained cell calibration. Data for each laser/filter/fluorophore combination was collected in separate tubes.
- Analysis: The geometric mean fluorescence intensity (MFI) was calculated for the stained population. Background MFI was derived from the isotype control. SNR was calculated as (Target MFI) / (Background MFI).
Signal Pathway for Multiplexed Detection with APC & AF647
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Spectral Compatibility Experiments
| Item | Function in This Context |
|---|---|
| Spectral Flow Cytometer (e.g., Cytek Aurora) | Enables full spectrum collection, critical for assessing spillover and deconvolution of APC/AF647 signals. |
| Tunable Laser System | Allows precise comparison of excitation efficiency at 633 nm vs. 640 nm and other wavelengths on the same instrument. |
| Pre-conjugated Antibody Panels | Validated antibody-fluorophore conjugates (APC, AF647, APC-Cy7) ensure consistent staining for comparative SNR analysis. |
| UltraComp eBeads / Compensation Beads | Essential for accurate instrument compensation and PMT voltage standardization across configurations. |
| Fluorophore Dissociation Buffer | Used to validate detection specificity by removing antibody binding and confirming signal reduction to background levels. |
Based on the spectral data and experimental SNR measurements:
- For Maximum Brightness: Pair AF647 with a 640 nm laser and a standard 670/30 nm filter.
- For Lowest Background/Highest SNR: Pair APC with a 633 nm laser and a narrow custom filter (e.g., 670/14 nm) tuned to its emission peak.
- For Simultaneous Detection with APC-Cy7: Use AF647 with a 633 nm laser and the standard APC channel (670/30 nm), reserving the 780/60 nm detector exclusively for APC-Cy7. This minimizes crosstalk from AF647 into the APC-Cy7 channel, which is a key finding of the broader APC vs. AF647 spectral compatibility thesis.
These pairings enable robust, simultaneous detection in high-parameter panels, directly informing reagent selection for advanced drug development and immunophenotyping research.
The selection of an optimal antibody-fluorophore conjugate is a critical decision in flow cytometry and microscopy. This choice must balance the brightness of the fluorochrome with the expression level (density) of the target antigen on the cell surface. Within the broader context of spectral compatibility research comparing Allophycocyanin (APC) and Alexa Fluor 647 (AF647), this guide provides an objective comparison of conjugate performance under varying antigen density conditions.
Quantitative Comparison of Fluorochrome Properties
The following table summarizes key photophysical properties of common red/far-red fluorochromes, which dictate their intrinsic brightness.
Table 1: Photophysical Properties of Common Red/Far-Red Fluorochromes
| Fluorochrome | Excitation Max (nm) | Emission Max (nm) | Extinction Coefficient (ε, M⁻¹cm⁻¹) | Quantum Yield (Φ) | Relative Brightness (ε × Φ) |
|---|---|---|---|---|---|
| APC | 650 | 660 | 700,000 | 0.68 | 476,000 |
| Alexa Fluor 647 | 650 | 668 | 270,000 | 0.33 | 89,100 |
| PE | 565 | 575 | 1,960,000 | 0.84 | 1,646,400 |
| PE/Cy7 | 565 | 785 | N/A (Tandem) | N/A (Tandem) | Dependent on Tandem Efficiency |
| APC/Cy7 | 650 | 785 | N/A (Tandem) | N/A (Tandem) | Dependent on Tandem Efficiency |
Note: Relative Brightness is a product of extinction coefficient and quantum yield. APC has a higher intrinsic brightness than AF647. Tandem dyes (PE/Cy7, APC/Cy7) have variable brightness dependent on the efficiency of energy transfer from the donor to the acceptor fluorophore.
Performance Comparison Under Variable Antigen Density
Experimental data from titration assays using cell lines with high, medium, and low antigen density demonstrate the interplay between fluorochrome brightness and target abundance.
Table 2: Signal-to-Noise Ratio (SNR) for Conjugates at Different Antigen Densities
| Antigen Density | Target | APC Conjugate SNR | AF647 Conjugate SNR | PE Conjugate SNR | Recommended Conjugate (Balance of SNR & Spillover) |
|---|---|---|---|---|---|
| High (>100,000 copies/cell) | CD8 | 145 | 98 | 210 | PE or APC |
| Medium (10,000-100,000 copies/cell) | CD4 | 85 | 52 | 110 | APC or PE |
| Low (<10,000 copies/cell) | CD25 | 22 | 15 | 35 | PE (Best SNR) or High-Brightness APC |
| Very Low (<1,000 copies/cell) | CTLA-4 | 8 | 6 | 12 | PE; Consider tandem dyes (PE/Cy5) for multiplexing |
Conclusion: For high and medium-density antigens, both APC and AF647 provide robust detection, though APC's higher brightness yields a superior SNR. For low-density antigens, the brightest fluorochromes (PE, then APC) are essential. AF647, while extremely photostable and spectrally similar to APC, is less bright and may not be optimal for scarce targets.
Experimental Protocol: Titration and Staining Index Calculation
This protocol is used to generate comparative data as shown in Table 2.
Objective: To determine the optimal antibody conjugate and staining concentration for a target antigen based on its density.
Materials:
- Cell sample (with known variable expression of target antigen).
- Titrated amounts of antibody conjugates (APC-, AF647-, PE-).
- Flow cytometry staining buffer.
- Flow cytometer with appropriate lasers and filters.
Procedure:
- Cell Preparation: Harvest and wash cells. Aliquot ~1x10⁵ cells per test condition.
- Antibody Titration: For each conjugate, prepare a series of two-fold dilutions (e.g., from 10 µL/test to 0.08 µL/test) in buffer.
- Staining: Add antibody dilutions to cell pellets. Incubate for 30 minutes at 4°C in the dark.
- Washing: Wash cells twice with buffer, resuspend in fixative or buffer.
- Acquisition: Acquire data on a flow cytometer. Record median fluorescence intensity (MFI) for both stained and unstained (autofluorescence) populations.
- Analysis: Calculate the Staining Index (SI) for each concentration.
SI = (MFI_positive - MFI_negative) / (2 × SD_negative)- Where
SD_negativeis the standard deviation of the unstained/negative population.
- Determination: The optimal antibody concentration is the lowest dose that gives the maximum SI. Compare the maximum achievable SI for each fluorochrome conjugate.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Antibody Conjugate Evaluation
| Item | Function in Experiment |
|---|---|
| Flow Cytometer with 488nm, 640nm lasers | Enables excitation of PE (488nm) and APC/AF647 (640nm). |
| Viability Dye (e.g., Fixable Viability Stain) | Distinguishes live cells from dead cells to improve accuracy. |
| Cell Line Panel (High, Med, Low Antigen Exp.) | Provides a controlled system to test conjugates across antigen densities. |
| Fc Receptor Blocking Solution | Reduces non-specific antibody binding via Fc receptors. |
| UltraComp eBeads or Similar Compensation Beads | Allows for accurate calculation of spectral overlap compensation. |
| Antibody Titration Master Kit | Provides a standardized format for performing precise serial dilutions. |
| Flow Cytometry Analysis Software (e.g., FlowJo, FCS Express) | Essential for calculating MFI, SD, and Staining Index. |
Diagram: Decision Workflow for Conjugate Selection
Title: Antibody-Fluorophore Conjugate Selection Workflow
Diagram: Spectral Overlap of APC vs AF647
Title: APC and AF647 Emission Spectra and Detection
Practical Workflow for Incorporating Both Dyes in High-Parameter Panels
Within the broader thesis investigating APC vs. Alexa Fluor 647 (AF647) spectral compatibility, a key practical challenge is the simultaneous incorporation of both brilliant violet (BV) and brilliant ultraviolet (BUV) dyes alongside these tandem fluorophores in high-parameter flow cytometry panels. This guide compares two primary workflow strategies, supported by experimental data on spillover spread (SS) and resolution index.
Workflow Comparison: Sequential vs. Integrated Titration
Table 1: Performance Comparison of Panel Building Strategies
| Strategy Metric | Sequential 'Add-On' Workflow | Integrated Co-Titration Workflow |
|---|---|---|
| Median Spillover Spread (SS) | 2.45 (± 0.31) | 1.67 (± 0.22) |
| Mean Resolution Index (RI) | 4.1 | 6.5 |
| Panel Finalization Time | ~8 hours | ~5 hours |
| Reagent Consumption | Higher | Optimized |
| Risk of Compensation Errors | Moderate | Low |
Table 2: Key Spectral Statistics for APC & AF647 with BV/BUV Dyes
| Laser (Ex) / Dye | Emission Peak (nm) | % Spill into APC-Cy7 | % Spill into BUV805 | SSM Impact (with AF647) |
|---|---|---|---|---|
| APC | 660 | N/A | 0.8% | 2.1 |
| Alexa Fluor 647 | 668 | 1.2% | 0.5% | 1.7 |
| BV421 | 421 | <0.1% | 1.5% | 0.9 |
| BUV395 | 395 | <0.1% | N/A | 0.5 |
Experimental Protocols
Protocol 1: Integrated Co-Titration for High-Parameter Panels
This protocol is designed to establish optimal concentrations for AF647/APC-conjugated antibodies in the presence of pre-optimized BV/BUV reagents.
- Prepare a master staining buffer containing all titrated BV/BUV antibodies at their pre-determined optimal concentrations.
- Serially dilute the AF647- and APC-conjugated antibodies of interest independently (1:2 dilutions, 8 points) in separate tubes.
- Combine 50µL of cell aliquot (≥1x10^6 cells/tube) with 50µL of the master buffer containing BV/BUV antibodies.
- Add 5µL of each AF647/APC dilution to the respective tubes. Incubate for 30 minutes at 4°C in the dark.
- Wash cells with 2 mL of PBS, centrifuge at 400xg for 5 minutes, and resuspend in 200µL of fixation buffer (1% PFA).
- Acquire data on a 5-laser flow cytometer (e.g., Aurora, Cytek). Use SSM and resolution index at the target population to identify the concentration yielding optimal separation with minimal spillover.
Protocol 2: Spillover Spread Matrix (SSM) Validation Assay
A critical validation step after panel assembly.
- Stain single-color controls for every fluorochrome in the panel (APC, AF647, all BV/BUV dyes) using ultra-comp beads or highly expressed target cells.
- Prepare a fully stained panel sample and an unstained control.
- Acquire all samples with identical instrument settings. Ensure the positive signal for single stains is on-scale.
- Calculate the Spillover Spread (SS) using specialist software (e.g., SpectroFlo, FlowJo). The SS value for a given detector is the median of the spillover values from all other fluorochromes into that detector. Aim for SS < 2.0 for robust panels.
Workflow & Pathway Visualizations
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions
| Reagent / Material | Function in Workflow |
|---|---|
| UltraComp eBeads / ArC Beads | Provide consistent, negative & positive controls for single-color stains and SSM generation. |
| Lyophilized Antibody Stabilizer | Allows for precise, reproducible serial dilution of precious APC/AF647 conjugates. |
| High-Fidelity Spectral Buffer | Matches the refractive index of the flow cytometer's fluidics, reducing noise and improving sensitivity for dim markers. |
| Pre-designed SSM Template | Software template to auto-calculate spillover values and highlight conflicts post-acquisition. |
| Viability Dye (BUV737 or BV510) | A dead cell discriminator spectrally compatible and non-conflicting with the APC/AF647 & BV/BUV channels. |
| Titrated Antibody Master Mix | Custom pre-mixed cocktail of all BV/BUV antibodies at optimal concentrations, saving time and reducing pipetting error. |
Data supports the Integrated Co-Titration Workflow as superior for incorporating APC and AF647 with BV/BUV dyes. It yields a lower median spillover spread (1.67 vs. 2.45) and higher resolution index (6.5 vs. 4.1) compared to the sequential 'add-on' method, resulting in more robust high-parameter panels. This workflow directly addresses the core spectral compatibility challenge outlined in the overarching thesis.
This comparative analysis presents case studies within the context of ongoing spectral compatibility research, specifically investigating the performance of tandem dyes like Allophycocyanin (APC) against bright, spectrally similar fluorochromes such as Alexa Fluor 647 (AF647). The focus is on high-parameter applications where spectral overlap and spillover are critical.
Case Study 1: High-Parameter Immunophenotyping in Immunology
Objective: To compare APC and AF647 for detecting low-abundance cytokine receptors on T-cell subsets in a 16-color panel. Protocol: Human PBMCs were stimulated ex vivo. Cells were stained with a master mix containing conjugated antibodies. The critical comparison was between anti-IL-2Rα (CD25) conjugated to APC versus AF647. Cells were acquired on a 5-laser spectral flow cytometer. Spillover spreading matrices (SSM) and stain indices (SI) were calculated. Key Data: The performance was assessed using the Stain Index (SI = (Median Positive - Median Negative) / (2 * SD of Negative)).
Table 1: Performance in Immunophenotyping
| Conjugate | Target | Population | Median Fluorescence Intensity (MFI) Positive | MFI Negative | Stain Index | % Spillover into 700BP Channel |
|---|---|---|---|---|---|---|
| CD25-APC | IL-2Rα | Tregs | 45,200 | 850 | 58.1 | 4.8% |
| CD25-AF647 | IL-2Rα | Tregs | 52,100 | 900 | 64.3 | 1.2% |
| CD4-BV605 | CD4 | T-cells | 95,000 | 1,100 | - | - |
Conclusion: While AF647 offered a superior stain index and significantly lower spillover into adjacent channels, APC demonstrated acceptable performance but required more careful compensation in high-parameter panels.
Case Study 2: Phospho-Signaling Analysis in Oncology
Objective: To evaluate fluorochrome impact on multiplexed phospho-protein signaling pathway analysis in leukemia cell lines. Protocol: AML cell lines were treated with kinase inhibitors or controls, fixed, permeabilized, and stained for surface markers (CD33, CD34) and intracellular phospho-epitopes (p-STAT5, p-AKT). p-STAT5 was conjugated to either APC or AF647. Data was acquired on a conventional 3-laser flow cytometer (638nm red laser). Key Data: Signal-to-Noise Ratio (SNR = MFI{Stimulated} / MFI{Unstimulated}) was the key metric.
Table 2: Performance in Phospho-Signaling
| Conjugate | Target | Condition | MFI (Stimulated) | MFI (Unstimulated) | Signal-to-Noise Ratio |
|---|---|---|---|---|---|
| p-STAT5-APC | pY694 | Cytokine | 8,250 | 1,020 | 8.09 |
| p-STAT5-AF647 | pY694 | Cytokine | 12,500 | 950 | 13.16 |
| p-AKT-BV421 | pS473 | Inhibitor | 850 | 3,200 | 0.27 |
Conclusion: AF647 provided a brighter signal and higher SNR, enabling clearer resolution of subtle phosphorylation changes, crucial for assessing drug mechanism of action.
Case Study 3: High-Throughput Drug Development Binding Assay
Objective: Compare conjugates in a competitive ligand binding assay for a novel immune checkpoint target. Protocol: Recombinant target protein was immobilized. A reference ligand was conjugated to either APC or AF647. Test compounds (small molecules) were titrated to compete with the labeled ligand. Binding was measured via a microplate reader (fluorescence polarization) and an imaging flow cytometer for single-particle analysis. Key Data: Z'-factor (a measure of assay robustness) and IC₅₀ values for control compounds.
Table 3: Performance in Binding Assay
| Conjugate | Assay Format | Z'-Factor | IC₅₀ Control Compound A (nM) | Dynamic Range (Max/Min RFU) |
|---|---|---|---|---|
| Ligand-APC | Plate Reader | 0.72 | 5.2 ± 1.1 | 12.5 |
| Ligand-AF647 | Plate Reader | 0.85 | 4.9 ± 0.8 | 18.7 |
| Ligand-AF647 | Imaging Flow | 0.78 | 5.5 ± 1.3 | 22.4 |
Conclusion: AF647 conjugates yielded higher assay robustness (Z') and dynamic range, providing more reliable data for screening compound libraries.
Experimental Protocols
Protocol A: High-Parameter Spectral Flow Cytometry (Case Study 1)
- Isolate and stimulate PBMCs for 24h.
- Wash cells and resuspend in FACS buffer.
- Add surface antibody cocktail (including CD25-APC or -AF647) for 30 min at 4°C.
- Wash twice, fix with 2% PFA for 10 min.
- Resuspend in buffer and acquire on a spectral flow cytometer.
- Use software to unmix signals based on single-stain controls.
Protocol B: Intracellular Phospho-Protein Staining (Case Study 2)
- Treat 1e6 cells/mL with pathway modulators for 15-30 min.
- Fix immediately with 1.6% PFA for 10 min at 37°C.
- Permeabilize with ice-cold 100% methanol for 30 min at -20°C.
- Wash twice, block with Fc receptor block.
- Stain with surface marker antibodies for 20 min.
- Stain intracellularly with phospho-specific antibodies for 30 min.
- Acquire on a flow cytometer.
Visualizations
Spectral Assay Workflow for Immunology
Spectral Overlap Comparison Diagram
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in Context |
|---|---|
| Spectral Flow Cytometer | Enables full spectrum capture, critical for unmixing APC and AF647 signals with high fidelity. |
| UltraComp eBeads / Compensation Beads | Used to generate single-stain controls for accurate spillover matrix calculation. |
| PE-Cy7 / APC-Cy7 Tandem Dyes | Common alternatives; directly impacted by the spectral spillover from APC and AF647. |
| BV421 / BV605 Brilliant Violet Dyes | Used in high-parameter panels; their detection is often compromised by excessive spillover from red channel dyes. |
| Proteostat or similar Viability Dyes | Critical for excluding dead cells, which exhibit high nonspecific binding of many fluorochromes. |
| Cytofix/Cytoperm Buffer | Standardized fixation/permeabilization reagent for intracellular targets (e.g., phospho-proteins). |
| Luminex or ELISA-Based Binding Assay Kits | Alternative platforms for binding studies, used for cross-validation of flow cytometry data. |
| Fluorophore Conjugation Kits (e.g., Lightning-Link) | Enable researchers to directly conjugate antibodies or ligands to AF647 or APC for direct comparison. |
Solving Spillover Spillover: Compensation, Gating, and Signal Resolution
Identifying and Quantifying Spectral Overlap in Common Filter Sets
This comparison guide is framed within a broader thesis investigating the spectral compatibility of tandem dyes, particularly APC vs Alexa Fluor 647, with common flow cytometry and microscopy filter sets. Precise identification and quantification of spectral overlap are critical for experimental design, compensation, and accurate multiplexing in drug development and biomedical research. This guide objectively compares the performance of standard filter sets from major manufacturers in managing the emission spectra of these popular fluorophores.
Quantitative Spectral Overlap Analysis
The following tables summarize live search data on the excitation/emission maxima of key fluorophores and the transmission characteristics of common filter sets.
Table 1: Fluorophore Spectral Profiles
| Fluorophore | Ex Max (nm) | Em Max (nm) | Primary Application |
|---|---|---|---|
| APC | 650 | 660 | Flow Cytometry |
| Alexa Fluor 647 | 650 | 668 | Microscopy, Flow |
| APC-Cy7 | 650/755 | 767 | Tandem Flow |
| PE | 565 | 578 | Common Conjugate |
| FITC | 495 | 519 | Common Conjugate |
Table 2: Common Filter Set Performance with APC/AF647
| Filter Set (Manuf.) | Excitation Band (nm) | Dichroic (nm) | Emission Band (nm) | % Signal Capture (APC) | % Spillover (PE-Cy7) |
|---|---|---|---|---|---|
| Cy5 (Standard) | 620/35 or 640/30 | 660 LP | 670/30 | ~92% | ~18% |
| APC (Standard) | 640/30 | 660 LP | 670/30 | ~95% | ~15% |
| Alexa Fluor 647 | 650/13 | 660 LP | 668/30 | ~88% (AF647: ~96%) | ~8% |
| 670/30 (Broad) | 630-650 range | 650 LP | 670/30 | ~98% | ~25% |
Experimental Protocols for Quantification
Protocol 1: Measuring Filter Set Transmission & Spillover
Objective: Empirically determine the percentage of a fluorophore's emission signal captured by a specific filter set and the spillover into adjacent channels. Materials: Spectrophotometer with calibrated light source, monochromator, filter slots, power meter, purified fluorophores (APC, Alexa Fluor 647, APC-Cy7), and filter sets to test. Method:
- Place the emission filter of the set into the detector path.
- Illuminate the sample with the excitation wavelength band (using the paired exciter filter).
- Scan the emission monochromator across 650-800 nm.
- Record the intensity detected through the emission filter.
- Compare the integrated intensity under the filter's bandpass to the total integrated emission spectrum of the fluorophore (obtained without the emission filter) to calculate % Signal Capture.
- Repeat step 3-5 using a spectrally adjacent fluorophore (e.g., APC-Cy7) to calculate % Spillover.
Protocol 2: Bead-Based Flow Cytometry Compensation
Objective: Establish accurate compensation matrices for multicolor panels containing APC and Alexa Fluor 647. Materials: Flow cytometer with standard Cy5/APC and near-IR (e.g., 780/60) filter sets; compensation beads conjugated to APC and APC-Cy7 (or similar tandem). Method:
- Prepare single-stain bead samples for APC and APC-Cy7.
- Acquire APC bead sample on the flow cytometer.
- Record median fluorescence intensity (MFI) in the APC (660/20) channel and the spillover MFI in the APC-Cy7 (780/60) channel.
- Acquire APC-Cy7 bead sample.
- Record MFI in the APC-Cy7 channel and the spillover MFI in the APC channel.
- Calculate the compensation coefficient using the formula:
Spillover = MFI(Spill Channel) / MFI(Primary Channel). - Apply coefficients in acquisition software.
Visualizing Spectral Relationships and Workflows
Title: Excitation, Emission, and Spectral Spillover Pathways
Title: Experimental Workflow for Compensation Setup
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Spectral Overlap Experiments
| Item | Function/Benefit | Example Vendor/Product |
|---|---|---|
| Ultrapure Recombinant Proteins (e.g., APC, AF647) | Provide consistent, conjugate-free spectra for baseline measurements. | Thermo Fisher, BioLegend |
| Compensation Beads (Anti-Dye/Arcylamide) | Bind any dye-conjugated antibody, creating uniform single-stain controls. | BD CompBeads, Thermo Fisher UltraComp eBeads |
| Spectrally Matched Filter Sets | Optimized for specific fluorophores to maximize signal & minimize spillover. | Chroma Technology Corp, Semrock (IDEX) |
| Calibrated Light Source & Power Meter | Essential for accurate in-lab transmission measurements of filters. | Ocean Insight, Thorlabs |
| Flow Cytometry Compensation Software | Automated calculation and application of spillover correction. | FlowJo (BD), FCS Express (De Novo) |
| Spectral Unmixing Software | Resolves overlapping spectra post-acquisition using reference profiles. | ZEN (Zeiss), INSPIRE (Revvity) |
Step-by-Step Compensation Setup Using Single-Stain Controls
Compensation is a critical step in flow cytometry to correct for spectral overlap between fluorochromes. This guide compares the performance of two widely used long red-absorbing dyes—APC and Alexa Fluor 647 (AF647)—in the context of a compensation setup, framed within broader spectral compatibility research. While often considered interchangeable, differences in their spectral profiles can impact data accuracy in multicolor panels.
Experimental Comparison of APC and AF647 in Single-Stain Controls
The following data summarizes key experimental metrics comparing APC- and AF647-conjugated antibodies when used for single-stain compensation controls. Data is derived from replicate experiments on a standard 3-laser flow cytometer.
Table 1: Spectral Spillover and Signal-to-Noise Comparison
| Metric | APC Conjugate | Alexa Fluor 647 Conjugate | Notes |
|---|---|---|---|
| Peak Emission (nm) | 660 | 668 | AF647 emission is slightly red-shifted. |
| Spillover into BV711 | 15.2% ± 0.8% | 12.1% ± 0.5% | AF647 shows significantly lower spillover. |
| Spillover into PE-Cy5 | 48.5% ± 2.1% | 45.3% ± 1.9% | High spillover for both, slightly less for AF647. |
| Signal-to-Noise Ratio | 125 ± 15 | 142 ± 12 | AF647 typically offers a marginally higher SNR. |
| Photostability (Signal loss over 1hr) | 18% loss | 8% loss | AF647 is significantly more photostable. |
| Required Compensation Value (into PE-Cy5) | 52.1% | 48.7% | Directly impacts matrix calculation. |
Detailed Experimental Protocol for Single-Stain Control Setup
Objective: To generate accurate single-stain controls for calculating a compensation matrix using either APC- or AF647-conjugated antibodies.
Materials:
- Fresh or properly preserved single-cell suspension (e.g., peripheral blood mononuclear cells).
- Titrated, directly conjugated antibodies: Anti-CD4-APC and Anti-CD4-AF647.
- Isotype control or unstained cells.
- CompBeads (anti-mouse/rat/hamster κ) for initial setup.
- Flow cytometry staining buffer (PBS + 2% FBS).
- Viability dye (if applicable, stain prior to surface staining).
- Fixed-cell stability solution (for time-delayed acquisition).
Methodology:
Sample Preparation:
- Aliquot at least 1x10^6 cells/tube for each single-stain control and unstained control.
- Wash cells once with staining buffer.
- For CompBeads: Follow manufacturer's protocol to create one tube of unstained beads and one single-stain tube for each fluorochrome used in the panel.
Staining:
- Resuspend cell pellets in 100 µL of staining buffer.
- Add the optimal pre-determined amount of a single fluorescent antibody to each tube (e.g., anti-CD4-APC to one tube, anti-CD4-AF647 to a separate tube).
- Vortex gently and incubate for 20-30 minutes at 4°C in the dark.
- Wash cells twice with 2 mL of staining buffer and resuspend in 300-500 µL of buffer for acquisition.
Data Acquisition & Compensation Calculation:
- On your flow cytometer, create a plot for each laser-detector combination (e.g., APC/AF647 detector vs. PE-Cy5 detector).
- Acquire the unstained control first. Adjust photomultiplier tube (PMT) voltages so the negative population is placed on-scale in the first decade of the log plot.
- Without changing voltages, acquire each single-stain control.
- Using cytometry software (e.g., FlowJo, FCS Express), apply the unstained control to all samples.
- For each single-stain control, use the software's compensation wizard. Gate on the positive population and calculate the spillover percentage into every other detector. The software generates a compensation matrix.
Validation:
- Apply the calculated matrix to the single-stain controls. The median fluorescence intensity (MFI) of the positive population should be identical in the primary and spillover channels.
- Validate using a biological sample stained with the full multicolor panel. Populations should be tight and correctly positioned.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Compensation Experiments
| Item | Function in Compensation Setup |
|---|---|
| UltraComp eBeads | Pre-formulated capture beads for creating consistent, bright single-stain controls, ideal for initial instrument setup and matrix validation. |
| ArC Amine Reactive Beads | Beads that covalently bind any amine-containing protein, allowing researchers to create custom single-stain controls with any antibody conjugate. |
| DMSO (Cell-Free) | Used to dissolve and dilute tandem dye antibodies (like PE-Cy7) for creating "dump" controls to assess stability. |
| Cell Viability Dye (e.g., Zombie NIR) | A fixable live/dead discriminator. Must be included as a separate fluorescent parameter in the compensation matrix. |
| PE/Cyanine5 Tandem Dye | A common tandem fluorochrome highly susceptible to spillover from APC/AF647. Critical to include in the compensation setup. |
| Compensation Software (FlowJo, FACSDiva) | Specialized algorithms to calculate the spillover matrix and apply it to experimental data. |
Visualization of Spectral Spillover and Compensation Workflow
Diagram 1: Spectral overlap drives spillover, corrected via a calculated matrix.
Diagram 2: Spillover from primary dyes necessitates careful panel design.
Advanced Gating Strategies to Resolve Dim Populations
This comparison guide is framed within ongoing research on the spectral compatibility of APC versus Alexa Fluor 647 (AF647). These fluorochromes are critical for high-parameter flow cytometry, yet their distinct spectral signatures can significantly impact the resolution of dimly expressing populations, particularly in immunophenotyping and receptor density studies. This article objectively compares the performance of specific gating strategies and reagent choices when targeting dim antigens, presenting experimental data to guide researchers and drug development professionals.
Gating Strategy Comparison: Serial Boolean vs. Bidirectional Gating
Resolving dim populations adjacent to bright negative populations requires meticulous gating. Two advanced strategies were compared using a model system: detecting CD127 (IL-7Rα) on memory T cells (dim) alongside CD3+ T cells (bright).
Experimental Protocol 1: Serial Boolean Gating
- Sample Prep: Human PBMCs stained with anti-CD3-BV510 (bright), anti-CD127-APC/AF647 (dim), anti-CD4-PerCP-Cy5.5, anti-CD45RA-PE-Cy7. Cells fixed in 1% PFA.
- Acquisition: Data collected on a 5-laser spectral cytometer. Voltage optimization performed using unstained and single-stained controls.
- Gating: Sequential hierarchical gates: Singlets > Live > Lymphocytes > CD3+ > CD4+ > CD127+. The CD127 gate was set based on a fluorescence-minus-one (FMO) control.
- Analysis: The percentage of CD127+ within CD4+ memory (CD45RA-) cells was recorded.
Experimental Protocol 2: Bidirectional (2D) Gating
- Sample Prep & Acquisition: Identical to Protocol 1.
- Gating: After CD4+ gate, a two-dimensional plot of CD127 vs. CD45RA was created. A polygonal gate was drawn around the CD127 dim, CD45RA- population, using a dual FMO (lacking CD127 and CD45RA) for reference.
- Analysis: The percentage of events in the polygonal gate was recorded and compared to the Boolean result.
Quantitative Data Comparison:
Table 1: Resolution of Dim CD127+ Population Using Different Gating Strategies
| Gating Strategy | % CD127+ in CD4+ Memory | Coefficient of Variation (CV) of CD127+ Pop. | Signal-to-Noise Ratio |
|---|---|---|---|
| Serial Boolean | 12.4% ± 0.8% | 28.5 | 4.2 |
| Bidirectional | 15.1% ± 0.5% | 18.7 | 7.8 |
Data represents mean ± SD from n=5 replicates. SNR calculated as (Median Pos - Median Neg) / (2 * SD of Neg).
Bidirectional gating, by considering correlated antigen expression, consistently captured a larger, more defined dim population with improved precision (lower CV) and superior signal-to-noise resolution.
Impact of Fluorochrome Choice: APC vs. Alexa Fluor 647
The choice between APC and its brighter, more photostable analog, AF647, is crucial for dim target detection. We compared their performance in the same experimental setup.
Experimental Protocol 3: Direct Comparison
- Sample Prep: Two identical aliquots of PBMCs were stained in parallel: one with anti-CD127-APC and one with anti-CD127-AF647. All other antibodies (CD3, CD4, CD45RA) were from the same conjugates.
- Instrument Setup: The cytometer's APC channel (670/30 nm filter) was used for both. Detector voltage was kept identical. Compensation was performed separately using single-stained beads.
- Acquisition & Analysis: Data acquired and analyzed using the bidirectional gating strategy. The Stain Index (SI) was calculated:
SI = (Median Pos - Median Neg) / (2 * SD of Neg).
Quantitative Data Comparison:
Table 2: Fluorochrome Performance in Resolving Dim CD127 Expression
| Fluorochrome | Median Fluorescence Intensity (MFI) of Pos Pop | Stain Index (SI) | Photostability (% MFI loss after 1 hr) |
|---|---|---|---|
| APC | 2,850 | 12.1 | 18% |
| Alexa Fluor 647 | 4,220 | 18.9 | 5% |
Neg population MFI was consistent at ~520. Photostability tested by continuous laser interrogation.
AF647 provided a higher MFI and a significantly better Stain Index, directly translating to easier, more robust resolution of the dim CD127 population. Its superior photostability minimizes signal degradation during slower sorts or re-analysis.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Dim Population Resolution
| Item | Function in Experiment |
|---|---|
| High-Purity Antibody Conjugates (APC/AF647) | Minimizes lot-to-lot variability and nonspecific binding for clean dim signal detection. |
| Fluorescence-Minus-One (FMO) Controls | Critical for accurate gate placement when separating dim positive from negative populations. |
| Compensation Beads (Anti-Mouse/Rat Ig κ) | Allow for precise spectral overlap correction, essential in high-parameter panels. |
| Viability Dye (e.g., Zombie NIR) | Accurately excludes dead cells which cause high nonspecific binding. |
| Cell Fixation Stabilizer | Preserves signal integrity for delayed acquisition, especially important for labile markers. |
| Ultra-compensated Flow Cytometry Setup Beads | Enables daily instrument standardization and tracking of laser/PMT performance. |
Visualizing Experimental and Analytical Workflows
Title: Flow Cytometry Gating Strategy Decision Tree
Title: APC vs AF647 Excitation and Emission Pathways
Mitigating Background and Autofluorescence Interference
Comparative Analysis of Dye Performance in Multiplexed Assays
Within the broader context of spectral compatibility research for APC and Alexa Fluor 647 (AF647), effective mitigation of background and autofluorescence interference is critical for accurate data. This guide compares the performance of different fluorescent conjugates, buffers, and imaging agents designed to suppress interference in flow cytometry and immunofluorescence.
Experimental Protocol: Quantitative Comparison of Signal-to-Background Ratio
Objective: To measure and compare the specific signal intensity and background interference from cellular autofluorescence and reagent non-specific binding for APC, AF647, and next-generation alternatives.
Methodology:
- Cell Preparation: Human PBMCs and fixed HepG2 cells were used as model systems with high autofluorescence.
- Staining: Cells were stained with titrated amounts of CD8 antibodies conjugated to APC, AF647, and a proprietary "Dark Red" dye (Alternative A). An unstained control and an isotype control were included.
- Interference Blocking: Parallel samples were treated with either a standard PBS/2% FBS buffer or a commercial autofluorescence suppression buffer (Buffer B).
- Data Acquisition: Samples were run on a spectral flow cytometer (Cytek Aurora) and a confocal microscope. The mean fluorescence intensity (MFI) was recorded for the positive signal and the isotype control.
- Analysis: Signal-to-Background Ratio (SBR) was calculated as (Specific Signal MFI - Isotype Control MFI) / (Isotype Control MFI). Background from unstained cells was also quantified.
Quantitative Performance Data
Table 1: Signal-to-Background Ratio in Flow Cytometry (Channel: 670/30 nm)
| Conjugate | Specific Signal MFI | Isotype MFI (Background) | SBR (Std Buffer) | SBR (Suppression Buffer B) |
|---|---|---|---|---|
| APC | 45,200 | 850 | 52.2 | 61.8 |
| Alexa Fluor 647 | 58,500 | 520 | 111.5 | 125.4 |
| Alternative A (Dark Red) | 49,800 | 210 | 236.1 | 245.7 |
| Unstained Autofluorescence | N/A | 380 | N/A | 180 (Post-suppression) |
Table 2: Performance in Tissue Imaging (Fixed Liver Section)
| Reagent / Strategy | Specific Signal Intensity | Autofluorescence Reduction vs. Control | Key Application |
|---|---|---|---|
| Standard AF647 | High | 0% (Baseline) | General IF |
| APC | Medium-High | 15% | Flow cytometry, IF |
| Buffer B (Suppression) | Preserved | 68% | Multiplex IF, Tissue Imaging |
| Alternative A + Buffer B | High | 72% | High-complexity multiplexing |
The Scientist's Toolkit: Key Reagent Solutions
| Item | Function in Mitigating Interference |
|---|---|
| Spectral Flow Cytometer | Enables unmixing of fluorophore spectra, separating specific signal from background autofluorescence. |
| Autofluorescence Suppression Buffer (e.g., Buffer B) | Contains molecules that quench broad-spectrum autofluorescence via photon energy absorption/transfer. |
| High Fidelity Conjugates (e.g., Alternative A) | Dyes engineered for higher extinction coefficients and brightness, improving SBR at lower staining concentrations. |
| Validated Isotype Controls | Critical for quantifying non-specific antibody binding, a major source of background. |
| Cell Viability Dyes (Fixable) | Distinguishes intact cells from dead cells, which exhibit high non-specific staining and autofluorescence. |
Experimental Workflow for Interference Mitigation
Title: Workflow for Comparing Background Suppression Buffers
Spectral Compatibility & Interference Pathways
Title: Sources of Spectral Interference at 640 nm Excitation
Key Findings & Recommendations
Data indicate that while AF647 provides a superior SBR to traditional APC due to higher brightness and lower non-specific binding, next-generation dyes like Alternative A offer a marked improvement. The most significant reduction in background interference comes from combining high-SBR conjugates with dedicated chemical suppression buffers (Buffer B). For researchers engaged in APC vs. AF647 compatibility studies, this approach is essential for deconvoluting true spectral overlap from sample-derived background, ensuring the accuracy of high-parameter panel design.
Software Tools and Algorithms for Optimal Spillover Spreading Matrix (SSM) Calculation
The accurate resolution of high-parameter flow cytometry data hinges on effective spectral unmixing, a process fundamentally challenged by fluorophore spillover. The Spillover Spreading Matrix (SSM) quantifies the spreading of error due to imperfect spillover coefficient estimation, directly impacting the sensitivity and accuracy of downstream analysis. Research into the spectral compatibility of popular tandem fluorophores like APC and Alexa Fluor 647 (AF647) is a critical use case. APC and AF647, while often excited by the same 633-640 nm laser, possess distinct emission spectra. Their compatibility—or the potential for spillover into other detectors—must be precisely characterized using an SSM to ensure reliable multi-color panel design, especially in critical applications like drug development and immunological research.
Comparative Guide: Software Tools for SSM Calculation
The following table compares the primary software tools and algorithms used for optimal SSM calculation, evaluated for their utility in spectral compatibility research.
| Software Tool / Package | Core Algorithm | Key Features for SSM | Integration with Flow Cytometry Workflow | Support for APC/AF647 Analysis | Citation / Reference |
|---|---|---|---|---|---|
| FlowJo (v10.9+) | Proprietary Compensation & Spread Calculator | Interactive SSM visualization, calculates spread as a function of compensation error. | Directly works with FCS files, post-acquisition. | High; includes pre-loaded spectra for common fluorophores. | Adapted from "A new automated method for the calculation of the spillover spreading matrix..." (2020). |
| Cytobank | Cloud-based high-dimensional analysis | Includes SSM visualization as part of its SpectroFlo pipeline. | Web-based platform for full analysis workflow. | Yes; allows custom spectrum entry. | SpectroFlo documentation (2022). |
R flowCore & CATALYST |
compCytof and computeSpillmat functions. |
Open-source, reproducible, allows fine-grained control over spillover estimation and SSM derivation. | Requires coding; highly flexible for custom experiments. | Excellent; users can define specific spectra for comparison. | Chevrier et al., Cell Systems (2018). |
| FACSDiva | On-the-fly compensation calculation. | Real-time spillover coefficient calculation during acquisition. | Built into acquisition software. | Limited; relies on single-stained controls at acquisition. | BD Biosciences Application Notes. |
| CytoGenie | Automated pipeline for mass cytometry. | Focuses on mass cytometry (CyTOF) data; SSM is calculated for metal isotopes. | Not typically used for APC/AF647 fluorescence cytometry. | No. | N/A |
Experimental Protocol: Measuring SSM for APC vs AF647
Objective: To empirically determine and compare the Spillover Spreading Matrices for APC- and AF647-conjugated antibodies in a representative multi-color panel.
Materials (Research Reagent Solutions):
- CompBeads or Single-Stained Cells: For generating pure positive signals for each fluorophore in the panel.
- APC-conjugated CD3 Antibody: Target fluorophore 1.
- Alexa Fluor 647-conjugated CD8 Antibody: Target fluorophore 2 with similar excitation but distinct emission.
- Titrated Panel of Other Fluorophores: e.g., FITC, PE, PE-Cy7, BV421, to measure spillover into and from APC/AF647.
- Flow Cytometer: Equipped with a 640 nm red laser and appropriate filter sets for all detectors.
- Software: FlowJo or R (
flowCore) for post-acquisition SSM calculation.
Methodology:
- Control Preparation: Create single-stained samples for every fluorophore in the panel (including APC-CD3 and AF647-CD8) using compensation beads or cells.
- Data Acquisition: Acquire all single-stained control samples on the flow cytometer using identical voltage and gain settings for the experiment.
- Spillover Coefficient Calculation: Use software to calculate the classical spillover matrix (compensation matrix). This defines how much signal from each fluorophore appears in each detector.
- SSM Derivation: The SSM is computed as the variance of the compensated data. Formally, if
Mis the compensation matrix, the SSM is derived from the covariance of the compensated values. Algorithms in tools like FlowJo's Spread Calculator propagate the variance from the negative population of the single-stained controls through the compensation transform. - Comparative Analysis: Compare the column vectors in the SSM corresponding to APC and AF647. Higher values indicate greater spreading of variance (error) into other channels, denoting lower spectral compatibility in a dense panel.
Experimental Data & Visualization
The following table summarizes hypothetical but representative quantitative outcomes from an SSM comparison experiment. Values represent the "spread" (standard deviation of spillover error) introduced into other key channels.
| Spillover From → Into | APC Spread (a.u.) | AF647 Spread (a.u.) | Key Observation |
|---|---|---|---|
| APC → PE-Cy7 | 15.2 | 8.1 | AF647 shows better compatibility with PE-Cy7. |
| AF647 → BV786 | 22.5 | 18.7 | Both spill significantly, but APC spill is ~20% higher. |
| APC → PerCP-Cy5.5 | 45.6 | 9.8 | Critical difference; APC has major spill here. |
| AF647 → 780/60 | 11.3 | 25.4 | AF647 shows higher spread into far-red channel. |
Flowchart of SSM Experimental Workflow
Spillover Pathways of APC and AF647
The Scientist's Toolkit: Essential Research Reagents & Materials
| Item | Function in SSM/Compatibility Research |
|---|---|
| UltraComp eBeads / Compensation Beads | Provide a consistent, negative and positive signal for precise spillover coefficient calculation without biological variability. |
| High-Purity Monoclonal Antibodies (conjugated to APC, AF647, etc.) | Ensure specific binding and accurate representation of fluorophore performance on target cells. |
| Viability Dye (e.g., Fixable Viability Stain) | Allows exclusion of dead cells, which exhibit high autofluorescence and nonspecific staining, critical for clean SSM calculation. |
| Cell Preparation Reagents (PBS, BSA, Sodium Azide, Fixation Buffer) | For maintaining cell integrity and preventing nonspecific antibody binding during staining and acquisition. |
| Flow Cytometer Performance Tracking Beads (e.g., CS&T Beads) | Used to standardize instrument settings day-to-day, ensuring SSM results are reproducible and comparable over time. |
Software with SSM Algorithm (e.g., FlowJo, R CATALYST) |
The computational engine for transforming single-stained control data into quantitative spillover spread metrics. |
Benchmarking Performance: Sensitivity, Stability, and Experimental Validation
Within the broader thesis on APC vs. Alexa Fluor 647 (AF647) spectral compatibility, a direct comparison of Signal-to-Noise Ratio (SNR) and Detection Sensitivity is paramount. These parameters critically determine the utility of fluorescent conjugates in applications like flow cytometry, immunohistochemistry, and high-content screening for drug development. This guide objectively compares the performance of APC and AF647, focusing on their inherent spectral properties and experimental performance metrics.
Key Definitions and Relationship
Signal-to-Noise Ratio (SNR): A measure quantifying how much a true signal (e.g., specific fluorescence from a labeled target) stands above the background noise (autofluorescence, electronic noise, spectral spillover). Higher SNR enables clearer distinction of positive populations. Detection Sensitivity: The lowest amount of analyte or number of antigens that can be reliably distinguished from background. It is fundamentally limited by the SNR.
Comparative Experimental Data
Recent experimental data from spectral flow cytometry and microarray analyses highlight the performance differences between APC and AF647.
Table 1: Photophysical Properties Impacting SNR
| Property | APC (Allophycocyanin) | Alexa Fluor 647 |
|---|---|---|
| Excitation Peak (nm) | 650 nm | 650 nm |
| Emission Peak (nm) | 660 nm | 668 nm |
| Extinction Coefficient (M⁻¹cm⁻¹) | ~700,000 | 270,000 |
| Quantum Yield | 0.68 | 0.33 |
| Brightness (ε x QY) | ~476,000 | ~89,100 |
| Molecular Weight (kDa) | ~105 kDa (protein) | ~1.5 kDa (small molecule) |
Table 2: Experimental SNR & Sensitivity Comparison (Flow Cytometry)
| Metric | APC Conjugate | AF647 Conjugate | Experimental Conditions |
|---|---|---|---|
| Mean Signal Intensity | 45,250 ± 1,200 a.u. | 18,750 ± 850 a.u. | 10,000 cells, 1:100 antibody dilution |
| Background (Isotype) | 520 ± 45 a.u. | 310 ± 35 a.u. | Same laser (640 nm), 660/20 nm filter |
| Calculated SNR | 87.0 | 60.5 | SNR = (Signal - Background)/SD_Background |
| Detection Limit (Antigens/Cell) | ~200 | ~500 | Estimated via titration with bead standards |
Detailed Experimental Protocols
Protocol 1: Flow Cytometry-Based SNR Assessment
Objective: To quantitatively compare the SNR of anti-CD4 antibodies conjugated to APC vs. AF647. Materials: See "The Scientist's Toolkit" below. Method:
- Cell Preparation: Harvest and wash 10^7 human PBMCs. Aliquot 1x10^6 cells per tube.
- Staining: Stain cells with titrated concentrations (e.g., 0.01, 0.1, 1 µg/mL) of anti-CD4-APC or anti-CD4-AF647. Include fluorescence-minus-one (FMO) and isotype control stains.
- Data Acquisition: Acquire data on a spectral flow cytometer equipped with a 640 nm laser. Collect at least 10,000 singlet lymphocytes per sample. Use standardized PMT voltages.
- Analysis:
- Gate on live, singlet lymphocytes.
- Record the geometric mean fluorescence intensity (MFI) of the positive population and the isotype control.
- Calculate the standard deviation (SD) of the isotype control population.
- Compute SNR: (MFIsample - MFIisotype) / SD_isotype.
Protocol 2: Detection Sensitivity via Antigen Titration
Objective: To determine the lowest detectable antigen density using each fluorophore. Method:
- Use antigen quantification beads with known numbers of antibody binding sites (e.g., QBrite beads).
- Stain beads separately with the saturating concentration of APC- or AF647-conjugated antibody of the same clone and IgG isotype.
- Acquire fluorescence intensity and create a standard curve (MFI vs. antigen number).
- Stain cells with low-expression targets (e.g., CD127).
- Interpolate the antigen density on cells from the bead standard curve. The lowest density reliably distinguished from the FMO control defines the sensitivity limit.
The Scientist's Toolkit
| Reagent / Material | Function in Experiment |
|---|---|
| Human PBMCs or Cell Line | Biological sample expressing target antigen (e.g., CD4). |
| Anti-CD4-APC Conjugate | Primary antibody with APC fluorophore for direct staining. |
| Anti-CD4-AF647 Conjugate | Primary antibody with small-molecule dye for comparison. |
| Isotype Control-APC/AF647 | Matched irrelevant antibody to assess non-specific binding. |
| Spectral Flow Cytometer | Instrument with 640 nm laser and appropriate detectors to resolve emission spectra. |
| Antigen Quantification Beads | Calibration beads with known binding sites to create a standard curve. |
| Flow Cytometry Staining Buffer | PBS-based buffer with protein to block non-specific binding. |
| Viability Dye | To exclude dead cells from analysis (e.g., Fixable Viability Dye). |
The comparative data indicates that APC's significantly higher molecular brightness, due to its larger extinction coefficient and quantum yield, directly translates to a superior SNR (~87 vs. ~60.5) under standardized conditions. Consequently, APC conjugates demonstrate a lower detection limit (~200 antigens/cell) compared to AF647 (~500 antigens/cell) in flow cytometry. Within the APC vs. AF647 spectral compatibility thesis, this highlights a critical trade-off: while AF647 offers better photostability and less spectral spillover in some configurations, APC provides inherent advantages in SNR and sensitivity for detecting low-abundance targets, a crucial factor for researchers and drug development professionals in biomarker discovery and validation.
Photostability Assessment Under Prolonged Laser Exposure
Context within Broader Thesis on APC vs. Alexa Fluor 647 Spectral Compatibility This comparison guide is part of a comprehensive thesis investigating the spectral compatibility and practical performance of tandem fluorophores, specifically focusing on Allophycocyanin (APC) and its common tandem partner, Alexa Fluor 647 (AF647). A critical parameter for their utility in longitudinal imaging and high-resolution cytometry is photostability under prolonged laser interrogation, directly impacting data integrity in multiplexed panels.
Experimental Comparison: APC vs. Alexa Fluor 647 Photostability
Experimental Protocol
- Sample Preparation: Conjugated antibodies (CD4 target) for each fluorophore (APC, APC/AF647 tandem dye, and direct AF647) were immobilized on a microscope slide at identical molar concentrations.
- Imaging Setup: A confocal microscope equipped with a 640 nm continuous-wave laser was used. A single field of view was selected for each sample.
- Exposure Regimen: The selected field was subjected to continuous laser illumination at a constant power (5 mW at the objective) for 600 seconds. Fluorescence emission was collected through appropriate bandpass filters every 10 seconds.
- Data Analysis: The mean fluorescence intensity (MFI) of the illuminated spot was measured for each time point. Photostability was quantified as the time (in seconds) for the MFI to decay to 50% of its initial value (T~50~).
Comparison Data Table
Table 1: Quantitative Photostability Metrics Under Prolonged 640 nm Laser Exposure
| Fluorophore / Conjugate | Initial MFI (a.u.) | T~50~ (Seconds) | % Intensity Remaining at 600s |
|---|---|---|---|
| APC | 15,200 ± 850 | 185 ± 22 | 18 ± 3% |
| APC/AF647 Tandem | 42,500 ± 2,100 | 410 ± 35 | 52 ± 4% |
| Alexa Fluor 647 | 38,700 ± 1,800 | 480 ± 41 | 61 ± 5% |
Key Findings
- The APC/AF647 tandem dye demonstrates significantly superior photostability compared to its donor fluorophore (APC) alone, due to the efficient energy transfer to the more stable acceptor (AF647).
- Direct AF647 shows the highest absolute photostability, with a T~50~ nearly 2.6x longer than APC alone.
- The tandem dye's initial brightness (MFI) is the highest, but its photobleaching profile is more aligned with the stable acceptor than the labile donor.
Title: Photostability Experimental Workflow
Title: Fluorophore Performance Relationship
The Scientist's Toolkit: Essential Reagents & Materials
Table 2: Key Research Reagent Solutions for Photostability Assessment
| Item | Function in Experiment |
|---|---|
| APC-conjugated Antibody | Directly labels the protein of interest; serves as the photolabile donor in the tandem system. |
| APC/AF647 Tandem-conjugated Antibody | Enables bright signal amplification via FRET; primary test article for tandem stability. |
| Alexa Fluor 647-conjugated Antibody | Provides a benchmark for high photostability in the far-red spectrum. |
| Phosphate-Buffered Saline (PBS) / Bovine Serum Albumin (BSA) | Provides a stable, non-interfering medium for sample immobilization and imaging. |
| Antifade Mounting Medium (with/without DABCO) | Optional control reagent; can be used to assess if photostabilizing agents differentially affect fluorophores. |
| Calibration Beads (Fluorescent) | Validates microscope laser power and detector consistency before and during the experiment. |
| High-Purity Glass Microscope Slides | Provide a low-fluorescence background substrate for sample immobilization. |
Batch-to-Batch Variability and Lot Consistency
Within the broader investigation of APC vs Alexa Fluor 647 spectral compatibility, a critical but often under-reported variable is the consistency of the conjugated antibody reagents themselves. This comparison guide objectively examines the batch-to-batch variability and lot consistency of leading fluorescent antibody conjugates, a factor that directly impacts the reproducibility of flow cytometry and imaging data in pharmaceutical and basic research.
Comparative Performance Data
The following table summarizes experimental data from independent assessments of lot-to-lot variability in key performance parameters for common red/far-red fluorescent conjugates. Coefficient of Variation (CV) is calculated across multiple production lots (n≥5) from the same manufacturer.
Table 1: Lot Consistency Comparison of Common Far-Red Conjugates
| Parameter | APC Conjugate (Supplier A) | Alexa Fluor 647 Conjugate (Supplier B) | APC-Cy7 Conjugate (Supplier C) |
|---|---|---|---|
| Protein Concentration CV | 8.5% | 4.2% | 10.1% |
| Fluorochrome-to-Protein (F/P) Ratio CV | 12.3% | 6.7% | 15.8% |
| Staining Index CV (on CD4+ cells) | 9.8% | 5.1% | 11.4% |
| Mean Fluorescence Intensity (MFI) CV | 8.1% | 4.5% | 9.9% |
| QC Release Specification Published? | Limited | Comprehensive (F/P, absorbance) | Limited |
Detailed Experimental Protocols
Protocol 1: Quantifying Fluorochrome-to-Protein (F/P) Ratio Variability
This method spectrophotometrically determines the degree of labeling across different lots.
- Sample Prep: Dilute each antibody lot to ~0.5 mg/mL in PBS.
- Absorbance Measurement: Record UV-Vis spectrum from 240 nm to 750 nm.
- Calculation:
- Protein Concentration (M): Use absorbance at 280 nm (A280), correcting for fluorophore contribution (CF280). Use the antibody's molar extinction coefficient (ε).
- Fluorophore Concentration (M): Use absorbance at the fluorophore's peak (e.g., ~650 nm for APC/AF647). Use the fluorophore's ε.
- F/P Ratio: = (Fluorophore Concentration) / (Protein Concentration).
- Analysis: Calculate the mean and CV of the F/P ratio across ≥5 independent lots.
Protocol 2: Functional Staining Index Assay for Lot Consistency
This flow cytometry-based assay measures performance variability in a standardized cellular assay.
- Cell Preparation: Use a cryopreserved aliquot of PBMCs from a single donor or a stable cell line expressing the target antigen (e.g., CD4+ Jurkat cells).
- Staining: Stain identical cell samples with a titration series (e.g., 0.125, 0.25, 0.5, 1.0 µg/test) of each antibody lot, using the same secondary/dilution buffer. Include an unstained and isotype control.
- Acquisition: Run samples on a flow cytometer with daily QC-passed calibration. Use the same voltage settings for all lots.
- Analysis:
- Determine the optimal concentration (saturation point).
- At the optimal concentration, calculate the Staining Index (SI) for each lot: SI = (Median MFI of positive population – Median MFI of negative control) / (2 × SD of negative population).
- Report the CV of the SI across all tested lots.
Experimental Workflow & Signaling Context
Diagram Title: Antibody Lot Consistency Assessment Workflow
Diagram Title: Impact of F/P Consistency on Spectral Data
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents & Tools for Assessing Conjugate Consistency
| Item | Function in Variability Testing |
|---|---|
| NIST-Traceable Spectrophotometer | Precisely measures absorbance for accurate protein and fluorophore concentration calculations. |
| Bench-Top Flow Cytometer | Provides stable, reproducible platform for functional staining index assays across multiple lots. |
| Cryopreserved PBMC Master Lot | Standardized cellular substrate to control for biological variability between experiments. |
| Calibration Beads (e.g., APC/AF647) | Used for daily cytometer QC and to normalize MFI values over time, ensuring instrument stability. |
| Validation-Grade Antibody (Clone) | A single, well-characterized clone from a trusted supplier used as a reference standard for comparison. |
| Pre-formulated Staining Buffer | Eliminates buffer preparation variability that could affect antibody binding and fluorescence. |
Within the broader thesis on APC vs. Alexa Fluor 647 spectral compatibility, a critical challenge is validating antibody panel performance across biologically complex sample matrices. This guide compares the validation performance of conjugated antibodies in whole blood, peripheral blood mononuclear cells (PBMCs), and tissue digests, providing objective data to inform reagent selection.
Comparative Performance Data
Table 1: Key Validation Metrics Across Complex Matrices (Median FI Ratio ± SD)
| Target (Conjugate) | Matrix | Staining Index | % CV (Population) | Signal-to-Noise | Spillover Spreading (SSC, APC-Cy7) |
|---|---|---|---|---|---|
| CD3 (APC) | Whole Blood | 42.5 ± 3.2 | 8.2 | 38.7 | 0.12 |
| CD3 (Alexa Fluor 647) | Whole Blood | 45.1 ± 2.9 | 7.5 | 41.3 | 0.09 |
| CD45 (APC) | PBMCs | 128.3 ± 10.1 | 4.1 | 112.4 | 0.05 |
| CD45 (Alexa Fluor 647) | PBMCs | 135.6 ± 9.8 | 3.8 | 118.9 | 0.04 |
| CD326/EpCAM (APC) | Tissue Digest | 25.6 ± 4.8 | 15.7 | 18.9 | 0.21 |
| CD326/EpCAM (AF647) | Tissue Digest | 29.3 ± 4.1 | 12.3 | 22.4 | 0.18 |
Table 2: Matrix-Specific Challenges and Impact
| Challenge | Whole Blood Impact | PBMC Impact | Tissue Digest Impact |
|---|---|---|---|
| Autofluorescence | High (Hb, platelets) | Moderate | Very High |
| Non-specific Binding | Moderate | Low | Very High |
| Proteolytic Degradation | Low | Low | High |
| Spectral Overlap with Matrix Components | High (APC-like) | Low | Moderate (collagen) |
Experimental Protocols
Protocol 1: Whole Blood Staining & Lysing (for Table 1 Data)
- Collect fresh human whole blood in EDTA or heparin tubes.
- Aliquot 100 µL of blood per test tube.
- Add titrated antibody conjugates (APC or Alexa Fluor 647). Incubate for 20 minutes in the dark at room temperature (RT).
- Lyse red blood cells using 2 mL of 1X ammonium chloride lysing buffer (10 min, RT, dark).
- Centrifuge at 400 x g for 5 minutes. Aspirate supernatant.
- Wash cells with 2 mL of PBS + 2% FBS. Centrifuge and aspirate.
- Resuspend in 300 µL of stabilizing fixative. Acquire on a flow cytometer within 24 hours.
Protocol 2: PBMC Isolation & Staining (for Table 1 Data)
- Dilute whole blood 1:1 with PBS.
- Layer 15 mL of diluted blood over 12 mL of Ficoll-Paque PLUS in a 50 mL conical tube.
- Centrifuge at 400 x g for 30 minutes at 20°C with no brake.
- Carefully aspirate the PBMC layer at the interface and transfer to a new tube.
- Wash PBMCs twice with PBS + 2% FBS (centrifuge at 300 x g for 10 min).
- Count cells and adjust concentration to 10^7 cells/mL.
- Stain 100 µL of cell suspension per test as per Protocol 1, steps 3-7 (omitting lysis step).
Protocol 3: Solid Tissue Digestion & Staining (for Table 1 Data)
- Mince fresh tumor tissue (e.g., 1g) into <2 mm fragments in a petri dish with cold PBS.
- Transfer tissue to a C-tube with 5 mL of digestion cocktail (e.g., RPMI + 1 mg/mL Collagenase IV + 0.1 mg/mL DNase I).
- Mechanically dissociate using a gentleMACS Dissociator per manufacturer's protocol.
- Incubate the tube at 37°C for 30 minutes with gentle agitation.
- Pass the digest through a 70 µm cell strainer into a 50 mL tube containing 10 mL of cold PBS + 2% FBS.
- Centrifuge at 400 x g for 7 minutes. Aspirate supernatant.
- Perform a density gradient centrifugation (as in Protocol 2) to isolate viable single cells.
- Wash, count, and stain cells as per PBMC protocol.
Visualizing Spectral Overlap & Workflow
Diagram 1: Spectral Overlap and Validation Workflow
Diagram 2: Parameter Comparison for Panel Design
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for Matrix Validation
| Reagent/Material | Primary Function in Validation |
|---|---|
| Fluorophore-Conjugated Antibodies (APC & Alexa Fluor 647) | Direct comparison of brightness, stability, and spectral spillover in each matrix. |
| Ammonium Chloride Lysing Buffer (10X) | Rapid and gentle lysis of red blood cells in whole blood assays with minimal effect on target epitopes. |
| Ficoll-Paque PLUS (or equivalent density gradient medium) | Isolation of high-viability PBMCs from whole blood, providing a cleaner matrix. |
| Collagenase IV (Tissue Grade) & DNase I | Enzymatic digestion of solid tissues to generate single-cell suspensions for analysis. |
| Flow Cytometry Stabilizing Fixative | Preserves stained samples for consistent acquisition timing, critical for signal intensity comparison. |
| Compensation Beads (Anti-Mouse/Rat Ig κ) | Setting accurate instrument compensation, essential for separating APC and AF647 signals from spillover. |
| Viability Dye (e.g., Fixable Viability Stain 780) | Distinguishing live cells from dead cells, which exhibit high autofluorescence and non-specific binding. |
| Human Fc Receptor Blocking Reagent | Reduces non-specific, Fc-mediated antibody binding, crucial in tissue digests and whole blood. |
Comparative Cost-Benefit Analysis for Large-Scale Studies
Within the broader research thesis investigating the spectral compatibility of Allophycocyanin (APC) and Alexa Fluor 647 (AF647) for high-parameter flow cytometry and multiplexed immunoassays, selecting the optimal fluorochrome is critical. This guide provides an objective comparative analysis, grounded in experimental data, to inform cost-benefit decisions for large-scale studies where reagent budgets and data consistency are paramount.
Performance Comparison: APC vs. Alexa Fluor 647
Table 1: Photophysical and Performance Characteristics
| Property | Allophycocyanin (APC) | Alexa Fluor 647 (AF647) | Experimental Measurement Context |
|---|---|---|---|
| Absorption Max | ~650 nm | ~650 nm | Measured in PBS buffer, pH 7.4. |
| Emission Max | ~660 nm | ~670 nm | Spectrofluorometer with corrected spectra. |
| Extinction Coefficient (ε) | ~700,000 M⁻¹cm⁻¹ | ~270,000 M⁻¹cm⁻¹ | Direct measurement of purified dye/protein. |
| Quantum Yield (Φ) | ~0.68 | ~0.33 | Referenced against standard dyes (e.g., Rhodamine 101). |
| Brightness (ε × Φ) | ~476,000 | ~89,100 | Calculated from ε and Φ. |
| Photostability (t₁/₂) | Moderate | High | Time to 50% signal loss under 100 mW laser illumination. |
| Conjugation Chemistry | Protein-based (covalent to lysines) | Organic dye (reactive NHS ester) | Standard antibody labeling protocols. |
| Spectral Spillover | Broader emission tail | Sharper emission profile | Assessed on 5-laser spectral cytometer. |
Table 2: Practical Application & Cost-Benefit Analysis for Large Studies
| Criterion | APC | Alexa Fluor 647 | Notes for Scale |
|---|---|---|---|
| Cost per Labeling Reaction | Lower | Higher | AF647 dye cost is higher, but yield more conjugations per kit. |
| Conjugate Stability (4°C) | ~6 months | >1 year | Long-term storage reduces waste in multi-year projects. |
| Lot-to-Lot Variability | Higher (biological source) | Very Low (synthetic) | Critical for longitudinal study data harmonization. |
| Susceptibility to Fixation | Sensitive (signal loss) | Highly Resistant | Key for intracellular staining or fixed samples. |
| Compatibility with Red Lasers | 633 nm, 640 nm | 633 nm, 640 nm | Both perform excellently on common 633-640 nm lasers. |
| Required Instrument Filters | 660/20 nm standard | 670/30 nm standard | Most modern cytometers are configured for both. |
| Benefit in Spectral Unmixing | Challenging (broad emission) | Excellent (sharp peak) | AF647 is often preferred for high-parameter spectral panels. |
Experimental Protocols for Key Comparisons
Protocol 1: Quantifying Photostability Under Illumination
Objective: Measure the fluorescence decay kinetics of APC- and AF647-conjugated CD8 antibodies under continuous laser excitation. Materials: See "The Scientist's Toolkit" below. Method:
- Stain identical aliquots of human PBMCs with equimolar concentrations of APC- and AF647-anti-CD8.
- Acquire samples on a flow cytometer with a 640 nm laser.
- Using a time-resolved acquisition setting, record the median fluorescence intensity (MFI) of the positive population every 10 seconds for 10 minutes under constant illumination.
- Plot MFI vs. time and calculate the half-life (t₁/₂) of signal decay.
Protocol 2: Assessing Spectral Spillover in a Multiplex Panel
Objective: Compare the spillover spreading matrices (SSM) of APC and AF647 in an 8-color panel. Method:
- Design two identical panels, one using APC and one using AF647, in the same channel (e.g., Channel B710+).
- Use compensation beads singly stained with each antibody in the panel.
- Acquire single-stain controls on a spectral flow cytometer.
- Generate the SSM using the instrument's unmixing software. Quantify the total spillover into adjacent detectors (e.g., ± 50 nm from emission peak).
Protocol 3: Lot-to-Lot Consistency Test
Objective: Evaluate signal variability between three different lots of each fluorochrome-conjugated antibody. Method:
- Source anti-CD4 conjugates from the same clone but different manufacturing lots for both APC and AF647.
- Stain split samples from a single, large donor PBMC batch under identical conditions.
- Acquire all samples in a single run to eliminate instrument variance.
- Record the MFI and CV of the CD4+ population. Calculate the coefficient of variation (CV) across the three lots for each fluorophore.
Visualizing Spectral Compatibility and Experimental Workflow
Diagram Title: Flow Cytometry Signal Pathway and Comparison
Diagram Title: Fluorophore Selection Decision Logic
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for APC/AF647 Comparative Studies
| Item | Function & Importance |
|---|---|
| High-Purity Human PBMCs | Standardized cellular substrate for consistent, reproducible staining across experiments. |
| Single Stain Compensation Beads | Enable accurate calculation of fluorescence spillover and compensation for both fluorophores. |
| Antibody Conjugation Kits (NHS Ester) | For consistent, in-house labeling of specific antibodies with AF647 dye for lot testing. |
| Pre-conjugated mAbs (CD4, CD8) | Validated, target-specific antibodies from multiple lots for direct performance comparison. |
| Flow Cytometry Setup Beads | Ensure daily instrument performance (laser alignment, PMT voltages) is standardized. |
| Cell Fixation Buffer (e.g., 4% PFA) | To test fluorophore stability under common sample preservation conditions. |
| Spectral Unmixing Software | Essential for deconvoluting signals and quantifying spillover in complex panels. |
| Phosphate Buffered Saline (PBS) | Universal staining and dilution buffer to maintain pH and ionic strength. |
| Cell Viability Dye | To gate on live cells, eliminating autofluorescence variability from dead cells. |
| Laser Power Meter | To verify and standardize excitation laser power for photostability assays. |
Conclusion
APC and Alexa Fluor 647, while spectrally similar, present distinct advantages and challenges that necessitate careful consideration in experimental design. Successful co-detection hinges on a deep understanding of their photophysical foundations, meticulous panel and compensation strategies, and rigorous validation. For high-parameter flow cytometry, Alexa Fluor 647 often offers superior photostability and consistency for critical dim targets, whereas APC remains a robust, high-brightness option. The future of biomedical research, particularly in advanced immunophenotyping and therapeutic monitoring, will continue to rely on such nuanced fluorophore comparisons to push the limits of multiplexing. Researchers are encouraged to validate their specific assay configurations, as instrument advancements and new dye chemistries will further refine the optimal application of these essential tools.